Atlas Denture Comfort®
Denture retention/stabilization system is more user-friendly and less technique-sensitive for both dentists and patients.
By now, most people who have seen the many articles and advertisements regarding the denture patient should be familiar with the McGill Consensus, which states that a two-implant retained overdenture should be the first choice of treatment for the restoration of the edentulous mandible.1
As a result, a new category of implants—known as narrow-body implants—has been used extensively to accomplish the same results. Today, after being clinically scrutinized in numerous university trials, there are approximately 10 implant manufacturers that make these small-diameter implants. The procedures to place these implants are more affordable, less invasive, and easier to implement than those required for the conventional larger-diameter implant systems.
In addition to being less expensive, among the many advantages of narrow-body implants over conventional implants with locator attachments or bars are that they are generally placed with flapless surgery, can be used when there is a lack of horizontal or vertical bone, and require only one surgery without the added need for bone augmentation surgeries. All of these implant-retained systems are effective in securing the patient's denture to the point where there is a markedly improved quality of life.
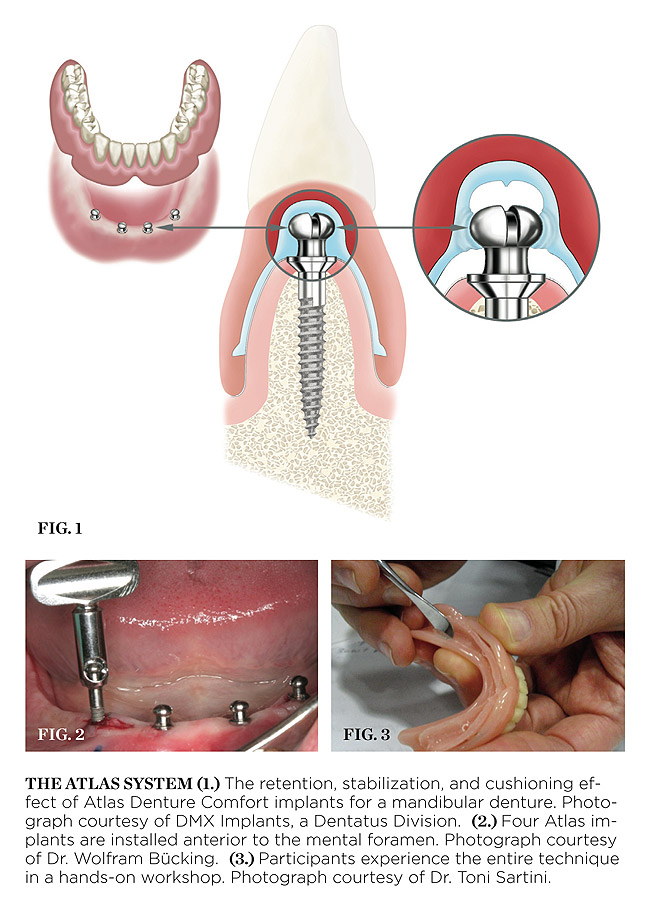
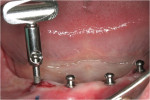
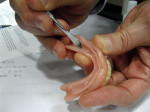
Yet, there is one system that sets itself apart from all the other systems with new technology that eliminates many of the inherent problems associated with o-rings, housings, and acrylic: Atlas® Denture Comfort™ by Dentatus USA (Figure 1, Figure 2 and Figure 3). This system is much less technique-sensitive and very user-friendly.
Any system that uses o-rings and/or housings creates difficulties for the clinician. For example, if there are parallelism discrepancies in implant placement, the retrofitted housings will transmit severe lateral stresses to the implants each time the denture is removed or placed back over the implants, which could lead to implant failure. With this chairside procedure, it is not uncommon to encounter old and worn dentures. Often, there is insufficient vertical denture height, so when drilling to retrofit the housing the lingual of the denture may be perforated or the entire denture weakened by drilling too close to the denture teeth. And, any practitioner who has ever worked on a full or partial denture can attest to the frustration of the time-consuming process of grinding away excess acrylic with the drill.
These problems and concerns are eliminated with Atlas Denture Comfort implants, manufactured by the DMX Implants division of Dentatus. In this procedure, the Tuf-Link® resilient silicone liner is firmly retained in the periphery of the denture all without the use of adhesive. The one-piece Tuf-Link reline is only 1 mm to 1.5 mm in thickness, so between the low (3-mm) profile of the implant head above the tissue and the thinness of the silicone, parallelism discrepancies are non-existent; the really good news is that the silicone is quickly and easily trimmed with scissors. Finishing the reline material and denture adjustments is quick and easy, and there is rarely a need to schedule the patient for a follow-up visit.
DMX has made it exceptionally easy to learn and implement this procedure with their half-day Hands-on Workshops (HOW). Every dentist already has the skill to surgically and safely place four Atlas implants anterior to the mental foramen; only the protocol needs to be learned. DMX has an extensive schedule of hands-on workshops to learn this technique. Participants of these workshops perform each step from A to Z and return to their practices with a professional model that can be used for patient case presentation and staff education and training. For more information or workshop schedules, see the company contact information below. All workshop participants receive free listings of their practices on the consumer website, www.DentureComfort.com.
Reference
1. McGill Consensus, McGill University. 2002
For more information, contact:
Dentatus USA
Phone: 800-323-3136
www.dentatus.com.
Disclaimer
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry.