Using Probiotics to Help Patients be Proactive
Novel approach can enable the prevention of root caries in a periodontal geriatric population.
By Gregory E. Oxford, DDS, MS, PhD
As the US population ages, people are becoming increasingly health conscious, and, similarly, a greater percentage of patients are keeping more teeth. The desire to remain fit as people age has led to an increasing desire to maintain a healthy, functional dentition. The major cause of tooth loss for adults is from periodontal diseases. Therefore, teeth retention for a large portion of adults may require some periodontal therapy. A common result of periodontal therapies is gingival recession and, ultimately, root exposure. Additionally, as people age and become less self-reliant, their oral hygiene measures typically diminish and the likelihood of developing root caries increases. Age- or medication-induced xerostomia also diminishes the innate ability of saliva’s protective response. Root surfaces are uniquely more susceptible to caries as they are more porous and likely to develop biofilms and, ultimately, dental caries. Streptococcus mutans is the known acidogenic etiologic pathogen in dental caries. Consequently, myriad approaches have been suggested to prevent these exposed roots from developing dental caries. Recently, a new oral probiotic entered the marketplace that can uniquely help prevent root surface decay. By continually inoculating the oral cavity with probiotic bacteria that out-compete naturally occurring S. mutans, an environment is created that combats the development of dental caries.
Root Surface Susceptibility
The average age of the US population has generally been increasing over the past few decades. Americans are also becoming increasingly more concerned about maintaining their health as they age. Consequently, dental health and function have kept pace. Individuals in most countries have more teeth, according to decayed, missing and filled indices (DMFT), than in years past.1 Accordingly, more effort and resources have been allocated for teeth maintenance.2
The media has also played an important role in promoting individuals of all age groups to have a beautiful, healthy smile, which has added to the demand for dental services and maintenance of teeth. Nonetheless, the major reason for tooth loss in the adult population continues to be periodontal diseases.3 Periodontal diseases are a group of bacterial infections leading to inflammatory changes in the tissues around teeth, ultimately resulting in loss of supporting bone and, if left untreated, loss of teeth.
A variety of treatment methods are used to help combat periodontal diseases. Once periodontitis, or loss of attachment to the teeth, has occurred, most treatment remedies result in recession of the gingival tissues,4 even if only non-surgical modalities are used. The recession may be more pronounced if periodontal surgery is needed to control the disease. Most cases of periodontal therapy result in exposure of the root surfaces.
Although both crown and root surfaces are ectodermally derived from Hertwig’s epithelial root sheath, they are structurally and functionally different.5 The coronal surfaces are covered with enamel, a hard surface designed to withstand the masticatory role. Root surfaces were embryologically evolved to aid in attachment of the tooth to the bony housing. Root surfaces are covered with an ectodermally derived layer of cementum that overlies the root and is very porous. The root surface, ideally, is never exposed to the oral cavity. Through disease, trauma (including tooth brushing or the controlled trauma of orthodontic care), or treatment, recession of gingival tissues may occur and lead to exposure of the root surface. Because the cementum or underlying dentinal surface is porous, it is more susceptible to biofilm development and plaque than is the enamel surface, thus predisposing the root surface to greater susceptibility to caries than enamel surfaces.
The development of a biofilm on a tooth surface begins to occur shortly after cleaning. The biofilm is the matrix into which plaque can then attach. It has been known for years that S. mutans is the etiologic factor of dental caries.6 S. mutans can produce large amounts of lactic acid through a fermentative process that reduces the pH in the micro-environment of the tooth or root surface, leading to dissolution of the mineral phase of enamel or dentin surfaces until clinically visible cavitation is seen. Plaque samples obtained from cavitated exposed root surfaces have a greater concentration of cariogenic S. mutans than that found in smooth surface lesions.7
Traditional Approaches
Dental caries continues to be a worldwide problem.8 The most common method of addressing this problem is the prevention of decay via fluoridation of water, which has led to a marked reduction of dental caries in western industrialized countries.9 Fluoridated toothpaste, fluoride gels, and electric toothbrushes have also been employed to help control dental caries, yet it remains a common malady.
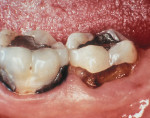
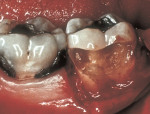
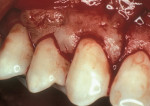
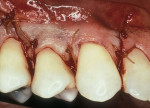
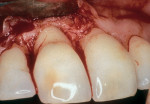
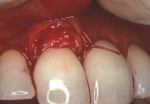
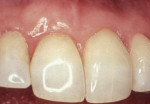
As people maintain more teeth for longer periods of time, an increased likelihood of root exposure through disease, traumatic factors, or extensive care exists and, thus, an increased prevalence for root surface decay. A common difficulty in addressing carious root surfaces is that current bonding materials for most restorative applications are not well suited for dentinal surfaces, especially when compared to that of enamel (Figure 1, Figure 2 and Figure 3).
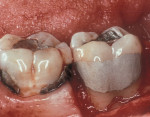
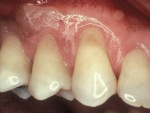
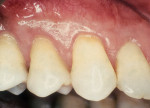
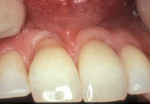
Another method of addressing exposed root surfaces is to prevent their prolonged exposure and, thus, limit their susceptibility to dental caries with soft-tissue grafts to replace the recessed gingival tissues. This is most frequently indicated when there is a minimal amount of keratinized tissue present and no significant loss of attachment has occurred. Root coverage procedures for a pure recession lesion may restore the clinical environment to cover the root and, thus, reduce the susceptibility to root surface caries (Figure 4, Figure 5, Figure 6 and Figure 7). Root coverage procedures employ a variety of tissues, including autogenous grafts with epithelial or connective tissue (Figure 8, Figure 9, Figure 10 and Figure 11) or allografts.
Novel Approach
A new approach to dental caries prevention is on the horizon: probiotics. Probiotics are live microorganisms that confer a health benefit when administered in sufficient amounts. The beneficial bacteria in a probiotic formulation can help maintain a healthy balance of bacteria. Examples of common probiotic applications are yogurt containing live cultures, acidophilus capsules to improve digestion, and products for improved immune system and vaginal and urinary tract health. Some groups have suggested an oral benefit from taking a lactobacillus strain isolated from the gut.10 It is well documented in probiotics literature that the best benefits come from taking probiotics that are of human origin and that are natural colonizers of the site where a benefit is desired.
Oral probiotics are a spinoff from the medical community. A recently marketed oral probiotic, EvoraPlus™, employs three bacterial strains (ProBiora3™) that normally colonize the mouth and have been isolated from orally healthy humans. These strains are different from those isolated from the gut, which do not naturally colonize the oral cavity and have a short-lived benefit.
ProBiora3 is a blend of three naturally occurring strains of oral bacteria with documented oral health benefits. The three bacteria are Streptococcus oralis, Streptococcus uberis, and Streptococcus rattus, originally isolated from the oral cavity of healthy subjects. Repeated application of probiotic gut bacteria could actually disturb the natural balance of the oral flora. Additionally, probiotics for the gut are typically encapsulated to protect them during transit through the stomach, so they would not be available to attach in the mouth. Finally, it has been suggested that gut probiotics may elicit a beneficial systemic immune response that could positively influence oral health; however, this idea is highly controversial and not scientifically well grounded.
The scientific basis for the oral health benefits provided by the three strains of bacteria isolated from the oral cavity in ProBiora3 has been documented over the past 30 years. This unique blend of bacteria has the ability to suppress the levels of S. mutans, the principal etiologic agent of tooth decay. S. rattus is a very close relative of S. mutans, and until recently was considered a subspecies of S. mutans. A completely natural strain of S. rattus (JH145) that does not make lactic acid from metabolized sugar has been isolated.11 The genetic defect in JH145 was identified as a spontaneous deletion mutation in the structural gene for the enzyme lactate dehydrogenase, preventing this strain from making lactic acid and rendering it incapable of causing tooth decay. The inclusion on S. rattus, which a patient inoculates repeatedly with daily applications of the probiotic, helps prohibit the development of dental caries.
Additionally, a strong inverse relationship has been established between the presence of certain species of viridans streptococci and the bacteria that cause most types of periodontal disease. In a healthy periodontal site, S. oralis (previously called S. sanguis type II) and S. uberis are commonly found in significant amounts, while levels of periodontal pathogens including Tannerella forsythensis, Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Prevotella intermedia, Peptostreptococcus micros, Campylobacter rectus, and Prevotella melaninogenica are quite low. The opposite situation prevails in disease sites where S. oralis and S. uberis are usually undetectable. Furthermore, it has been demonstrated that S. oralis and S. uberis inhibit the growth of periodontal pathogens by producing hydrogen peroxide. This production of hydrogen peroxide was demonstrated to be sufficient to produce a substantial whitening effect on tea- and chlorhexidine-stained ceramic disks. Additionally, they also oxidize volatile sulfur compounds (VSC) responsible for bad breath.12
ProBiora3 is available in four over-the-counter formulations, including versions for adults, children, and pets, as well as a professional-strength concentration (Evora plus™, Evora kids™, Teddy’s Pride, and Evora pro™, respectively). The human probiotics are provided as flavored tablets intended for twice-daily use by adults and children after brushing their teeth.
Clinical Application
For the past 24 months, the author has been prescribing Evora plus™ (Figure 12) to elderly patients, institutionalized patients, those with excessive areas of root surface exposure, those with a documented high caries incidence, and patients who have physical and dexterity limitations. Although no significant longitudinal data exists, the authors’ clinical impressions have been overwhelmingly convincing. The incidence of root surface caries has diminished if not eradicated in a portion of the authors’ population. As always, compliance is an issue. Empirically, patients who have a history of decay-related concerns, are self-pay, and are not institutionalized are most compliant and benefit the greatest. The authors are attempting to incorporate the administration of this oral probiotic into their institutionalized population and include it in their patients’ formularies, although it is available in several national pharmacies.
Conclusion
Root surface caries is a problem in patients with exposed root surfaces. Prevention of root surface caries continues to be the mainstay of clinical approaches to this problem rather than traditional methods of addressing caries after it has developed. A new probiotic has been developed that reduces the likelihood of caries development. The ease of application makes this an excellent tool in the clinician’s arsenal to combat caries of all types.
A population that could benefit tremendously from this new approach to caries prevention is the highly susceptible youth and adolescents. These individuals have recently been treated largely with pharmaceutical devices (such as chlorhexidine gluconate and fluoride gels) that have significant side effects such as staining and floral alterations. Alternatively, two of the bacteria in the ProBiora3 formulation (S. oralis and S. uberis) produce hydrogen peroxide sufficient to produce a substantial whitening effect. Thus, daily inoculations with these probiotics seem to be an appropriate approach for these young individuals. Similarly, probiotics could be well employed in patients who have started orthodontic care and may have limited access to effective oral hygiene measures due to their orthodontic appliances such as brackets and bands.
Clearly, though, the groups that may benefit the most are elderly patients or mentally or physically handicapped patients who are limited in their ability to perform even the most rudimentary oral hygiene procedures. The elderly may also be compromised by age- or medication-induced xerostomia. With this decrease in salivary flow, these patients have a reduced innate protective and defense mechanism against caries and, thus, have an increased risk of developing caries, especially root surface caries. They, therefore, could benefit greatly from this novel new approach to preventing decay.
References
1. Campus G, Sacco G, Cagetti M, Abati S. Changing trend of caries from 1989 to 2004 among 12-year old Sardinian children. BMC Public Health. 2007;Mar(7):28.
2. Ferguson DA, Steinberg BJ, Schwien T. Dental economics and the aging population. Compend Contin Educ Dent. 2010; 31(6):418-425.
3. Martin JA, Page RC, Loeb CF, Levi PA Jr. Tooth loss in 776 treated periodontal patients. J Periodontol. 2010;81(2):244-250.
4. Sarfati A, Bourgeois D, Katsahian S, et al. Risk assessment for buccal gingival recession defects in an adult population. J Periodontol. 2010;81(10):1419-1425.
5. Bhaskar SN. Development and growth of teeth. In: Orban’s Oral Histology and Embryology. 8th ed. St. Louis, MO: Mosby; 1976:37-43.
6. Gibbons RJ, van Houte J. Dental caries. Annu Rev Med. 1975;26:121-136.
7. Takahashi N, Nyvad B. Caries ecology revised: microbial dynamics and the caries process. Caries Res. 2008;42(6):409-418.
8. Edelstein BL. Pediatric caries worldwide: implications for oral hygiene products. Compend Contin Educ Dent. 2005;26(5 suppl 1):4-9.
9. Bratthall D. Estimation of global DMFT for 12-year-olds in 2004. Int Dent J. 2005; 55(6):370-372.
10. Caglar E, Cilder SK, Ergeneli S, et al. Salivary mutans streptococci and lactobacilli levels after ingestion of probiotic bacterium Lactobacillus reuteri ATCC 55739 by straws or tablets. Acta Odontol Scand. 2006;64(5):314-318.
11. Hillman JD, McDonell E, Cramm T, et al. A spontaneous lactate dehydrogenase deficient mutant of Streptococcus rattus for use as a probiotic in the prevention of dental caries. J Appl Microbiol. 2009;107(5):1551-1558.
12. Kang MS, Kim BG, Chung J. Inhibitory effect of Weissella cibaria isolates on the production of volatile sulfur compounds. J Clin Periodontol. 2006;33(3):226-232.
About the Author
Gregory E. Oxford, DDS, MS, PhD
Private Practice
St. Augustine, Florida
Courtesy Professor of Graduate Periodontics
University of Florida
College of Dentistry
Gainesville, Florida