Bioactive Dental Materials
Composition, properties, and indications for a new class of restorative materials
Steven R. Jefferies, MS, DDS, PhD
In the past 25 years, a new classification of dental restorative material has emerged. Bioactive cements, also termed bioactive chemical-bonded ceramics,1,2 have significantly improved the care we can give our patients in many clinical applications. Bioactive dental cements appeared with the introduction of ProRoot® MTA (DENTSPLY Tulsa Dental Specialties, www.tulsadentalspecialties.com) in the mid 1990s. Since that time, this category of dental materials has continued to evolve rapidly, and its bioactivity has created some confusion and controversy. The purpose of this article is to address basic questions and provide some clarity concerning this interesting class of restorative materials.
Defining Bioactivity
The terms bioactivity and bioactive material have recently emerged in both the dental literature and advertisements for new dental products. As when any new concept in dentistry emerges, the quest for understanding its relevance to clinical practices becomes critical.
The term bioactive material appears to have originated with Dr. Larry Hench as a result of his development of the calcium silicophosphate glass, Bioglass.3 His initial primary interest was the development of improved graft materials for orthopedic and bone reconstruction. Therefore, Hench’s research led to the discovery that Bioglass could chemically bond to bone. This property became the initial and longest-standing definition of a bioactive material.
However, the in vivo usage test needed to confirm that a material was bioactive by this definition was expensive and complicated. Based on the more straightforward test suggested by Kokubo and colleagues,4 a growing consensus has emerged regarding a more universal definition of bioactivity among materials scientists and clinician researchers. In the literature, the term bioactivity has the following definition when applied to dental materials: Bioactivity is the property of a biomaterial to form apatite-like material on its surface when immersed in a simulated body fluid (SBF) for a period of time. A standardized, internationally developed test is available from the International Standards Organization (ISO 23317:2007) that provides a specific protocol and evaluation standard to establish whether a material is, in fact, bioactive. This standard specifies that any material claimed to be bioactive must meet the endpoint of this international test—the ability to form measureable surface apatite by 28 days in a specific SBF containing inorganic phosphate. Based on this scientifically validated definition, the traditional formulations of glass ionomers, resin-modified glass ionomers (RMGIs), and fluoride-releasing composites would not be bioactive unless they demonstrated the formation of surface apatite in a SBF.5,6 Gandolfi and colleagues6 suggest terminology to differentiate the ion-releasing behavior seen in nonbioactive materials, such as glass ionomers or fluoride-releasing composites, by classifying such materials as biointeractive. This differentiates such materials from calcium silicate and calcium aluminate biomaterials, which the literature demonstrates are both biointeractive and bioactive (ie, apatite-forming).1,2,6
Defining bioactivity is an interesting proposition for the dental materials researcher, but the clinician needs more practical information to effectively select and utilize bioactive products in clinical practice. This includes how the properties of bioactive materials compare with those of other classes of dental materials and what they provide in terms of clinical efficacy; whether these materials are necessary in clinical practice, and if so, how and when they should be used; and if all resin formulations that contain bioactive components are equally effective.
Material Composition and Properties
Bioactive materials available commercially and utilized in clinical dentistry today fall primarily in one of two compositional classes: calcium silicates and calcium aluminates.1,2 Both materials, when their powder component is mixed with water, set with an acid-base chemical reaction and produce an alkaline pH after setting. The role of pH in bioactivity is potentially important, as all of the strictly water-based, calcium silicate/calcium aluminate cements significantly elevate the local pH level in solutions in which they are immersed. Accordingly, the most active of the bioactive materials demonstrate a high pH level in storage solutions. High pH levels (7.5 or higher) appear to stimulate more active and complete bioactivity.
Because these materials are primarily water-based, polysalt, chemically bonded-ceramic cements, their physical strength properties are similar to other cement types such as zinc orthophosphate, calcium hydroxide, polycarboxylate, zinc oxide eugenol, and certain conventional glass ionomers and RMGIs. The place of the bioactive calcium silicate and calcium aluminate cements in the evolution of dental cements can be seen in Table 1.
In terms of mechanical-strength properties, bioactive cements have evolved from cements with relatively low levels of strength (ie, compressive strength) to compositions with levels of strength virtually equal to glass ionomer and self-adhesive resin-based cements. For water-based cements, the mechanical strength conforms to an ISO test method. The earliest dental bioactive cements, such as mineral trioxide aggregate (MTA), displayed relatively low compressive strengths, which were approximately 30 to 50 MPa at 24 hours. Some of the latest materials for restorative and luting applications, such as Biodentine® (Septodont, www.septodontusa.com) and Ceramir® C&B (Doxa Dental, www.doxadentallearning.com), have 24-hour compressive strength values of approximately 150 MPa, a significant improvement.
Several other physical properties of bioactive cements have potentially significant implications for clinical use, including tendencies toward high water sorption and solubility and slower setting times. Both of these properties can have significant impact on the handling and efficacy of these materials.
A recent study examining a calcium silicate–based material reported a water sorption of 2.4% to 2.7% at 24 hours and as high as 6% to 7% at 60 days, which is approximately equal to or grless fail as an open sandwich material when used to cover the gingival wall in Class II restorative preparations within as little as 3 years due to dissolution.8 However, use eater than that of a conventional glass ionomer.7 As a result, use of a bioactive cement (such as a calcium silicate-based cement used as a lining or base material) as an open sandwich liner or base (despite apparently low in vitro microleakage) may not be advisable at this time. The rationale for this recommendation is derived from the fact that glass ionomers, which have even lower water solubility, neverthein a closed sandwich technique, in which a bioactive material used for pulp capping or lining is not exposed to the oral environment, appears to be acceptable.
MTA-type (complex calcium silicate-based) materials can have setting times of several hours. The relative solubility together with the prolonged setting behavior of bioactive cements requires use of selective adhesive coating agents to stabilize them during setting and reduce their dissolution as a result of exposure to the oral environment. It may prove useful to apply and photopolymerize a self-etch adhesive (eg, Xeno® III, DENTSPLY International, www.dentsply.com)9 or a self-adhesive lining material such as a RMGI liner or flowable compomer (eg, Dyract®flow, DENTSPLY International)10 over the partially set cement prior to placement of a definitive restorative.
Other dental material chemistries are being combined with these two calcium-based compositions, producing hybrid chemistries. For example, a bioactive luting cement, which has been available for the past 4 years, is a hybrid chemistry of calcium aluminate and glass ionomer (Ceramir C&B). This material is also bioactive, has good physical properties, and has been tested up to 3 years’ recall in a standard controlled clinical evaluation.11
Clinical Indications
The first bioactive materials, calcium silicate cements, were designed primarily for use as endodontic root replacement materials. However, the indications for use for these materials have expanded (Table 2), and now include root-end filling after apicoectomy, obturation in areas of internal and external root resorption, pulp capping (vital pulp therapy), lateral or furcation perforation, and apexification/apexogenesis.1 The original MTA/Portland cement formulations, such as ProRoot MTA, were originally indicated for pulp capping and later expanded the vital pulp therapy approach into vital pulpotomies for both primary and permanent teeth.
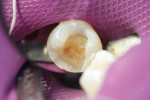
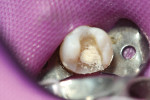
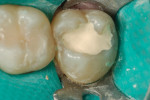
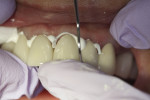
Later formulations such as tricalcium silicate cements (Biodentine) and a calcium aluminate/glass-ionomer formulation (Ceramir C&B) have broadened clinical use of bioactive cements for indications such as use as a liner/base (Figure 1 and Figure 2), temporary restorative (Figure 3), and permanent cementation-luting agent (Figure 4).2 Although use of calcium silicate cements as a root canal sealer/filling material have been mentioned almost since the material’s inception, dedicated bioactive, calcium silicate-based root canal sealers such as iRoot® SP root canal sealer (Innovative BioCeramix, www.ibioceramix.com), EndoSequence® BC Sealer™ (Brasseler USA, www.brasselerusadental.com), and BioRoot™ RCS (Septodont) have more recently appeared in this indication group.
Adhesion to Tooth Structure
Laboratory research findings indicate that the calcium silicate– and calcium aluminate–based cements display adhesion to tooth structure, specifically dentin. Recent findings indicate levels of adhesion to dentin approximately equal to that of conventional glass ionomer cements.12 In vitro measurement of adhesive retentive force of crowns cemented with a bioactive cement (Ceramir C&B) also indicates levels of retention comparable with self-adhesive resin cements.11 Nevertheless, the precise nature of the interaction between these bioactive cements and both dentin and enamel is not yet fully understood, requiring further research concerning dentin-cement interaction and dentin adhesion.12
Resin-Based Materials
The incorporation of calcium silicate fillers in a polymerizable resin matrix may produce a dental restorative material with evidence of bioactivity. One research team has reported evidence of surface apatite formation with "higher calcium-releasing ability and lower solubility" than MTA or calcium hydroxide products with a resin-calcium silicate material (TheraCal™ LC, BISCO, ). However, another another research group reports that "hydration" of this resin-calcium silicate material, "was incomplete because of the limited moisture diffusion within the material." There was also lower or no formation of calcium hydroxide, and "a lower calcium ion leaching."14 In addition, preliminary research findings in the author’s laboratory suggest that these resin-based materials containing potential bioactive additives (as a general group), can be bioactive but produce characteristics of surface apatite formation (quantity, rate of formation) that may be different than nonresin bioactive materials. A usage test utilizing a primate pulpal histology model and a single-observation point of 4 weeks (28 days), employing this same resin-calcium silicate (TheraCal LC, BISCO, www.bisco.com has indicated dentin bridge formation and acceptable healing after direct mechanical pulp exposure.15
Other restorative materials with various calcium and phosphate components (eg, Activa™ resin-modified glass ionomer restorative materials, Pulpdent, www.pulpdent.com) have also appeared in the marketplace.16 In the laboratory study referenced above, “the Activa enhanced-RMGIs demonstrated comparable flexural strength and flexural fatigue to flowable composites,” and also “demonstrated flexural strength and flexural fatigue significantly greater than other tested materials” that are classified as resin-modified glass ionomers.16 The evaluation of these methacrylate-based, resin materials (compared to longer-standing, non-resin, calcium silicate and calcium aluminate materials) is still in its early stages and needs more evaluation, especially with respect to their bioactivity and biocompatibility.
Preliminary findings regarding these newer resin-based materials should be considered in the context of the numerous published human and animal histologic findings that confirm the exceptional ability of MTA to stimulate pulpal healing and bridge formation in direct and indirect pulp exposures.2 Nevertheless, resin-calcium silicate hybrid materials may display some aspects of bioactivity and could be beneficial for their recommended intended uses. More research on these materials is clearly needed.
Future of Bioactive Materials
Bioactive materials have clearly changed and improved our ability to treat patients’ conditions. The excellent biocompatibility and regenerative properties of these materials have saved countless teeth that, under other therapeutic modalities, may have been condemned to extraction. As mechanical and physical properties improve in these various modalities and their chemistries evolve, their range of use and clinical indications may well continue to expand in our clinical materials repertoire.
Disclosure
Steven R. Jefferies, MS, DDS, PhD, holds common stock in Dentsply International, the company that markets ProRoot MTA, which was cited in this review article. Dr. Jefferies’ institution, Temple University’s Maurice H. Kornberg School of Dentistry, has received prior research funding from Doxa Dental AB, the company that markets the calcium aluminate/glass ionomer (Ceramir Crown & Bridge Cement) also mentioned in this review.
References
1. Jefferies SR. Bioactive and biomimetic restorative materials: a comprehensive review. Part I. J Esthet Restor Dent. 2014;26(1):14-26.
2. Jefferies SR. Bioactive and biomimetic restorative materials: a comprehensive review. Part II. J Esthet Restor Dent. 2014;26(1):27-39.
3. Jones JR. Review of bioactive glass: from Hench to hybrids. Acta Biomater. 2013;9(1):4457-4486.
4. Kokubo T, Kushitani H, Sakka S, et al. Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W. J Biomed Mater Res. 1990;24(6):721-734.
5. Lööf J, Svahn F, Jarmar T, et al. A comparative study of the bioactivity of three materials for dental applications. Dent Mater. 2008;24(5):653-659.
6. Gandolfi MG, Siboni F, Botero T, et al. Calcium silicate and calcium hydroxide materials for pulp capping: biointeractivity, porosity, solubility and bioactivity of current formulations. J Appl Biomater Funct Mater. 2015;13(1):43-60.
7. Singh S, Podar R, Dadu S, et al. Solubility of a new calcium silicate-based root-end filling material. J Conserv Dent. 2015;18(2):149-153.
8. van Dijken JW. A 6-year evaluation of a direct composite resin inlay/onlay system and glass ionomer cement-composite resin sandwich restorations. Acta Odontol Scand. 1994;52(6):368-376.
9. Jefferies S, Pameijer C. Hybridizing ProRoot MTA in vital pulp capping—a pilot study [abstract 0936]. Presented at: ADEA/AADR/CADR Meeting & Exhibition; March 10, 2006; Orlando, Florida.
10. Iwamoto CE, Adachi E, Pameijer CH, et al. Clinical and histological evaluation of white ProRoot MTA in direct pulp capping. Am J Dent. 2006;19(2):85-90.
11. Jefferies SR, Pameijer CH, Appleby DC, et al. A bioactive dental luting cement—its retentive properties and 3-year clinical findings. Compend Contin Educ Dent. 2013;34(spec no 1):2-9.
12. Kaup M, Dammann CH, Schäfer E, Dammaschke T. Shear bond strength of Biodentine, ProRoot MTA, glass ionomer cement and composite resin on human dentine ex vivo. Head Face Med. 2015;11:14. doi: 10.1186/s13005-015-0071-z.
13. Gandolfi MG, Siboni F, Prati C. Chemical-physical properties of TheraCal, a novel light-curable MTA-like material for pulp capping. Int Endod J. 2012;45(6):571-579.
14. Camilleri J, Laurent P, About I. Hydration of Biodentine, Theracal LC, and a prototype tricalcium silicate-based dentin replacement material after pulp capping in entire tooth cultures. J Endod. 2014;40(11):1846-1854.
15. Cannon M, Gerodias N, Viera A, et al. Primate pulpal healing after exposure and TheraCal application. J Clin Pediatr Dent. 2014;38(4):333-337.
16. Pameijer CH, Garcia-Godoy F, Morrow BR, Jefferies SR. Flexural strength and flexural fatigue properties of resin-modified glass ionomers. J Clin Dent. 2015;26(1):23-27.