Do Bib Clips Pose a Cross-Contamination Risk at the Dental Clinic?
An excerpt from Compendium of Continuing Education in Dentistry
Dental instruments are either disposable or meant for multiple patient uses. Multiple-use items include dental mirrors, ultrasonic units, and patient napkin holders (ie, bib chains). These items require sterilization or disinfection procedures to reduce the risk of transmitting infection. Items contacting intact skin only, such as bib chains, have a relatively low risk of transmitting infection, and as such may be subject to Centers for Disease Control and Prevention (CDC) regulations requiring the use of intermediate- or low-level disinfectants.1 A previous study by Molinari demonstrated that bib chains harbored bacteria on their surface following dental treatments.2 Furthermore, that study found that a brief wipe of the chains with an EPA-approved, intermediate-level disinfectant between patient appointments reduced, but did not eliminate, the microbial load found on these chains.2 Similarly, the presence of bacterial contamination on bib clips after treatment and after disinfection was also found in a study performed at the Dental School of Witten/Herdecke University in the Witten area in Germany.3 However, no published studies document the bacterial loads on bib clips following different dental treatments in specialty clinics, such as Endodontics and Orthodontics, and the effectiveness of disinfection of bib clips after patient care.
The primary objectives of this study were as follows: 1) to determine the presence of bacterial contaminants on two types of clips of reusable patient bib chains following dental procedures at Endodontics and Orthodontics clinics; and 2) to evaluate the effectiveness of disinfection of these clips using a commercially available disinfectant.
Two types of bib clips were used in this study—metal clips that are attached to metal bib chains (Henry Schein napkin holder, Henry Schein, Inc., www.henryschein.com) and rubber-faced metal clips that are attached to rubber bib chains (Slip-Nots™, Kerr TotalCare, Metrex Research Corp., www.metrex.com). Bib clips were sampled during the morning (AM) and afternoon (PM) sessions at the Endodontics and Orthodontics department clinics at Tufts University School of Dental Medicine (TUSDM). In each department, a total of 40 chains were studied; among them, 20 had metal clips and 20 had rubber-faced metal clips. From each chain, only the clips were sampled for bacterial contamination. The study was conducted according to an approved protocol by Tufts Medical Center and Tufts University Health Sciences Institutional Review Board (IRB) office.
Bacterial Sample Collection
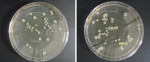
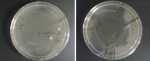
Bib clips were sampled for bacterial contaminants immediately following dental treatments provided to patients. Sampling was done both before and after the clips were disinfected by wiping them with CaviCide® wipes (CaviWipes®, Metrex Research Corp.) and letting them dry according to the manufacturer’s instructions and the disinfection protocol followed at TUSDM. Immediately after patient dismissal, bacterial contaminants were directly transferred from the clips onto Trypticase soy agar plates in the operatory. A direct transfer was achieved by firmly pressing each of the four sides of each clip onto a separate area on a nutrient agar plate.4 Thereafter, the clips were disinfected by wiping them with CaviCide and sampled again in the same manner for bacterial contaminants on a separate agar plate. Plates were then transferred to a microbiology laboratory at TUSDM and incubated aerobically at 37°C for 48 hours. Bacterial samples were collected from both the right and left clips of each chain, and the total number of bacterial colonies developed from inoculation was considered to be one sample. For each plate, the number of bacterial colonies that grew on the agar was recorded, and representative plates were imaged (Figure 1 and Figure 2).
Discussion
This study sought to determine the presence of bacterial contaminants on bib clips and the effectiveness of disinfecting these clips. Both the Endodontics and the Orthodontics specialty clinics participated in this study, representing a wide spectrum of patients, dental procedures, and appointment times. The procedures performed in the Endodontics clinic included consultation, nonsurgical endodontic therapy, and orthograde retreatment. Procedures performed in the Orthodontics clinic included consultation, impressions, bracket and band placement, appliance adjustment, orthodontic wire adjustments, as well as braces removal, polishing, and debriding.
Similar to the findings reported by Molinari,2 bib clips sampled in both clinics exhibited bacterial contamination immediately after dental procedures. Molinari’s study, however, does not identify the types of clinics or treatments performed. Following treatment and before disinfection, the present study found a greater microbial load on bib clip samples from the Orthodontics clinic when compared to samples from the Endodontics clinic.
This finding raises the question as to why a higher level of bacterial load is found in the orthodontic setting as opposed to the endodontic setting. The statistically significant difference in bacterial contamination found on bib clips following treatment is likely due to the nature of the treatments provided to patients in these clinics. It may result primarily from the use of rubber dams during an endodontic treatment, which limits salivary contamination of the bib clips. In orthodontics, a significant aspect of the treatment involves entering and re-entering the oral cavity with appliances, thus bacterial contamination from the gloves could be transferred to the patient’s bib clips. If the rubber dam in the endodontics setting reduces the bacterial load, it must be understood that the use of a rubber dam in an orthodontics setting is actually impractical. In addition, this difference may also result from the application and adjustment of the clips by the patients themselves in the Orthodontics clinic.
Collectively, the study’s findings suggest that due to the nature of the dental treatment, different clinics may be exposed to different bacterial loads and bacterial species on the clips, and, therefore, to a different contamination risk.
The present study also revealed a significant and clinically important difference in bacterial loads between the two types of bib clips that were sampled following treatments. In comparison to the metal bib clips, the rubber-faced metal clips demonstrated a significantly greater number of bacterial contaminants. One possible explanation is that due to its nature, the rubber material may exhibit microscopic porosities, which can support bacterial adherence and, in general, may decrease disinfection efficiency. In contrast, the smooth surface of metal bib clips may reduce bacterial adherence and, in general, their disinfection may be more effective.
Molinari’s study suggested that bacterial loads on bib chains can occur due to extended contact of the bib chain with the patient’s skin, exposure of the chain to microbe-containing aerosols during treatment, and handling the chains with contaminated gloves during the treatment.2 By sampling only the bib clips, the present study excluded bacterial contaminants that can be found on the rest of the chain, specifically the areas that are in close contact with the patient’s skin. Thus, its findings represent the actual bacterial load that resides on the bib clips due to the dental treatment and/or the surrounding environment—ie, airborne bacteria—after dental treatment and after disinfection. However, immediately after treatment, skin commensals are likely to be found on the bib clips from young patients adjusting the bib chain in the Orthodontics clinic.
In addition, while no significant difference was found between the clinics or the type of clip in the post-cleaned samples, the P value for the comparison of bacterial counts obtained in the morning and afternoon sessions could be considered approaching significance. Clinically, these results suggest that bacteria may be accumulating on the bib clips between the morning and the evening sessions, as patients are treated throughout the day.
Finally, the findings of the present study demonstrate that despite the initial, marked difference in bacterial load on pre-cleaned bib clips in both clinics and on both clip types, effective bacterial reduction is achieved by the disinfection protocol. However, whereas 70% to 80% of the clips were left with no detectable bacteria after disinfection, 20% to 30% of the disinfected clips still harbored bacterial contaminants on their surfaces that may or may not pose a cross-contamination risk during patient care at the dental clinic. These results are similar to those recently found in a study performed by the University of North Carolina at Chapel Hill’s School of Dentistry.4
In the current study, the mere presence of bacterial contaminants on the disinfected clips has not been shown to be an infectious disease problem. However, the identity of the bacteria found on the pre- and post-disinfected clips remains unknown. To address cross-contamination issues by bib clips in patient care in different clinics, further investigation should be performed to determine the species and strains of the bacterial contaminants present on the bib clips after dental treatment and following disinfection.
Conclusions
Results of this study indicated that following dental procedures, a significantly greater bacterial burden is present on bib clips in the Orthodontics clinic as compared to those in the Endodontics clinic. Furthermore, when metal clips are compared to clips with rubber facings, greater numbers of bacteria were found on the latter. While disinfecting the clips was proven to be significantly effective, 20% to 30% of both clip types still harbored bacteria, posing a potential yet undetermined risk for cross-contamination.
This novel study design, which was implemented in specialty clinics, establishes baseline data for further analyses in additional clinics. Moreover, further research should be performed in order to determine the identity of bacterial species in samples from both pre- and post-disinfected bib clips and to determine whether or not they include disease-causing bacterial species.
Disclosure
The study was sponsored in part by Dux Dental (www.duxdental.com).
For the full research report and tables, read the Compendium article here.
References
1. Centers for Disease Control and Prevention. Guidelines for infection control in dental health care settings. MMWR. 2003;52(RR-17):1-61.
2. Molinari JA. Microbial contamination of patient napkin holders. The Dental Advisor. June 2010, Number 29.
3. German Study Finds Bacteria on 70% of Dental Bib Holders [press release]. S. Zimmer, University of Witten/Herdecke in Witten, North Rhine-Westphalia, Germany. Dr Bicuspid Hygiene Community; March 19, 2012.
4. Study Finds Bib Chain Potential Source of Bacteria [press release]. Oral Microbiology Laboratory, Chapel Hill’s School of Dentistry, University of North Carolina. Dental Health Magazine; March 16, 2010.