Development of a Processed Composite Resin Restoration: Preparation and Laboratory Fabrication—Part I
Douglas A. Terry, DDS, and Karl F. Leinfelder, DDS, MS
Advances in adhesive restorative biomaterials by researchers, clinicians, scientists, and manufacturers have resulted in adhesive restorations that provide increased retention, marginal adaptation and seal, and reduced microleakage. This evolution in the development of adhesive dental technology, with adhesive materials and techniques, has dramatically changed the way dentistry is practiced in the modern dental office. Modern adhesive restorative materials and techniques have provided clinicians more conservative treatment avenues that preserve tooth structure while improving the longevity and esthetics of the restoration.
From the wide range of restorative biomaterials, indirect laboratory-processed composite resin systems provide an esthetic alternative for intracoronal posterior restorations. Laboratory-processed inlays and onlays fabricated with composite resin provide esthetic results that may also reinforce tooth structure. Because this adhesive procedure strengthens the cusps and provides additional support for the dentition, a more conservative preparation design can be used. Thus, these systems restore mechanical and biological function while achieving optimal esthetic results with minimal resin cement shrinkage and limited tooth reduction. Additional clinical benefits include precise marginal integrity, wear resistance similar to enamel, and wear compatibility with opposing natural dentition but also ideal proximal contacts, excellent anatomical morphology, and optimal esthetics.1-3
Next-Generation Indirect Systems
The use of laboratory-processed composite resin systems for intracoronal restoration of posterior teeth has increased dramatically with the improvements in physical and mechanical properties of these resin systems and patient demand for tooth-colored restorations. These next-generation resin systems (TESCERA ATL™, Bisco, Inc, Schaumberg, IL; Gradia™ Light-Cured Micro-Ceramic Composite, GC America, Inc, Alsip, IL; Sculpture® Plus, Pentron Laboratory Technologies, Wallingford, CT) maintain a higher density of inorganic ceramic microfillers compared to the earlier-generation direct and indirect systems.4 These materials have been noted for possessing the advantages of composite resins and porcelains without being confined by their inherent limitations.5
The biomaterial, known as “microhybrids,” include a combination of inorganic particles (fillers) and an organic polymer (matrix), with a filler content that contains twice the organic matrix content (approximately 66% inorganic fillers and 33% resin matrix). The filler is the primary determinant of the clinical and physiochemical properties of composite resin material. These submicron-particle fillers demonstrate exceptional surface characteristics such as polishability and wear resistance.6 The wear is influenced by the filler size, filler shape, filler load, and filler/matrix bonding.7-9 In fact, a significant reduction in wear resistance has been reflected by simply decreasing the size of the filler particle.7,10
In addition, the various methods of postcuring (eg, light, heat pressure, vacuum, nitrogen) allow for secondary curing of the composite by increasing the conversion of the material from monomer to polymer.11 This heightened but controlled degree of polymerization increases fracture toughness, flexural and diametral tensile strength, wear resistance, incisal edge strength, and color stability.12,13 Whereas many articles have examined the plethora of uses for indirect resin-reinforced systems, this discussion will focus on the onlay restoration employing an indirect resin-reinforced system that uses three curing mechanisms—pressure, light, and heat underwater. Part I will describe each of the systems’ mechanisms and the specific material properties of this next generation indirect composite resin system (TESCERA ATL) including a detailed review of the preparation and laboratory fabrication.
Components of the System
An understanding of a specific indirect composite resin system requires a discussion of the components of the system: the resin material and the curing mechanism. The indirect composite resin system used in this case presentation, TESCERA ATL, contains a combination of three different forms of composite resin material: dentin, body, and incisal components.
Composite Resin Material
In selecting a resin material, the particle size represents crucial information on the determination of how to best use composite materials.14 The filler particle size, distribution, and the quantity incorporated dramatically affects the mechanical properties and clinical success of composite resins.15 The filler particles are silanated for suitable adhesion to the organic matrix. This indirect system’s filler composition varies for the dentin material and the body and incisal. The dentin material is a highly filled hybrid (85% by weight, 73% by volume) similar to the proprietary mixture of the direct restorative LITE™ LS (Bisco, Inc). This increased filler loading allows a volumetric shrinkage of 1.5% while maintaining a high flexural strength. The body and the incisal material consists of a reinforced microfill (70% by weight) similar to the proprietary mixture of the direct restorative MICRONEWT™ (Bisco, Inc).16 Added to the nanoparticles is a relatively large “reinforcement” particle that averages 1 µm in size, compared to the main filler, which is 0.04 µm in size. The average particle size for this composite is approximately 50 nm (0.05 µm). The presence of these 1-µm reinforcement particles are reported to contribute to the strength by acting as a “crack arrester,” while the increased particle concentration of the microfill particles provides improved clinical performance through increased polishability, durability of the polish, wear resistance, and fracture resistance.16
The matrices for the dentin, body, and incisal material consist of various combinations of diluents: Bis-GMA (bisphenol A-glycidyl methacrylate), urethane dimethacrylate, ethoxylated bis “a” dimethacrylate, and tetraethylene glycol dimethacrylate. However, the matrix for the incisal differs from that of the dentin and body in that the incisal uses a low Bis-GMA concentration, whereas the dentin and body materials have a higher concentration. A study of the incisal material by Ferracane and Condon at Oregon Health Sciences University indicates a greater abrasion resistance than other indirect systems tested.16 The manufacturer suggests that this improved wear resistance of the incisal material is a result of the change in concentration of Bis-GMA.17
Curing Mechanism
The polymerization process for this indirect system combines two curing mechanisms—light and heat under water. The “artificial dentin” is initially completely pressurized (60 psi) in a light cup before the light-curing cycle is initiated. The initial pressurization eliminates the incorporation of internal voids and bubbles during the incremental build-up process. The light-cup contains white reflection beads which provide support to the working die while reflecting and diffusing light around the chamber and onto the composite surface. During the incremental build-up process, each light-cure cycle requires 2 minutes and stabilizes the restoration during build-up, which allows placement of subsequent increments without deforming the underlying composite layer. After complete development of the restoration, the final cure is accomplished in a heat cup with the restoration submerged in water. Any residual free oxygen in the water is removed by adding an oxygen-scavenger tablet that absorbs the residual oxygen. This is beneficial since oxygen limits the degree of polymerization by competing at the carbon double-bond sites. Therefore, removing oxygen allows for a more complete cure because no air-inhibited layer remains uncured,18 and this may improve the physical and mechanical properties at the surface. The final restorations are cured using an initial full cycle of pressure (60 psi) with light and heat (peak heat of 130°C and temperature decreases to approximately 90°C before the pressure is released) for approximately 10 minutes to 13 minutes depending on the size of the restoration and the initial temperature of the water. The final curing process with heat under pressure increases the polymer conversion and eliminates the residual monomers. The resulting composite material provides increased strength and homogeneity, excellent esthetics with enhanced optical properties and fluorescence, low water sorption and solubility, color stability, and superior resistance to wear and deformation.2
Fiber Reinforcement
For large restorations or teeth with minimal remaining enamel, fibers should be included as a base on which to veneer the composite.19 An important consideration for achieving optimal long-term clinical success of laboratory-fabricated resin inlays /onlays is tooth reinforcement. To reinforce the composite resin, additional fibers (TESCERA Reinforcement Material, Bisco, Inc; Ribbond®, Ribbond, Seattle, WA) are integrated into the resin matrix20,21 during fabrication and before the curing process. These fibers have been surface-treated to enhance the adhesion to any synthetic restorative material. Although no long-term clinical trials are available to determine the clinical success of these materials, a recent short-term study on 60 single-crown restorations demonstrated no breakage after 1 year.5,22 Because the flexural strength and fracture resistance of the restoration is increased by the addition of composite reinforced fibers,5,23 the authors believe it is prudent to incorporate them to reduce fractures in regions of increased occlusal stress.
A recent development of another type of reinforcing structure for these indirect composite resin systems is the TESCERA structural fibrous material, which consists of pre-tensed quartz fibers that are cured into a resin matrix to provide a rigid, strong reinforcing structure. These materials consist of different shapes and configurations (ie, U-Bars, barrels, sleeves, and fiber bundles) which have been surface-treated to enhance the adhesion to any synthetic restorative material.
Preparation, Impression, and Provisionalization
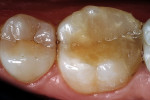
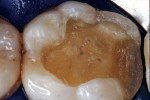
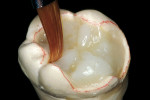
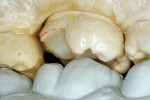
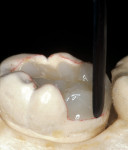
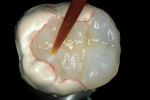
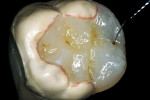
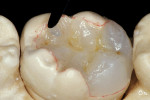
The adhesive preparation design preserved sound tooth structure and required no extension for prevention. The preparation was limited to access to the defect, since the composite required less volume to resist clinical fracture than would have an amalgam.24,25 Upon removal of the existing recurrent caries, the cavity design followed the preparation guidelines for indirect inlay/onlay restorations (Figure 11):
- All enamel supported by sound, healthy dentin;
- All internal angles and edges rounded to avoid stress and facilitate the fabrication of the restoration;
- Isthmus width should be at least 2 mm with a minimum depth of 1.5 mm;
- All proximal walls should be flared or diverged 5 degrees to 15 degrees with no undercuts;
- Gingival margins should be prepared to a 90-degree cavosurface line angle (ie, butt joint);
- Sharp cavosurface margins should be maintained;
- Occlusal margins should not coincide with occlusal contact site;
- No feather-edge preparation.2,5,11,26
As a general guide, when the isthmus preparation exceeds one half of the distance from the central fossa to the cusp tip, a restoration with cuspal coverage should be considered. In areas of low stress and where there is minimal potential of tooth flexure, thinner areas of tooth structure may be judiciously inlayed. For larger restorations or weak teeth with minimal enamel as aforementioned, fibers should be included as a base on which to veneer the composite.5
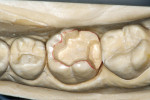
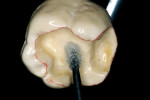
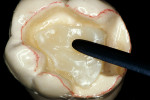
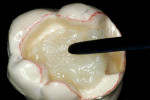
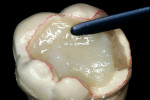
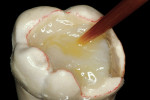
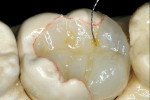
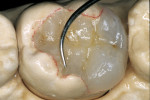
Before impression making, it was important to seal the dentin tubules with a hybrid layer.1,11,27,28 This protected the pulp from the invasion of microorganisms and reduced sensitivity during the provisional stage. Once the preparation was conditioned, a thin layer of adhesive (All-Bond® 2, Bisco, Inc; G-Bond, GC America; Gluma® Comfort Bond + Desensitizer, Heraeus Kulzer, Armonk, NY) was applied on the preparation surfaces with an applicator for 20 seconds, air-thinned for 5 seconds, and light-cured for 20 seconds. To prevent interaction of the dentin adhesive with the impression material, particularly polyethers, the adhesive layer was covered with a layer of glycerin and additionally light-cured for 20 seconds.29 A polyether impression (ie, Impregum™, 3M™ ESPE™, St. Paul, MN; Permadyne™, 3M™ ESPE™; Polyjel® NF, DENTSPLY Caulk, Milford, DE; P2™, Heraeus Kulzer) was made, including all cavosurface margins. A direct provisional restoration was placed with a matrix band (Automatrix®, DENTSPLY Caulk) using a light-cured, semiflexible material (ie, Fermit, Ivoclar Vivadent, Amherst, NY), and the occlusion was evaluated.
Laboratory Development
The laboratory procedure shown in Figure 22; Figure 3; Figure 4; Figure 5; Figure 6; Figure 7; Figure 8; Figure 9; Figure 10; Figure 11; Figure 12; Figure 13; Figure 14; Figure 15; Figure 16; Figure 17; Figure 18A and Figure 18B illustrates the interpretation of the clinical information and describes how this information can be used with laboratory-pro-cessed composite resins to integrate the existing color of the natural tooth with the optical properties of the restorative material, TESCERA ATL.
Conclusion
While new products and technological advancements impact our profession positively, a new burden rests on clinicians and technicians to continually educate themselves and their patients on the properties and applications of the new restorative biomaterials. This knowledge has provided patients and clinicians with alternative conservative treatment avenues to various clinical scenarios. However, it is essential to remember that the final restorative result is based on the experience and judgment of the clinician and technician and the communication and understanding between them. Part 1 of this discussion has reviewed the general properties of the next-generation, laboratory-processed composite resins, explained the components of a specific indirect composite resin system (TESCERA ATL) and provided a detailed description of the preparation and laboratory fabrication of an onlay restoration. Part 2 will describe the principles that should be followed to achieve long-term success, with an emphasis on adhesive bonding and finishing protocols.
References
1. Touati B, Aidan N. Second generation laboratory composite resins for indirect restorations. J Esthet Dent. 1997;9(3):108-118.
2. Howard NY. Advanced use of an esthetic indirect posterior resin system. Compend Contin Educ Dent. 1997;18(10):1044-1050.
3. Leinfelder KF. Indirect posterior composite resins. Compend Contin Educ Dent. 2005;26(7):495-503.
4. Douglas RD. Color stability of new-generation indirect resins for prosthodontic application. J Prosthet Dent. 2000;83(2):166-170.
5. McLaren EA, Rifkin R, Devaud V. Considerations in the use of polymer and fiber-based indirect restorative materials. Pract Periodontics Aesthet Dent. 1999;11(4):423-432.
6. Soderholm KJM. Filler systems and resin interface. In: Vanherle G, Smith DC (eds). International Symposium on Posterior Composite Resin Dental Restorative Materials. St Paul, MN: Minnesota Mining + Mfg.Co.; 1985:139-159.
7. Suzuki S, Leinfelder KF, Kawai K, et al. Effect of particle variation on wear rates of posterior composites. Am J Dent. 1995;8(4):173-178.
8. Braem M, Finger W, Van Doren VE, et al. Mechanical properties and filler fraction of dental composites. Dent Mater. 1989; 5(5):346-348.
9. St. Germain H, Swartz ML, Phillip RW, et al. Properties of microfilled composites resins as influenced by filler content. J Dent Res. 1985;64(2):155-160.
10. Bayne SC, Taylor DF, Heymann, HO. Protection hypothesis for composite wear. Dent Mater. 1992;8(5):305-309.
11. Miara P. Aesthetic guidelines for second-generation indirect inlay and onlay composite restorations. Pract Periodontics Aesthet Dent. 1998;10(4):423-431.
12. Cook WD, Johannson M. The influence of postcuring on the fracture properties of photo-cured dimethacrylate based dental composite resin. J Biomed Mater Res. 1987;21(8):979-989.
13. Bausch JR, de Lange C, Davidson CL. The influence of temperature on some physical properties of dental composites. J Oral Rehabil. 1981;8(4):309-317.
14. Ferracane JL. Current trends in dental composites. Crit Rev Oral Biol Med. 1995;6(4):302-318.
15. Leinfelder KF. Composite resins. Dent Clin North Am. 1985;29(2):359-371.
16. Suh BI. New concepts and technology for processing of indirect composites.Compend Contin Educ Dent. 2003;24(8Suppl):40-42.
17. Oral conversation with Steven Duray chemist.
18. Shellard E, Duke ES. Indirect composite resin materials for posterior applications. Compend Contin Educ Dent. 1999;20(12):1166-1171.
19. Jackson RD. Indirect resin inlay and onlay restorations: a comprehensive clinical overview. Pract Periodontics Aesthet Dent. 1999;11(8):891-900.
20. Dickerson WG, Rinaldi P. The Fiber-Reinforced Inlay-Supported Indirect Composite Bridge. Pract Periodontics Aesthet Dent. (Supplement) 1996.
21. Touati B, Miara P, Nathanson D. Esthetic Dentistry & Ceramic Restorations. London, UK; Martin Dunitz; 1999:291.
22. Filled Polymer Crowns: 1 & 2 Year Status Report. Clinical Research Associates Newsletter. 1998;22(10);1-3.
23. Dyer SR, Sorensen JA. Flexural strength and fracture toughness of fixed prosthodontic resin composites. J Dent Res. 1998;77(special issue):160(Abstract No. 434).
24. Sturdevant CM, Roberson TM, Heymann HO, Sturdevant JR. The Art and Science of Operative Dentistry. 3rd ed. St. Louis, MO: Mosby-Year Book; 1995.
25. Leinfelder, KF. A conservative approach to placing posterior composite resin restorations. J Am Dent Assoc. 1996; 127(6):743-748.
26. Burke FJ, Watts DC, Wilson NFH, et al. Current status and rationale for composite inlays and onlays. Br Dent J. 1991;170(7):269-273.
27. Nakabayashi N, Kojima K, Mashuhara E. The promotion of adhesion by infiltration of monomers into tooth substrates. J Biomed Mater Res. 1982;16(3):265-273.
28. Pashley DH, Ciucchi B, Sano H, et al. Permeability of dentin to adhesive agents. Quintessence Int. 1993;24(9):618-631.
29. Magne P, Belser U. Bonded Porcelain Restorations in the Anterior Dentition A Biomimetic Approach 1st ed. Carol Stream, IL: Quintessence Publishing; 2002.
About the Authors
Douglas A. Terry, DDS
Assistant Professor, Department of Restorative Dentistry and Biomaterials
University of Texas Health Science Center, Dental Branch
Houston, Texas
Private Practice-Esthetic and Restorative Dentistry
Houston, Texas
Karl F. Leinfelder, DDS, MS
Adjunct Professor, Biomaterials Clinical Research
University of North Carolina
Chapel Hill, North Carolina
Professor Emeritus
University of Alabama School of Dentistry
Birmingham, Alabama