Getting Back to the Basics of Instrument Reprocessing
Eve J. Cuny
Instrument cleaning and sterilization are critical elements of a dental infection-control program. Patients expect that the instruments and devices used in connection with dental procedures are free from organic and inorganic materials from previous patients, and most importantly, free of organisms that have the potential to transmit disease from patient to patient.
Numerous obstacles to effective instrument reprocessing may present themselves. Time constraints can interfere with the ability of personnel to complete all necessary steps, malfunctioning equipment may compromise the effectiveness of the processes, equipment may have parts that are difficult to clean or cannot be heat sterilized, and personnel responsible for the process may lack appropriate training.
GUIDELINES AND REGULATIONS
One common misconception is that the Occupational Safety and Health Administration (OSHA) set the standard for sterilization in its Bloodborne Pathogens Rule. That regulation applies to the safety of workers, but does not address issues that are specific to patient safety. OSHA does require that personnel use appropriate precautions, such as personal protective equipment (PPE) when transporting, cleaning, and sterilizing contaminated instruments,1 but a more comprehensive guideline for infection control practices comes from the Centers for Disease Control and Prevention (CDC).2 The CDC, in its role as the United States Public Health Service (USPHS), develops guidelines and recommendations on a broad range of health topics, including infection control. The Guidelines for Infection Control in Dental Health-Care Settings–2003 covers topics that are relevant to both patient safety and occupational safety, making this a vital document to reference when developing and monitoring the dental infection-control program. Although the CDC does not have regulatory authority, many dental licensing agencies adopt the CDC guidelines in whole or in part into their requirements for licensees. The CDC classifies dental instruments as critical, semicritical, or noncritical items (Table 1). This classification system determines which instruments must be heat sterilized. In today’s dental practice nearly everything that enters the patient’s mouth is either disposable or heat stable, making it easy to reprocess between patients. Larger equipment, such as mechanical scalers and curing lights, often have detachable intraoral parts, which are possible to heat sterilize. It is simple to manage the areas that personnel touch during the procedure using impervious barriers. Always change equipment barriers between each patient.
CLEANING
The first and arguably most important step in instrument reprocessing is cleaning. Heat sterilizers that provide the terminal process of sterilization rely on the premise that the instruments are free from all debris and that the sterilizing agent (eg, steam, pressure, heat) will contact all surfaces of the items in the sterilizer. Debris such as dental materials, blood, and tissue interfere with this process and may result in instruments that are not suitable for use on patients. There are three available methods for cleaning dental instruments and it may be necessary to use a combination of two or more methods. Few offices have one team member dedicated solely to instrument reprocessing and, therefore, it is important to ensure the process is as efficient as possible. If possible, select one best cleaning method to save time.
Ultrasonic
Ultrasonic cleaners use sound waves to produce cavitation in a chamber of water and cleaning agent. Debris on instruments immersed in ultrasonic cleaners separates from the instrument under the pressure of cavitation. A properly functioning ultrasonic is more effective in cleaning instruments than hand scrubbing.3
Place instruments in a basket and immerse fully in the cleaning solution. Cover the ultrasonic tank before activation. Follow the manufacturer’s instructions for the operating time for cleaning and for selection of the proper liquid cleaning agent.
Although research indicates there is little escape of microorganisms during ultrasonic cleaning, use of the cover effectively reduces the contamination to nondetectible amounts.4 The ultrasonic does not reliably disinfect the instruments.4 Therefore, personnel must continue to wear heavy-duty gloves and other PPE during rinsing, drying, and packaging of instruments after cleaning and before sterilization (Figure 1).
Washer/Disinfector
Instrument washer/disinfectors are medical devices that will clean and disinfect instruments using a combination of hot water, pressure, and chemical agents (Figure 2). Available in both countertop and built-in models, these devices provide a reliable and effective means of preparing instruments for sterilization.5 Upon removal from the device, instruments are safe to handle since they have completed a disinfection process.
Some instrument washers do not have a thermal disinfection cycle. When using these devices for cleaning instruments, personnel must continue to wear heavy-duty gloves and other PPE while packaging the instruments and loading the packages into the sterilizer.
Hand Scrubbing
Manually cleaning dental instruments using a brush and running water is the least safe and efficient method available. Occasionally, there is a need to hand scrub instruments after mechanical cleaning processes if stubborn debris remains on the instrument. In this case, personnel performing this function must wear heavy-duty gloves and PPE to prevent splashes to the skin, eyes, or other mucous membranes.
STERILIZATION
Sterilization as the final step is still necessary to meet current standards for instrument reprocessing. Dental offices usually use one of three basic methods for heat sterilization; steam under pressure (autoclave), dry heat, or unsaturated chemical vapor.
The type of sterilization packaging for dental instruments is dependent upon the instruments, the instrument delivery system, and the type of sterilizer. Paper pouches with or without a clear plastic side work well with steam autoclaves and unsaturated chemical vapor sterilizers. Nylon tubing is appropriate for the higher temperatures of dry-heat sterilizers. Disposable CSR wrap is appropriate for instrument cassettes and trays. Unless the manufacturer indicates otherwise, all sterilization wraps and pouches are single-use.
Autoclave
Autoclaves use either gravity or mechanical pre-vacuum as the mechanism for creating the steam under pressure necessary to achieve sterilization. In gravity autoclaves, as the water heats and steam begins to fill the chamber, gravity forces air out through vents in the autoclave chamber. In pre-vacuum (also called Type B sterilizers), air is evacuated from the sterilization chamber, creating a vacuum before steam is introduced. Pre-vacuum sterilizers may also have an additional vacuum cycle before the drying cycle begins, which tends to result in faster drying time. Gravity sterilizers are appropriate for solid instruments and pre-vacuum sterilizers are appropriate for solid instruments and some hollow-bore instruments or devices.
Dry Heat
Dry-heat sterilizers use a higher temperature than do other heat sterilizers to achieve sterilization without steam or chemical vapor. Static dry-heat sterilizers rely on heated air rising in the chamber to contact all instruments and maintain a minimum temperature for the duration of the sterilization cycle. The hot air rising and falling can result in uneven temperatures, making static heating less effective than other methods. Convection dry-heat sterilizers circulate heated air throughout the sterilization cycle, helping maintain a uniform temperature throughout the process.
Unsaturated Chemical Vapor
Unsaturated chemical vapor sterilizers (eg, Chemiclave®, Barnstead®/Harvey®, Dubuque, IA) use a proprietary formula containing alcohol and formaldehyde to generate a heated chemical vapor under pressure to achieve sterilization. The low moisture helps prevent damage to corrosion-prone instruments, such as those that contain carbon steel. Proper ventilation is necessary for use of unsaturated chemical vapor. Current models have built-in ventilation systems.
SPECIAL CONSIDERATIONS
Technology in the dental office often presents additional challenges to asepsis. If the device has components that are semicritical or critical, heat sterilization of those components is preferable. Barriers that prevent contamination are one alternative to heat sterilization if the components are not detachable or are heat sensitive. High-level disinfection of intraoral devices is not the preferred method, but may be necessary in specific situations where barriers or heat sterilization are not possible.
A number of items are difficult to clean, and the dental team may find that switching to disposable product provides a more efficient alternative. Carbide burs are difficult to clean and may become dull during the heat sterilization process. Prophy angles often require multiple steps for maintenance and require careful cleaning before sterilization. Multi-dose containers of dental materials are difficult to manage if they have brushes or other tips that go into the patient’s mouth. Evaluation of alternative products, including disposable, unit-dose, and heat-stable items, is a continuous process that involves the entire dental team.
QUALITY ASSURANCE
Monitoring of the sterilization cycle parameters is one method of ensuring the sterilization process is effective. There are several methods of monitoring, each with a different level of assurance.
Physical monitoring includes viewing gauges for proper time, temperature, and/or pressure. Some sterilizers provide a tape or electronic record of each sterilization cycle (Figure 3). A periodic review of these records helps the dental team ensure that proper parameters are in use when sterilizing instruments.
Chemical monitoring involves the use of indicators that are sensitive to the parameters of the sterilizer (eg, steam, pressure, heat). These usually have a visual indicator, such as a change in color that alerts the user to the presence of the appropriate conditions for sterilization. Some chemical indicators measure one parameter, while others measure multiple parameters with a single test. Chemical monitoring helps the dental team tell the difference between sterilized and unsterilized packages or cassettes of dental instruments.
For pre-vacuum autoclaves, an air removal test, called a Bowie-Dick test, provides assurance that there is adequate air removal for the sterilization cycle.
Biological indicators (BI), also called spore tests, are the highest level of sterility assurance. BIs are either strips or vials that contain spores that are resistant to the heat-sterilization process. A weekly BI test helps ensure that the sterilizer is functioning properly and that the personnel responsible for instrument reprocessing are using proper methods of sterilization.
REFERENCES
1. US Department of Labor, Occupational Safety and Health Administration. 29 CFR Part 1910.1030. Occupational exposure to bloodborne pathogens; needlesticks and other sharps injuries; final rule. Fed Regist. 2001;66:5317-5325. As amended from and includes 29 CFR Part 1910.1030. Occupational exposure to bloodborne pathogens; final rule. Fed Regist. 1991;56:4174-4182.
2. Centers for Disease Control. Guidelines for Infection Control in Dental Health-Care Settings–2003. MMWR Recomm Rep. 2003;52(No. RR-17):1-61.
3. Cafruny WA, Brunick A, Nelson DM, Nelson RF. Effectiveness of ultrasonic cleaning of dental instruments. Am J Dent.1995;8(3): 152-156.
4. Bettner MD, Beiswanger MA, Miller CH, Palenik CJ. Effect of ultrasonic cleaning on microorganisms. Am J Dent. 1998;11(4): 185-188.
5. Miller CH, Tan CM, Beiswanger MA, et al. Cleaning dental instruments: measuring the effectiveness of an instrument washer/disinfector. Am J Dent. 2000;13(1): 39-43.
 |
 |
||||||
| Figure 1 Personal protective attire for handling contaminated instruments during reprocessing. | Figure 2 Dental office automated washer/ disinfector. | ||||||
 |
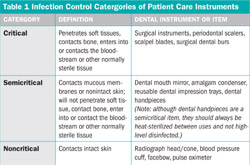 Table 1 |
||||||
| Figure 3 Autoclave cycle record. | Adapted from: CDC Guidelines for Infection Control in Dental Health-Care Settings—2003. | ||||||
|
|||||||




