Avoiding Endodontic Misadventure
Timeless, state-of-the-art strategies for preventing adverse events
Iatrogenic events in endodontics can be grouped into four categories: perforations; canal blockages and transportations; separated instruments and metallic fragments; and extrusion (of various filling materials, medicaments, and media) beyond the canal system/apex. These events can be prevented—or at least minimized—through careful preoperative case assessment and risk evaluation. Prevention is always better than repair, assuming repair is even possible. This article will describe the causes of the common iatrogenic events that may occur during endodontic treatment and provide the general dentist with time-honored, evidence-based clinical strategies for avoiding these complications.
Risk Factors for Latrogenic Events
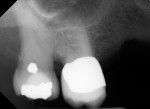
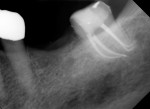
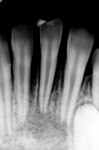
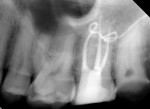
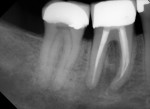
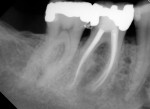
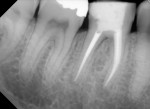
Several different risk factors may converge to increase the likelihood of an iatrogenic event. The primary risk factors can be traced to the patient, the tooth, or the clinician—and in some cases, a combination of these (Figure 1 through Figure 6).
Patient-Related Factors
Risk factors in this category may be behavioral or physical in nature. Patient-related risk factors include having limited ability to open the mouth; being unwilling or unable to hold reasonably still in the chair or to be reclined for a long period of time; exhibiting a fear of needles, dental phobia in general, and/or other psychological challenges; having a history of anesthesia difficulty, especially in the lower molars; having a need to swallow frequently; and having a sensitive gag reflex.
Tooth-Related Factors
There are numerous anatomic complexities that may make endodontic treatment difficult. Some of the tooth-related risk factors for an iatrogenic event include rotated and tipped teeth; high furcations; internal or external root resorption; unusually long or short or severely curved roots; atypical root numbers; roots lying over the mandibular canal and/or other vital anatomic structures; teeth with previous endodontic treatment, especially if an iatrogenic event occurred; immature apices; trauma; atypical canal numbers and location; calcified canals of all types; and existing crowns and bridges in the way of the preferred access location, among other factors.
As a side note, analyzing the tooth anatomy, root structure, canal anatomy, and the position of the tooth in the arch is essential. Cone-beam technology is the gold standard for preoperatively determining the anatomy. The second best option is to take multiple radiographs from different angles, such as mesial, distal, buccal, and straight on. Such visualization can illustrate any anatomical complexity and, more importantly, tell the clinician whether the case should be referred or what should be done to address the complexities that are present.
For example, in the case of a narrow, “high” furcation, if the canals are not located where they were initially expected (ie, in a horizontal, vertical, and buccal-to-lingual dimension), the clinician must stop and take a radiograph to realign the access location. Additionally, perforation and/or canal location can be especially problematic if a crown obscures the pulp chamber. The use of a surgical operating microscope (SOM) can be invaluable in such cases. Diligent and conservative removal of tooth structure under the SOM can avoid “hollowing out” the cervical third of the tooth and predisposing the tooth to subsequent vertical root and furcal floor fracture.
Clinician-Related Factors
Finally, there are several clinician-related factors that can contribute to difficulties with a case. These factors include the level of the clinician’s experience and training; the selection and/or use of radiography and instrumentation (eg, hand and/or rotary file system); and equipment factors, such inadequate or poorly organized equipment (eg, endodontic motor, heat source, and obturation device) for the task at hand.
Importance of Treatment Planning
Of the three types of factors, the author believes it is the clinician’s treatment planning skill that is the central predictor of case outcome. If the clinician properly identifies the risks, makes a plan to avoid iatrogenic events, and carries out the plan, clinical success is optimized. The astute clinician fully informs the patient about the procedure, including a discussion of alternatives and risks, and answers the patient’s questions completely. The second worse thing to happen after an iatrogenic event is for one to happen in a patient who has no idea that the procedure was a risk for complication in the first place. Seldom if ever is an uninformed patient prepared to be referred for the repair of an iatrogenic event or for the expense of that repair, and they are certainly not prepared, in the worst-case scenario, for the ramifications of losing the tooth entirely.
Treating any tooth with the belief that it can be referred for surgery or retreatment later is rarely productive, or is it in the best interest of any of the parties involved. There is much more value to carefully evaluating every clinical case for the patient-related, tooth-related, and clinician-related risk factors that make the achievement of an excellent outcome probable or improbable. Many endodontists will, at times, pass on certain clinical cases once they are recognized for their poor prognosis (eg, vertical fractures, severe resorption, unclear symptoms, nonrestorable cases, severe calcification with no visible canal, lack of true consent, etc). In essence, the value of knowing “when to say when” cannot be overstated.1-6
Aside from recognizing iatrogenic risk factors, once the treatment begins, the clinician must chose the appropriate path yet be prepared to make course corrections midstream when both expected and unexpected clinical challenges emerge. While there are an infinite number of possible scenarios, below are three common clinical examples.
Profound anesthesia. If the clinician cannot achieve profound anesthesia rapidly with common techniques, should they apply more local anesthesia using the same methods hoping for a different outcome, or should they immediately switch to an intraosseous technique? Most generally, they should switch to an intraosseous technique if clinically appropriate. Moving forward in a case without profound anesthesia is the harbinger of excessive tooth removal and apical transportation.
Canal access. If the canals are not where they were expected to be clinically, should the clinician keep searching by moving in the same direction, or should he or she stop and take a radiograph? Almost always, it is appropriate to stop and take a radiograph to determine where the canal is in relation to the removal of dentin at that stage. In a number of clinical scenarios, not taking a radiograph makes for an extreme risk of perforation.
Canal blockages. If a canal was patent initially but it is not now, what is the next step? Using a precurved small K file or stiff file, such as MANI K files or D Finders (MANI, Inc., www.mani.co.jp) can help to determine if the canal is truly blocked with canal debris or possibly a separated instrument. Taking a radiograph has great value. In this scenario, forcing large instruments to length in an attempt to break through a blockage or make apical progress is unlikely to be productive and more likely to cause additional iatrogenic harm.
From these examples, it should be clear that at any given moment during treatment there is one best forward action, whether it be referral or clinical action to advance the case, that will take the clinician to the desired outcome. Stopping when needed, assessing the situation, knowing what your strengths and liabilities are as the clinician, and considering the various courses of action can often mean the difference between a successful outcome and the creation of an iatrogenic event.
While no list of strategies is exhaustive, there are several key strategies for the prevention of the most common iatrogenic events (eg, perforations, canal blockages and transporations, separated instruments, and extrusion of filling materials beyond the canal system). These are discussed below.
Perforation
Perforation is always avoidable. To avoid it, recognizing the risk (eg, thin root walls, severe calcification, limited opening, rotated and tipped teeth, etc) and excellent tactile and visual control over the tooth is essential.
Generally, perforations are categorized by location, size, and the time since the perforation occurred. The larger the perforation, the longer it has remained unsealed, and the closer it is to the cervical region of the tooth, the worse the prognosis. Alternatively, a small perforation that is recent, has remained sealed to the oral environment, and is in the apical region can often be sealed with mineral trioxide aggregate or possibly apical surgery as indicated, depending on a number of clinical factors that include root and canal anatomy.
If the canal is not where it should be or is expected to be based on the radiographs, the clinician should stop immediately and take a radiograph. Strip perforations are a special case (related to size, extrusion of a foreign body beyond the strip, and the quality of the coronal seal since the perforation occurred). The management of virtually all perforations is a specialist procedure using the SOM.
Removing all caries, existing restorations (especially crowns and bridges) and bases and pins, etc, where indicated (ie, “clearing the tooth”) can prevent perforations. Mapping the pulpal floor after all such restorations have been removed is also beneficial. Clearing the tooth can guide the clinician in his or her assessment of whether the tooth is restorable because crowns often obscure caries and fractures that may influence the tooth’s prognosis.
Using specially designed burs for canal location (eg, MI Stainless, MANI, Inc.; Munce Discovery Burs™, CJM Engineering, Inc., www.cjmengineering.com) and ultrasonics where appropriate is far preferable for canal location than using a high-speed bur that is too large for the given indication. That said, a high-speed, surgical-length, 28-mm #2 bur (MANI, Inc.), when used under a SOM, is faster and just as precise in dentin removal when it is used appropriately. Whatever the means for canal location in highly calcified cases, caution and clinical judgment are always advised.
To avoid strip perforation, the clinician must first visualize the root curvature, calcification, and dimensions from mesial to distal, especially in younger patients who have canals that are initially larger and easily entered with Gates Glidden drills and orifice openers. Shaping the orifice too rapidly with Gates Glidden drills and nickel-titanium (NiTi) orifice openers can lead to excessive removal of tooth structure. It is important to note that .10 and .12 orifice openers and #5 and #6 Gates Glidden drills risk strip perforation especially below the coronal third. This can be avoided by determining the final prepared taper before beginning the procedure and never exceeding that taper in the initial orifice shaping. In the vast majority of cases, using a .08/25 orifice opener can avoid such over-enlargement.
Canal Blockages and Transportations
To avoid all kinds of canal blockages and canal transportations, there is no substitute for the diligent and progressive (ie, smaller to larger) use of stainless-steel hand files, especially before employing nickel-titanium (NiTi) instruments and/or Gates Glidden drills. This means using small hand files (such as the stiff MANI D Finders or K files) depending on the degree of calcification, starting with the smallest sizes (#6 and #8) and moving progressively to larger hand files until the canal is negotiated, a true working length is determined, and a glide path has been prepared. Blockages occur where access is not in a straight line, pulpal debris from the chamber is propelled apically, and large instruments are used too early and too forcefully in the canal negotiating sequence. The canal should be enlarged sequentially from its initial size to that of a minimum #15 to #20 hand file size before the “bulk” dentin removers (NiTi instruments) are used to shape the canal. Irrigation should be frequent and copious. Viscous ethylenediaminetetraacetic acid gel should be used in the presence of pulp in the chamber to avoid propelling pulpal fragments apically. The use of a reciprocating handpiece (eg, NSK ER-10, MounceEndo. https://mounceendo.com) is especially helpful in enlarging the canal slowly with hand files to prepare the glide path.
Fractured Instruments
There are three strategies to avoid the preventable clinical misadventure of fracturing NiTi instruments. First, the creation of a glide path using stainless-steel instruments allows subsequent NiTi instruments to flow down the canal with the least amount of torsion and cyclic fatigue. Second, the correct sequence and tactile control are essential. Generally, after the glide path is created, in complex cases the canal is shaped using the “step-back” technique (from smaller to larger tapers and tip sizes), and in more simple anatomy the canals are shaped “crown down” (from larger to smaller tapers and tip sizes). Insertion is gentle and passive, taking approximately 3 seconds and shaping approximately 4 mm of dentin per insertion. If the clinician is meeting undue resistance during insertion while moving crown down, he or she can immediately switch to an “apex up” sequence of file insertion, and vice versa. While not mandatory (and a matter of clinician preference), customizing the motor settings for speed (ie, rotations per minute) and torque control can otherwise minimize the possibilities of fracture in some clinical situations, especially in canals with abrupt apical curves.
Extrusion
To avoid extrusion of filling materials, media, and medicaments beyond the apex, a keen awareness of the position of the minor constriction of the apical foramen is essential at all times. Before starting the case, the clinician should estimate the working length, measure the true working length repeatedly, and verify the measurement before filling. A cone-fit radiograph is almost always indicated. True working length can and should be determined by a host of methods that include radiographic, tactile, bleeding point, electronic, and, optimally, cone-fit films as mentioned above.
Irrigation, instrumentation, and obturation must always be undertaken with the position, size, and anatomy of the apex in mind and with the utmost respect for the apical tissues. Open apices are always at higher risk for extrusion than closed apices. Although clinical opinions differ, this author believes that clinicians should consider using chlorhexidine as the primary irrigant in cases of perforation and/or cases where there is significant risk of extrusion due to open apices.
Conclusion
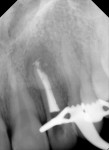
Diligent and comprehensive preoperative risk assessment of the patient-, tooth-, and clinician-related factors can help to predict clinical success or the possibility of an iatrogenic event. Such a risk assessment will tell the clinician where iatrogenic potential exists and provide strong clues to the appropriate clinical action that is needed to avoid any given complication (Figure 7 through Figure 9).
Disclosure
Dr. Mounce owns MounceEndo, LLC, which markets the rotary nickel-titanium MounceFile in Controlled Memory and Standard NiTi, as well as MANI products.
References
1. Pontes F, Pontes H, Adachi P, et al. Gingival and bone necrosis caused by accidental sodium hypochlorite injection instead of anaesthetic solution. Int Endod J. 2008;41(3):267-270.
2. Blanas N, Kienle F, Sàndor GK. Injury to the inferior alveolar nerve due to thermoplastic gutta percha. J Oral Maxillofac Surg. 2002;60(5):574-576.
3. Sharma S, Hackett R, Webb R, et al. Severe tissue necrosis following intra-arterial injection of endodontic calcium hydroxide: a case series. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(5):666-669.
4. Lindgren P, Eriksson KF, Ringberg A. Severe facial ischemia after endodontic treatment. J Oral Maxillofac Surg. 2002;60(5):576-579.
5. Ahlgren FK, Johannessen AC, Hellem S. Displaced calcium hydroxide paste causing inferior alveolar nerve paraesthesia: report of a case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96(6):734-737.
6. Mehra P, Clancy C, Wu J. Formation of a facial hematoma during endodontic therapy. J Am Dent Assoc. 2000;131(1):67-71.
About the Author
Richard Mounce, DDS
Private Practice, Endodontics
Chief Executive Officer, MounceEndo LLC
Rapid City, South Dakota