Lithium Disilicate for Custom Abutment Solutions
An anterior option that allows excellent esthetics and optimal placement
Dental implants have become a standard of care for treating various indications, from single unit restorations to partial or complete edentulism.1 They provide a stable, highly esthetic solution to myriad dental problems, and their surge in popularity is not undeserved.
The current popularity of dental implants does not preclude them from potential risks, however, not least of which includes complications due to inflammation. Nearly 80% of dental implant patients have or will experience inflammation and resulting complications, making it absolutely essential for patients to maintain healthy gingival tissues around their implants, as the survival of their implants depends on this good health.2,3
One of the most common issues to arise from implant placement is peri-implantitis, an inflammatory disease defined by bacterial contamination of the peri-implant surface.4 Causes of this condition vary, ranging from alcohol use, smoking, genetics, poor oral hygiene, and a number of iatrogenic factors.5,6 Failure to address peri-implantitis can ultimately lead to failure of the implant itself.6 While myriad treatments exist and have been recommended in the literature,7 proper planning, use of advanced materials, and careful placement of the implant and restoration can prevent the condition from occurring.
Custom Hybrid Abutments
The following case demonstrates a new way to provide custom abutments for implants to both address the problem of peri-implantitis and ensure highly esthetic results. Research has demonstrated that the color of an implant abutment has a direct influence on the color of the final implant-supported restoration when certain metal-free materials are used, and/or when proper thickness is not maintained.8 In such instances, a mask-out technique could be used to block out the color of the abutment. Porcelain-fused-to-metal (PFM) restorations, on the other hand, are not influenced by abutment color.
Potential show-through of abutment color is of great importance for restorations in the anterior region, where esthetics are key, particularly in garnering patient acceptance. In this case, a lithium disilicate material with exceptional light transmission, which necessitated the use of an esthetic-colored abutment, was used in the fabrication of a hybrid abutment crown to ensure natural esthetics. Neither PFM nor zirconia materials were used because they would not produce the desired light transmission.
Case Presentation
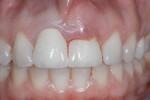
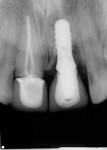
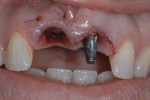
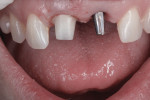
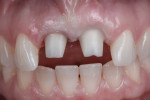
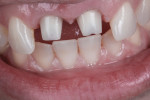
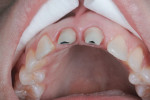
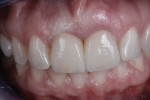
A 46-year-old woman presented with peri-implantitis surrounding tooth No. 9 and root resorption in tooth No. 8 (Figure 1). The crown restorations for both teeth demonstrated an improper fit along the gingival margins (Figure 2). In particular, the soft tissue at tooth No. 9 was inflamed as a result of poor implant abutment selection that placed the margin too deep within the tissue, which is contraindicated for cement-retained implant restorations. As noted previously, many causes of peri-implantitis are iatrogenic in nature,6 and cement-retained implant restorations require abutment placement at the correct margin level and precise removal of excess cement, to prevent development of inflammation.
A treatment plan was developed by Belkis del Puerto, DMD, and John Paul Gallardo, DDS, PA, to first address the peri-implantitis infection at tooth No. 9 and stabilize the already completely osseointegrated implant (Figure 3). Then, the soft tissue would be regenerated and recontoured (Figure 4). The clinical and laboratory protocol for creating the custom hybrid titanium and lithium disilicate abutments were dictated by the implant (eg, Straumann Tissue Level, Straumann, www.straumann.us) and materials (eg, IPS e.max®, Ivoclar Vivadent, www.ivoclarvivadent.com) used in this case.
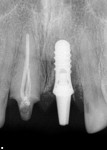
Subsequently, tooth No. 8 was extracted and a Straumann Tissue Level implant, similar to the one placed at No. 9 in 2005, was placed (Figure 5 and Figure 6) and provisionalized (Figure 7). The provisional in this case was splinted, using tooth No. 9 for support and tooth No. 8 as a pontic. Further design of the tissue architecture occurred at this time.
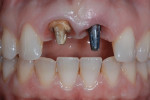
The patient was then allowed to heal for 6 months, after which impression copings were placed and an impression taken to fabricate a custom hybrid IPS e.max abutment (Figure 8). At the laboratory, the impression was poured in stone (FiJirOCK® stone, GC America, inc, www.gcamerica.com). To duplicate the actual position and architecture of the gingival tissue, the soft tissue was replicated on the model using a soft material (Zhermack, https://en.zhermack.com) and a model base (Giroform system, Amann Girrbach, www.amanngirrbachamerica.com). The custom abutment was selected for both teeth No. 8 and No. 9 to bring the margins as close to the tissue level as possible and avoid problems with cementation when delivering the definitive crown restorations. Additionally, by using a titanium base to engage the implant, fractures at the point of engagement could be avoided. The author has observed that in addition to demonstrating poor light transmission—which can affect the appearance of the soft tissue—zirconia-engaging abutments can break, whereas titanium-engaging abutments have been more predictable. Further advantages of this hybrid abutment included the ability to impart vitality within the soft tissue through the use of the tooth-colored IPS e.max ingot (versus what would be achieved with a metal-only abutment) (Figure 9).
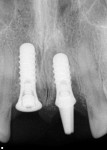
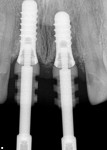
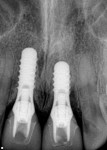
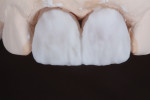
Three months later, an implant-level impression was taken of tooth No. 9 using an open tray impression technique and appropriate material. Titanium abutment components (Strauman Tissue Level) were selected for this case to prevent future fractures at the level of engagement. The emergence profile for the abutments and subsequent restorations was designed in wax and then pressed with an IPS e.max Press ingot in shade A2 LT (low translucency). Once the abutments were pressed and fit, the monolithic abutment design was cemented onto the implant components (Multilink® Implant, Ivoclar Vivadent). (Figure 10).
Monolithic crowns were then fabricated from shade A2 for cementation on top of the abutments. They were sent to the doctor for try-in to verify soft tissue support, color, occlusion, contours, and shape (Figure 11 and Figure 12). At try-in, it was determined that the abutments could remain in place, but the contours of the provisional restorations should be altered, since they were too wide at the cervical area, pushing the papilla. The doctor duplicated the crowns and created new provisionals with different contours to prepare the gingival tissues for the definitive restorations (Figure 13 and Figure 14).
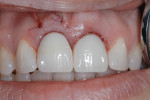
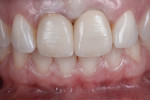
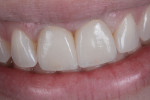
When the restorations were tried in the patient’s mouth, the crowns were monolithic. However, the restorations were returned to the laboratory for esthetic modifications. There, they were altered using equal ratios of EO1 and EO3 (Figure 15). The crowns were then sent back to the doctor’s office, where they were seated and cemented onto the customized abutments using Multilink Implant (Figure 16 and Figure 17).
Discussion
Esthetics were an important factor in the design of a treatment plan for this case, as the teeth involved were the patient’s maxillary central incisors. Therefore, it was decided that a custom abutment should be used. The customization afforded by this type of abutment permitted an esthetic and effective design to be implemented that also corrected for the previous problems encountered with the first restoration.
IPS e.max Press was chosen as the material of choice for this procedure, based on its outstanding esthetic and clinical properties. Research has shown that lithium disilicate provides exceptional strength and esthetics, being identified as the most robust material studied in the research published by the New York University College of Dentistry.9 A hybrid abutment developed from IPS e.max Press allows unique design and adjustment of the emergence profile, shape, and esthetic properties of the clinical situation at hand.10 This material demonstrates a strength of 400 MPa and is indicated for myriad situations, including hybrid abutment restorations in the anterior region.10
Combined, these characteristics make it an ideal alternative to stock or prefabricated abutments—or other custom abutments made from less-than-optimal ceramic or metal-ceramic materials (eg, gold, PFM, zirconia), which may not fully and/or adequately address a patient’s needs or desires. For example, custom abutments fabricated with gold may block out and/or prevent light transmission needs, creating an esthetically inappropriate appearance. Additionally, certain materials may absorb the color of a gold abutment, also rendering less-than-ideal esthetics.
The design and shape of the hybrid abutment also permits easy integration of the restoration, as the crown preparation margin will be at gingival level, ensuring that the removal of superfluous cement will be easy. This, in turn, greatly reduces the patient’s likelihood of developing peri-implantitis from improper cement removal.10 This ease of use is among the reasons IPS e.max Press stands as an ideal solution for fabricating implant-supported abutment/restorations.
Conclusion
Clinicians and laboratory technicians alike have long sought answers to the problems that prefabricated abutments bring, with their inability to be designed according to necessary patient specifications. With the use of IPS e.max Press, a solution to this problem and that of possible complications, such as peri-implantitis, is presented, as the custom abutments simultaneously permit placement at the correct gingival level while remaining a highly esthetic solution. This lithium disilicate option affords clinicians and technicians the ability to design esthetic, durable abutments and restorations that will prove long lasting for years to come.
References
1. Inan O, Secilmis A, Eraslan O. Effect of pontic framework design on the fracture resistance of implant-supported all-ceramic fixed partial dentures. J Appl Oral Sci. 2009;17(5):533-538.
2. Zeza B, Pilloni A. Peri-implant mucositis treatments in humans: a systematic review. Ann Stomatol (Roma). 2012;3(3-4):83-89.
3. Esposito M, Grusovin MG, Worthington HV. Interventions for replacing missing teeth: treatment of peri-implantitis. Cochrane Database Syst Rev. 2012;1:
CD004970. doi: 10.1002/14651858.CD004970.pub5.
4. Dörtbudak O, Haas R, Bernhart T, Mailath-Pokorny G. Lethal photosensitization for decontamination of implant surfaces in the treatment of peri-implantitis. Clin Oral Implants Res. 2001;12(2):104-108.
5. Shapoff CA, Lahey BJ. Crestal bone loss and the consequences of retained excess cement around dental implants. Compend Contin Educ Dent. 2012;33(2):94-96, 98-101; quiz 102, 112.
6. Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35(8 suppl):292-304. doi: 10.1111/j.1600-051X.2008.01275.x.
7. Nguyen-Hieu T, Borghetti A, Aboudharam G. Peri-implantitis: from diagnosis to therapeutics. J Investig Clin Dent. 2012;3(2):79-94. doi: 10.1111/j.2041-
1626.2012.00116.x.
8. Chaiyabutr Y, Kois JC, Lebeau D, Nunokawa G. Effect of abutment tooth color, cement color, and ceramic thickness on the resulting optical color of a CAD/CAM glass-ceramic lithium disilicate-reinforced crown. J Prosthet Dent. 2011;105(2):83-90. doi: 10.1016/S0022-3913(11)60004-8.
9. Guess PC, Zavanelli R, Silva NR, Thompson VP. Clinically relevant testing of dental porcelains for fatigue and durability with an innovative mouth motion simulator. Presented at: 39th Annual Session of the American Academy of Fixed Prosthodontics; February 2009; Chicago, IL.
10. Blanco J, Rego N. Using lithium-disilicate hybrid abutments for implant restorations. Inside Dental Technology. 2013;4(2):80-81.
ABOUT THE AUTHORS
Rafael Santrich, CDT
President
VM Lab Technologies
Aventura, Florida
For more information, contact:
Ivoclar Vivadent
800-533-6825
www.ivoclarvivadent.com