Bonding a Veneered Zirconia Anterior Fixed Partial Denture
Abstract:
This case report documents the rationale and procedure for bonding a veneered zirconia restoration. A three-unit zirconia fixed partial denture (FPD) was fabricated with facial and incisal porcelain veneering. The intaglio surface of the prosthesis was abraded with 50-µm aluminum oxide (Al2O3) particles and coated with one layer of a 10-methacryloxydecyl dihydrogen phosphate (MDP) primer. The FPD was tried into the mouth and occlusal adjustment was performed with a fine grit diamond, and then it was polished with zirconia polishing points. The intaglio surfaces of the abutment crowns were cleaned with 37% phosphoric acid and rinsed. A self-etch adhesive was applied to the tooth preparations and light-cured, and the crowns were filled with cement and seated. Excess cement was immediately wiped away with a brush, followed by spot-curing of the margins. The case demonstrates that, when properly designed, veneered zirconia restorations offer acceptable esthetic and mechanical properties for anterior FPDs.
Zirconia has become a ubiquitous material in dentistry. Research regarding the wear properties of zirconia has shown that it is more friendly to opposing enamel than veneering porcelain.1 Its wear properties dictate optimal methods for designing a coping in a veneered zirconia restoration and making intraoral adjustments. Unlike previous dental ceramics, such as lithium disilicate or leucite, zirconia does not contain glass and cannot be etched with hydrofluoric acid or bonded with silane. Therefore, new materials and techniques have been developed to bond zirconia to tooth structure. This case report will demonstrate the techniques and theory for designing, bonding, and adjusting a veneered zirconia restoration.
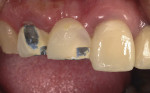
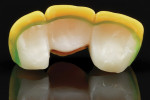
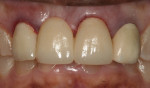
A 58-year-old female patient presented with a fractured metal-ceramic fixed partial denture (FPD) (Figure 1). The patient reported that she fractured the existing FPD while opening a bottle cap with her teeth. Her medical history included allergic rhinitis, bipolar depression, cerebrovascular disease (stroke), gastroesophageal reflux disease (GERD), hepatitis C, human immunodeficiency virus (HIV), and hypertension. Significant dental history included extraction of tooth No. 8 more than 5 years previously.
The patient requested an esthetic replacement for her fractured anterior restoration. Due to financial limitations, a dental implant for No. 8 was not considered as a treatment option. The patient was also not interested in a removable prosthesis. Based on her previous medical condition, there was no contraindication for the placement of an FPD. The choice to restore this case with a veneered zirconia prosthesis was based on the desire to obtain the incisal translucency afforded by veneering porcelain and the high strength but white hue present in a zirconia core. Other alternatives were a porcelain-fused-to-metal (PFM), monolithic/veneered lithium-disilicate, or monolithic zirconia FPD. PFM FPDs are tough/durable due to the ability of metal to deform without fracturing; however, the dark color of the metal coping compromises esthetics around the gingival margin and underlying hue of the restoration. Monolithic or veneered lithium disilicate could have been used for this case; however, lithium disilicate has a lower flexural strength and fracture toughness than zirconia.2 Monolithic zirconia was not used because it is too opaque to mimic translucent incisal edges. The patient expressed a desire to eventually replace tooth No. 10 with an all-ceramic crown due to the opacity of the restoration and visible facial metal margin. Her intraoral examination revealed that she had a deep overbite and canine guidance. Prior to removing the previous crowns, a custom anterior guide table was fabricated.
Preparation and Prosthesis Design
This FPD was prepared with a minimum reduction of 1.2 mm based on recommendations to reduce veneered zirconia to 1.2 mm (monolithic zirconia may be reduced to 0.6 mm).3 A chamfer margin was used for the preparation (Figure 2). Crowns with rounded chamfer margins have greater fracture strength than those with sharp shoulder margins.4 Previous in-vitro studies have shown that zirconia restorations fabricated on shoulderless (knife-edge) preparations produced greater fracture strength than those on chamfered preparations.5,6 Analysis of fractured crowns in-vivo, however, demonstrated that zirconia coping fracture typically occurs at the margin, either interproximally or lingually/palatally.7 For this reason, the authors chose to prepare a chamfer margin to allow adequate bulk of zirconia at the margin. All line angles were rounded to prevent over-milling of the internal aspects of the restoration (ie, areas where the CAM bur was wider than the tooth preparation).
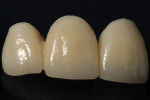
The prosthesis was designed to full anatomic contour in areas of function, leaving room for facial and incisal veneer porcelain in non-stress–bearing areas (Figure 3 and Figure 4). If the restoration were to be completely layered it would be important to use an anatomic coping. An anatomic coping implies that the external surface of the coping mimics the contours of the final restoration. This coping design allows an even thickness of veneering porcelain, which is adequately supported by the core zirconia. It has been shown that anatomic copings improve the fracture strength of zirconia crowns by 30% to 40%.8 The laboratory should be advised to pay close attention to the design of the anatomic copings for areas of over-reduction, such as cusps and marginal ridges of posterior teeth.
The veneer was applied as a pressed feldspathic porcelain, which was formulated to match the coefficient of thermal expansion of zirconia to help prevent chipping of the porcelain.9,10 In this case, the lingual of the crown was not veneered, because this portion of the crown would be in contact with the opposing dentition (Figure 5). Several studies have proven that polished zirconia causes less opposing enamel wear than feldspathic porcelain.11,12 The FPD connectors were designed to provide a connector area of at least 9 mm2 with at least 3 mm of height.
The Decision to Cement or Bond Restoration
A ceramic crown may be cemented to the preparation, thereby providing micromechanical retention, or bonded, in which case a chemical bond is created. The decision whether to cement or bond the restoration is based on the need for either retention or additional strength. Bonding, which will increase a crown’s retention, should be performed for short or over-tapered preparations. Bonding to tooth structure can strengthen a porcelain, leucite, or lithium-disilicate crown; however, bonding is not necessary to strengthen high-strength ceramics such as zirconia.13,14 Bonding is more technique-sensitive than cementing, as it is more affected by moisture. In this case, the authors chose to bond the FPD due to the ease of achieving ideal isolation. Cementing this restoration with a resin cement (without the use of an adhesive) or a resin-modified glass-ionomer cement would have also been acceptable.
Several types of resin cement could have been used to bond the zirconia FPD. In this situation, a modified total-etch system was utilized, consisting of a separate self-etch adhesive and cement. However, simplified cement systems could have been used, such as self-etch (primer plus cement) or self-adhesive (only cement) cements. Self-etch or self-adhesive cements, which provide similar bonding effectiveness but require less steps, are useful for bonding restorations in areas with less-than-ideal moisture control.15
Zirconia Preparation, Try-in, and Adjustment
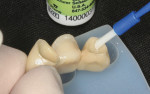
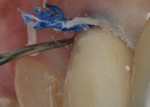
When the FPD was received from the dental laboratory, the internal aspects of the abutments were immediately air-particle abraded with 50-µm alumina particles for 30 seconds to increase micromechanical retention (Figure 6).16-18 Unlike with lithium disilicate, air abrasion of zirconia does not weaken the crown.16,19 The inside surface of both abutments were then treated with a priming agent (Z-Prime™, Bisco, Inc., www.bisco.com) (Figure 7). Z-Prime is a zirconia primer that contains 10-methacryloxydecyl dihydrogen phosphate (MDP), a difunctional monomer that bonds to both zirconia and methacrylates in resin cements. Other similar primers that can be used to bond zirconia to resin cements include Monobond Plus (Ivoclar Vivadent, www.ivoclarvivadent.com) and Clearfil™ Ceramic Primer (Kuraray, www.kuraraydental.com). Particle abrasion and MDP primers both improve the bond strength to zirconia, and when used in combination, have a compounded effect.20 Following the application of the MDP primer, the FPD was tried into the mouth.
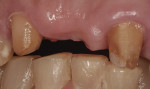
No internal adjustments to the FPD were necessary. If internal adjustment is needed, it is preferable to make adjustments to the preparation rather than adjust the intaglio surface of the crown. Because ceramics fail in tension and the intaglio surfaces of crowns are subjected to tensile forces, avoiding internal adjustment of ceramic crowns prevents sites of potential crack initiation. The use of a try-in paste can aid in determining where to adjust the preparation. All external adjustment to the FPD was performed with a fine-grit red-striped diamond bur. Following final adjustment, the zirconia was polished with a zirconia polishing system (Dialite® ZR, Brasseler USA, www.brasselerusadental.com). Several studies have shown that polishing zirconia produces less wear on opposing enamel than glazing.11,12 If the clinician prefers to glaze the zirconia, it should first be polished prior to glazing, as this will produce a smoother surface when the glaze wears off. The incisal embrasures were not “opened up” with a disc, as this would introduce flaws into the tensile side of the FPD. If adjustment of the incisal embrasure area is necessary, this area should be opened up with a diamond bur and then carefully polished afterwards.
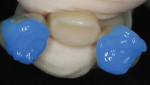
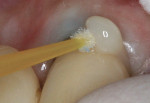
After try-in was complete, the abutment crowns were cleaned with 37% phosphoric acid for 60 seconds (Figure 8) and rinsed to remove any contaminants from saliva or the try-in paste. A cleaning paste (eg, Ivoclean, Ivoclar Vivadent) can also be used to clean the inside of the crown. This is particularly useful if the inside of the crown is contaminated prior to application of the MDP primer since phosphoric acid is thought to hinder the bonding of MDP to zirconia.21,22
Bonding
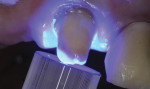
Isolation was obtained by placing a cotton roll in the labial vestibule. A portion of the crown margin was obstructed by a piece of gingival tissue; therefore, a retraction cord was placed to allow complete access to the margin. For this case, the authors chose a universal bonding agent (All-Bond Universal®, Bisco, Inc.), which can be used in a total-etch, selective-etch, or self-etch mode; they used a self-etch method. Some self-etch adhesives require a dual-cure activator to be mixed with the adhesive prior to use with a dual-curing cement. The universal adhesive used here, however, does not require a dual-cure activator. The preparations received two coats of adhesive and were scrubbed for 15 seconds (Figure 9). The adhesive was then air-dried for 15 seconds until there was no visible movement of the adhesive. Prior to light-curing the adhesive, the preparations were examined to confirm that there was no puddling of the adhesive, which would have prevented complete seating of the crowns. The adhesive was light-cured for 10 seconds (Figure 10). (Author’s note: The manufacturer’s instructions should be referenced to confirm that light-curing the adhesive is necessary, as some adhesive/cement combinations, such as Scotchbond™ Universal/RelyX™ Ultimate [3M ESPE, www.3MESPE.com], contain chemical initiators in the cement that polymerize the adhesive.)
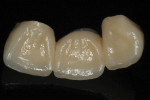
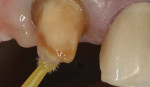
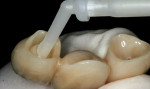
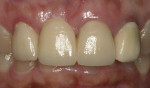
The abutment crowns were then filled with a dual-curing (light and chemical cure) resin cement (Duo-Link Universal™, Bisco, Inc.) (Figure 11). Light-cured cements provide higher bond strength and more color stability than chemically cured cements; however, the opacity of zirconia prevents light penetration and requires a cement with a chemical cure mechanism.23 The FPD was seated (Figure 12), and excess cement was lightly removed from the margins with a microbrush (Figure 13). The margins were spot-cured for 3 seconds at every quarter surface (mesio-facial, disto-facial, disto-lingual, and mesio-lingual) (Figure 14). The patient was then instructed to bite in maximum intercuspation for 60 seconds. The remainder of the excess cement was then cleaned with a scaler, and the retraction cord was removed (Figure 15). A final radiograph was taken to confirm complete seating of the FPD, and the occlusion was again evaluated with articulating paper. Figure 16 shows the restoration immediately postoperative.
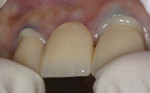
At the patient’s 2-month follow-up appointment, the temporary gingival inflammation associated with the restorative procedures (ie, use of packing cord and removal of cement) had healed. The patient was retaining plaque at the disto-facial surface of tooth No. 7, and erythema of the gingiva was noted (Figure 17). Oral hygiene instructions were reinforced. The gingival tissues had rebounded and the cervical embrasures were adequately filled with tissue. The patient was satisfied with the esthetic outcome of her treatment.
Conclusion
If properly designed—that is, with occlusion in zirconia and with use of long connectors and anatomic copings—partially veneered zirconia restorations have acceptable esthetic and mechanical properties for anterior fixed partial dentures. Zirconia restorations can be adhesively bonded to tooth structure by modifying the surface of the zirconia (via particle abrasion and MDP primer) and using a resin cement.
About the Authors
Nathaniel C. Lawson, DMD, PhD
Assistant Professor, Department of Clinical and Community Sciences, Division of Biomaterials, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama
Ramakiran Chavali, BDS, MS
Assistant Professor, Department of Restorative Sciences, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama
John O. Burgess, DDS, MS
Assistant Dean for Clinical Research, Department of Clinical and Community Sciences, Division of Biomaterials, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama
References
1. Lawson NC, Janyavula S, Syklawer S, et al. Wear of enamel opposing zirconia and lithium disilicate after adjustment, polishing and glazing. J Dent.2014 Sep 23. doi:10.1016/j.jdent.2014.09.008. [Epub ahead of print].
2. Lawson NC, Burgess JO. Dental ceramics: a current review. Compend Contin Educ Dent.2014;35(3):161-166.
3. Baladhandayutham B, Beck P, Litaker MS, et al. Fracture strength of all-ceramic restorations after fatigue loading [abstract]. J Dent Res. 2012;91(spec iss A). Abstract 24.
4. Jalalian E, Atashkar B, Rostami R. The effect of preparation design on the fracture resistance of zirconia crown copings. J Dent (Tehran). 2011<;8(3):123-129.
5. Beuer F, Aggstaller H, Edelhoff D, Gernet W. Effect of preparation design on the fracture resistance of zirconia crown copings. Dent Mater J. 2008;27(3):362-367.
6. Reich S, Petschelt A, Lohbauer U. The effect of finish line preparation and layer thickness on the failure load and fractography of ZrO2 copings. J Prosthet Dent. 2008;99(5):369-376.
7. Oilo M, Hardang AD, Ulsund AH, Gjerdet NR. Fractographic features of glass-ceramic and zirconia-based dental restorations fractured during clinical function. Eur J Oral Sci. 2014;122(3):238-244.
8. Lawn BR, Deng Y, Thompson VP. Use of contact testing in the characterization and design of all-ceramic crownlike layer structures: a review. J Prosthet Dent. 2001;86(5):495-510.
9. Fischer J, Stawarzcyk B, Trottmann A, Hammerle CH. Impact of thermal misfit on shear strength of veneering ceramic/zirconia composites. Dent Mater. 2009;25(4):419-423.
10. Blatz MB, Bergler M, Ozer F, et al. Bond strength of different veneering ceramics to zirconia and their susceptibility to thermocycling. Am J Dent. 2010;23(4):213-216.
11. Burgess JO, Janyavula S, Lawson NC, et al. Enamel wear opposing polished and aged zirconia. Oper Dent.2014;39(2):189-194.
12. Janyavula S, Lawson N, Cakir D, et al. The wear of polished and glazed zirconia against enamel. J Prosthet Dent. 2013;109(1):22-29.
13. Heintze SD, Cavalleri A, Zeiiweger G, et al. Fracture frequency of all-ceramic crowns during dynamic loading in a chewing simulator using different loading and luting protocols. Dent Mater. 2008;24(10):1352-1361.
14. Al-Wahadni AM, Hussey DL, Grey N, Hatamleh MM. Fracture resistance of aluminum oxide and lithium disilicate-based crowns using different luting cements: an in vitro study. J Contemp Dent Pract. 2009;10(2):51-58.
15. Inokoshi M, De Munck J, Minakuchi S, van Meerbeek B. Meta-analysis of bonding effectiveness to zirconia ceramics. J Dent Res.2014;93(4):329-334.
16. Yi YA, Ahn JS, Park YJ, et al. The effect of sandblasting and different primers on shear bond strength between yttria-tetragonal zirconia polycrystal ceramic and a self-adhesive resin cement. Oper Dent.2014 Aug 1. [Epub ahead of print].
17. Shin YJ, Shin Y, Yi YA, et al. Evaluation of the shear bond strength of resin cement to Y-TZP ceramic after different surface treatments. Scanning.2014;36(5):479-486.
18. Zandparsa R, Talua NA, Finkelman MD, et al. An in vitro comparison of shear bond strength of zirconia to enamel using different surface treatments. J Prosthodont.2014;23(2):117-123.
19. Amaral M, Valandro LF, Bottino MA, Souza RO. Low-temperature degradation of a Y-TZP ceramic after surface treatments. J Biomed Mater Res B Appl Biomater. 2013;101(8):1387-1392.20013;110(4):274-280.
21. Chen L, Suh B, Shen H. Minimize the contamination of zirconia restoration surface with saliva [abstract]. J Dent Res. 2013;92(spec iss A). Abstract 1654.
22. Koch S, Bock T, Meirer F, et al. Cleaning efficacy of a universal cleaning agent on ceramic surfaces [abstract]. J Dent Res. 2013;92(spec iss A). Abstract 413.
23. Latta MA, Kelsey WP 3rd, Kelsey WP. Effect of polymerization mode of adhesive and cement on shear bond strength to dentin. Am J Dent. 2006;19(2):96-100.