Reduction of Two Red Complex Bacteria by Sustained-Release Doxycycline and Correlation to Improvement in Mean Pocket Depth
By Clay B. Walker, PhD; Kenneth C. Godowski, MSc; Steven Garrett, DDS; Craig Wesselman, MS
Abstract
Objective: The objective was to evaluate the effects of an 8.5% sustained-release doxycycline-containing polymer formulation (SRDF) on deep pockets (pocket depth [PD] ≥ 7 mm) in chronic periodontitis. Total bacterial counts were used to estimate the number of viable bacteria present before treatment and for up to 6 months posttreatment.
Methods: All sites had PD ≥ 5 mm and bled on probing in 23 subjects who received treatment with SRDF. There was an average of 8.7 teeth or 23 sites for each subject. One deep pocket (≥ 7 mm) in each subject was selected for monitoring. This site was sampled prior to treatment and at 7, 21, 91, and 182 days after SRDF placement. The primary endpoints were changes in the viable counts of two red complex species, Porphyromonas gingivalis and Tannerella forsythia. Secondary endpoints were changes in the number of total anaerobic bacteria recovered and changes in PD.
Results: Relative to baseline, SRDF reduced the proportions of P gingivalis and T forsythia by 88% and 99%, respectively, at day 7. At the conclusion of the monitoring period—182 days—P gingivalis and T forsythia were present but at 19% to 20% of the pretreatment values. Total anaerobic counts were reduced by 96% at day 7; by 87% at day 21; and by 75% and 68% at days 91 and 182, respectively. Mean PD for the sample sites (initially ≥ 7 mm) was reduced 2 mm by day 21, and this difference persisted throughout the study.
Conclusions: This study demonstrates SRDF has a significant effect, not only statistically but also microbially and clinically, on deep periodontal sites in patients with chronic periodontitis. SRDF significantly reduced the number of red complex bacteria P gingivalis and T forsythia, as well as the number of total anaerobic bacteria. By day 21, PD was reduced by 2 mm, and this reduction was maintained for at least 6 months posttherapy.
The local delivery of an antimicrobial agent directly to the diseased periodontal pocket offers several advantages versus systemic delivery of an antibiotic. Namely, the drug would be primarily restricted to the area of infection and much higher concentrations could be achieved. In recent years, this has become increasingly important. One reason is because of a real concern about the development of antibiotic resistance. The second reason is the recognition that bacterial biofilms such as subgingival plaque are many orders of magnitude more antibiotic-resistant than the same bacteria grown planktonically. The use of a local delivery, continuous-release device enables the drug to be delivered to a defined region at much higher concentrations than can be safely achieved systemically. The American Academy of Periodontology has discouraged routine use of systemic antibiotic therapy for chronic periodontitis to reduce the potential rise of antibiotic-resistant bacterial strains and the emergence of opportunistic pathogens.1 The first commercial formulation and delivery mechanism of a local delivery drug was developed by J.M. Goodson in the late 1970s, with commercial marketing in the early 1980s. This consisted of tetracycline HCl placed into "hollow fibers" that would gradually release the drug over time.2 These fibers were physically placed around the tooth and pressed down between the tooth and gingival sulcus wall. Following the development of the tetracycline-containing fibers, various controlled-release polymers were developed and impregnated with different antimicrobial agents, including metronidazole, minocycline, doxycycline, and chlorhexidine. Many clinical trials have been conducted, testing various local delivery preparations mostly as adjuncts to mechanical therapy consisting of scaling and root planing (SRP).3-5 Conflicting results have been reported; some studies have reported beneficial effects while others have found no significant advantage relative to SRP. SRP by itself is an effective treatment for most patients with chronic periodontitis. Thus, it may be difficult to demonstrate an adjunctive effect with a locally delivered antimicrobial agent combined with SRP.
In this study, the authors were interested in the effects, clinically and microbially, obtained in patients with chronic periodontitis that was treated with a preparation of 8.5% doxycycline hyclate in a controlled-release, biodegradable vehicle (Atridox®, Tolmar, www.tolmar.com) without adding SRP. Each patient served as his or her own control. The primary outcome was the effect on the red complex bacteria Porphyromonas gingivalis and Tanneralla forsythia. Secondary outcomes were the effects obtained on total anaerobic counts and probing pocket depth (PD).
Materials and Methods
Study Population
The study protocol was approved by the Institutional Review Board of Hill Top Research Laboratories, West Palm Beach, Florida. Participants had been recruited via advertisements. Twenty-three patients between the ages of 25 and 75 years participated for the 6-month period. Entry criteria required each enrollee to present with chronic adult periodontitis characterized by having four nonadjacent periodontal pockets with a PD ≥ 5 mm and that bled on gentle probing in each of two quadrants of the mouth. Enrollees were in good health based on medical history, blood pressure and heart rate readings, and clinical judgment. Females were neither pregnant nor lactating and were either postmenopausal, sterile, or using an acceptable method of birth control. Exclusion criteria included periodontal therapy in the previous 3 months, systemic or local antibiotic therapy within 3 months of enrollment, a condition that required prophylactic antibiotics, a systemic condition that could influence the course of periodontal disease (eg, diabetes mellitus, autoimmune disease), gross dental decay, and allergies to any tetracycline.
Treatment
All participants were assigned to receive doxycycline treatment consisting of the local application of an 8.5% (w/w) sustained-release doxycycline-containing polymer formulation (SRDF) as a stand-alone treatment. They did not receive SRP. All sites with PD ≥ 5 mm that bled on probing were treated with the doxycycline-containing polymer (average of 8.7 teeth or 23 sites per patient). The treated sites were covered with a periodontal dressing that was not removed for 7 days. Baseline indices, both clinical and microbial, were collected prior to treatment with each participant serving as his or her own control.
Sampling
In each participant, a single site with PD ≥ 7 mm in one of the treatment quadrants was pre-selected for microbial sampling. Subgingival plaque samples from these sites were collected at the Hill Top Research dental operatories prior to the determination of periodontal clinical indices, immediately before treatment initiation (baseline), and at days 7, 21, 91, and 182 following treatment initiation. Subgingival plaque samples were collected using two fine endodontic paper points. The two points were simultaneously inserted into the gingival crevice following the removal of any apparent supragingival plaque. The points were inserted until resistance was encountered and then left in place for 10 seconds. The points were removed and immediately placed together into a tube containing 6 mL of prereduced, anaerobically sterilized (PRAS) Ringers solution,6 which was flushed while open with a gentle stream of oxygen-free N2 gas to maintain anaerobiosis. Plaque samples were placed on ice and delivered to the Hill Top Research microbiology laboratory for processing immediately after collection.
Sample Processing
The samples were dispersed by vortexing and sonication and then serially diluted in PRAS Ringers solution under anaerobic conditions. The samples were spiral-plated in duplicate onto a CDC anaerobic blood agar (CDC-ANA), an enriched nonselective media, for an estimate of total anaerobic count and P gingivalis count. These plates were incubated anaerobically at 35°C for 5 to 10 days. The samples were also spiral-plated in duplicate onto tryptic soy blood agar-containing N-acetyl muramic acid (TSBA-NAMA) for an estimate of T forsythia count. These plates were incubated anaerobically at 35°C for 10 to 12 days. All microbiology determinations were made based on viable counts recovered. Because only viable bacteria were of interest, neither DNA-DNA hybridization nor specific DNA probes were used because these would have detected an expected high number of killed bacteria 7 days after placement of the drug and possibly at 21 days.
Determination of Bacterial Counts and Bacterial Identification
After the prescribed incubation period, the CDC-ANA and TSBA-NAMA plates were examined for P gingivalis and T forsythia. Black glistening colonies on CDC-ANA plates that did not fluoresce under shortwave ultraviolet light, were confirmed as Gram-negative bacilli by Gram stain, and tested positive for trypsin-like activity were identified as P gingivalis. T forsythia was identified as small white- or pink-speckled raised colonies on TSBA-NAMA plates, Gram-negative bacilli by Gram stain, and trypsin-positive.
Pocket probing depths
Prior to the study, the clinical tools were calibrated to levels of accuracy and reproducibility for pocket probing depth measurements consistent with current standards. A periodontal probe determined the distance (millimeters) from the gingival margin to the bottom of the gingival pocket. Numbers were rounded to the nearest whole millimeter and taken at six location points around the circumference of each tooth. Seven to 14 days prior to the start of the study, patients were screened for qualifying periodontal sites, and this PD data was used as a baseline. At day 0 (study start), only microbial samples were taken. Due to the presence of SRDF in the sample pockets at 7 days, PD measurements were not taken at that time. PD was determined at days 21, 91, and 182.
Data Analysis
The primary endpoints in this study were changes in viable counts of P gingivalis and T forsythia relative to baseline. Secondary end points consisted of changes in total recovered anaerobic counts and PD measurements relative to baseline. Changes from baseline for P gingivalis, T forsythia, and PD (sampling sites ≥ 7 mm) were analyzed by performing a log conversion of the raw microbial counts and then testing for statistical significant differences using a nonparametric paired sign test to detect differences between baseline and each posttreatment point (day 7, 21, 91, and 182). P values ≤ .05 were considered to represent statistically significant differences from baseline. PD measurements were analyzed using a paired t-test on ranked PD measurements (nonparametric) to detect differences from baseline and each posttreatment point (day 21, 91, and 182).
Results
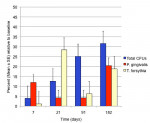
Figure 1 presents the mean counts for total anaerobic bacteria, P gingivalis, and T forsythia recovered from the sample sites at baseline and at 7, 21, 91, and 182 days following the placement of SRDF. Figure 2 presents the same data as a percentage of the baseline values. The total anaerobic colony-forming units (CFUs) recovered were significantly reduced (96%) at day 7 relative to baseline. The total anaerobic CFUs at 21, 91, and 182 days after the SRDF placement represented, respectively, 12.6%, 25.1%, and 31.6% of the counts present at baseline (Figure 2). The effects of SRDF on the two specific red complex species monitored are given in Table 1 and Figure 1. Table 1 and Figure 1 provide the mean CFUs obtained for each species at each time point. In addition, Table 1 gives the number of sample sites in which the target organisms were detected. Figure 2 expresses these data as the percent present relative to baseline. The numbers of both species were significantly reduced at all time points relative to baseline. However, even at 7 days, neither species was totally eliminated. There was a reduction of 88% of the total P gingivalis bacterial population at day 7 and, at day 21, a further reduction equivalent to 96% relative to baseline levels. This reduction was maintained through day 91, and only at day 182 was a repopulation trend seen. Table 1 lists the number of sites where P gingivalis was detected out of the total number of sites sampled. The number of P gingivalis-positive sites fell from 11 at baseline to 6 immediately after treatment and was maintained at this level for the study duration. The greatest effect on T forsythia was at day 7 when a mean reduction of 99% was noted relative to baseline. Throughout the remainder of the 6-month period, there was some variability in T forsythia levels with reductions from baseline levels ranging from 72% to 94%. In Table 1, the number of T forsythia-positive sites fell from 14 at baseline to 10 immediately after treatment and was maintained at approximately this level for the study duration. SRDF was effective at reducing the total anaerobic counts and both of the target species, P gingivalis and T forsythia, which resulted in significant decreases for the duration of the study of 6 months. The relationship of these reductions to the mean PD and statistical significance is illustrated in Figure 3 and Figure 4. In Figure 3, the P gingivalis mean CFU is plotted as a line graph with counts on the right y-axis. The mean pocket depth in millimeters is plotted as a bar graph with mm on the left y-axis. Figure 4 presents the T forsythia data in the same manner. The mean CFUs of P gingivalis were statistically significantly lower at day 21 (P = .0285), day 91 (P = .0266), and day 182 (P = .0327) relative to baseline. The mean pocket depth at the sampled sites, all of which had initial PD > 7 mm, was significantly reduced at days 21, 91, and 182 compared to baseline (P < .001). For T forsythia (Figure 4), the mean CFUs were statistically significantly lower at days 21, 91, and 182 with P values of .0260, .0285, and .0306, respectively. The mean pocket depth was also significantly reduced at all sample periods relative to baseline (P < .001).
Discussion
Periodontitis is induced by bacteria that tend to be site-specific in that the entire dentition does not demonstrate uniform disease activity at any given time. Some sites within the same individual will progress much more aggressively than other sites. In most instances, chronic periodontitis responds well to conventional mechanical therapy consisting of SRP. It is not unusual, however, for a few sites to not respond as well as the clinician would like. Often, practitioners include an adjunctive antibiotic as part of their therapy as a means to better control those sites that did not respond as well to SRP. A number of reviews discuss the benefits of systemic antibiotics as adjuncts to SRP.7-16 Meta-analyses of the effects obtained with the local delivery of metronidazole5 and tetracyclines4 have been conducted using only studies involving participants with chronic periodontitis. Both meta-analyses concluded that beneficial clinical outcomes were achieved with the local delivery of either metronidazole or the tetracyclines when used as adjuncts to SRP. This study lacks a control group. Ideally, a group treated with SRP would have been included as a positive control. This was not performed. The authors believe, however, that the study's longitudinal design with microbial samples and clinical indices collected immediately prior to treatment and at discrete intervals after treatment for 6 months in patients with severely diseased chronic periodontitis does provide useful information to the dental practitioner. In this study, the authors wanted to determine the effect that could be obtained with SRDF alone. The authors did not include in the study design a treatment arm of SRP alone or a treatment arm combining SRP and SRDP. A major reason that the authors did not include an SRP arm is that SRP, in itself, is very effective in controlling chronic periodontitis. An attempt to demonstrate an additive effect of SRDF over SRP alone would require a very large number of patients. Meta-analyses of studies that have compared SRP/local antibiotic treatment to SRP alone have reported local antibiotic delivery in conjunction with SRP did result in improved clinical parameters over SRP alone. Although such differences have been shown to be statistically significant, many investigators claim that the differences seen are clinically insignificant. Thus, instead of using a two-arm study, the authors used a single-arm treatment with the intent to determine the effects that could be contributed directly to the local delivery of 8.5% doxycycline hyclate without the presence of other treatment variables, such as SRP.
In this study, SRDF treatment alone resulted in significant decreases in total recoverable anaerobic flora and in the two periodontal pathogens, P gingivalis and T forsythia. A corresponding decrease was also seen in PDs. Mean PD (sites ≥ 7 mm) was reduced by 2 mm within the first 21 days of treatment and maintained throughout the 6-month course of the study. In two separate two-arm treatment designs involving locally delivered minocycline as an adjunct to SRP versus SRP alone in sites ≥ 5 mm, SRP alone gave a reduction in PD of approximately 1 mm within the first 30 days and the addition of minocycline resulted in approximately another 0.4-mm to 0.5-mm reduction.17,18 In these same two studies, greater reductions were present in the minocycline/SRP treatment than SRP alone in the total number of red complex bacteria, which included Treponema denticola, P gingivalis, and T forsythia. Mean red complex numbers were reduced 50% by minocycline/SRP at 30 days as compared to a 26% reduction by SRP alone. It has not been reported if these reductions obtained at 30 days were maintained for a longer period.
In the SRDF treatment, a 2-mm PD decrease was obtained in sites that originally probed > 7 mm within 21 days and was maintained for as much as 6 months. PD was not examined for changes at sites initially probing < 7 mm. Likewise, individual decreases in P gingivalis and T forsythia of 88% and 99%, respectively, were seen at day 7 and decreases of 80% were present for both red complex species at the study end at 6 months.
A systematic literature review for the added effectiveness of local adjuncts to SRP generally found the tetracyclines combined with SRP resulted in improved PD and clinical attachment levels relative to SRP alone.3 The use of tetracycline as an adjunct generally improved mean PD reduction by approximately 0.5 mm versus SRP alone. In the literature review, minocycline, based on studies involving 104 subjects in each arm, appeared to have more supportive evidence for its use as an adjunct than did other antibiotics or antimicrobials. Participants in two studies with ≥ 7-mm initial PD demonstrated 1.0-mm to 1.1-mm more PD reduction than did SRP alone.19,20
The authors' results with SRDF in patients with similar PD (≥ 7 mm) demonstrated a 2-mm decrease in PD at 21 days that would be equivalent to the combined effect obtained with minocycline and SRP reported above.
One of the interesting findings in this study was that viable counts were recovered at day 7 from all participants despite the presence of SRDF in the sample sites. SRDF has been demonstrated to immediately release doxycycline equivalent to 1600 µg/mL to 1800 µg/mL and to maintain levels in excess of 100 µg/mL for as much as 10 days after placement.21 The authors attributed this apparent resistance to the mature subgingival biofilm, which has been reported to require an antibiotic concentration for inhibition that is 500 to 1000 times that of the same bacterial strains grown planktonically.22
It should be noted that the microbial reductions reported here for SRDF were obtained from deep sites (PD ≥ 7). Deep sites tend to respond greater proportionally than do shallow-to-moderate depth sites. These findings that SRDF is highly effective in such sites is understandable. Sites this deep can be difficult to access without raising a flap. The nature of the polymer-containing doxycycline is such that it is fluid when first inserted into the periodontal pocket. This fluid enables flow and penetration to the base of the pocket regardless of the defect angle. After exposure to the gingival fluid, the polymer formulation changes to a wax-like solid and delivers high concentrations of the antibiotic to the very base of the pocket. Based on this study, it would appear that these concentrations are sufficient to inhibit a significant proportion of the P gingivalis, T forsythia, and total anaerobes present in the subgingival biofilm. It should also be noted that the use of SRDF alone does not remove subgingival calculus.
Conclusion
This study demonstrated SDRP is highly effective in significantly reducing periodontal pathogens of the red complex and the total bacteria mass in deep pockets. This study also showed SDRP has a significant clinical effect in reducing and maintaining a reduction in PD of such pockets for at least 6 months.
References
1. Research, Science and Therapy Committee of the American Academy of Periodontology. Treatment of plaque-induced gingivitis, chronic periodontitis, and other clinical conditions. J Periodontol. 2001;72(12):1790-1800.
2. Goodson JM. Pharmacokinetic principles controlling efficacy of oral therapy. J Dent. Res. 1989;68(spec iss):1625-1632.
3. Bonito AJ, Lux L, Lohr KN. Impact of local adjuncts to scaling and root planing in periodontal disease therapy: a systematic review. J Periodontol. 2005;76(8):1227-1236.
4. Pavia M, Nobile CG, Angelillo IF. Meta-analysis of local tetracycline in treating chronic periodontitis. J Periodontol. 2003;74(6):916-932.
5. Pavia M, Nobile CG, Bianco A, Angelillo IF. Meta-analysis of local metronidazole in the treatment of chronic periodontitis. J Periodontol. 2004;75(6):830-838.
6. Holdeman LV, Moore WE, Cato EP. VPI Anaerobic Lab Manual. 4th ed. Blacksburg, VA: Virginia Polytechnic Institute and State University; 1977.
7. Herrera D, Alonso B, León R, et al. Antimicrobial therapy in periodontitis: the use of systemic antimicrobials against the subgingival biofilm. J Clin Periodontol. 2008;35(8 suppl):45-66.
8. Loesche WJ, Giordano J, Soehren S, et al. Nonsurgical treatment of patients with periodontal disease. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;81(5):533-543.
9. Magnusson I, Clark WB, Low SB, et al. Effect of non-surgical periodontal therapy combined with adjunctive antibiotics in subjects with "refractory" periodontal disease. (I). Clinical results. J Clin Periodontol. 1989;16(10):647-653.
10. Mombelli A. Antimicrobial agents in periodontal prevention, therapy and maintenance: conclusions from the GABA Forum, 6 December 2002, Lyon, France. Oral Dis. 2003;9 (suppl 1):71-72.
11. Pantlin L. Is there a role for antibiotics in periodontal treatment? Dent Update. 2008;35(7):493-496.
12. Preshaw PM. Antibiotics in the treatment of periodontitis. Dent Update. 2004;31(8):448-456.
13. Slots J, Rams TE. Antibiotics in periodontal therapy: advantages and disadvantages. J Clin Periodontol. 1990;17(7 pt 2):479-493.
14. Slots J, Ting M. Systemic antibiotic in the treatment of periodontal disease. Periodontol 2000. 2002;28:106-176.
15. Walker C, Karpinia K. Rationale for the use of antibiotics in periodontics. J Periodontol. 2002;73(10):1188-1196.
16. Walker C, Karpinia K, Baehni P. Chemotherapeutics: antibiotics and other antimicrobials. Periodontol 2000. 2004;36:146-165.
17. Goodson JM, Gunsolley JC, Grossi SG, et al. Minocycline HCl microspheres reduce red-complex bacteria in periodontal disease therapy. J Periodontol. 2007;78(8):1568-1579.
18. Grossi SG, Goodson JM, Gunsolley JC, et al. Mechanical therapy with adjunctive minocycline microspheres reduces red-complex bacteria in smokers. J Periodontol. 2007;78(9):1741-1750.
19. van Steenberghe D, Bercy P, Kohl J, et al. Subgingival minocycline hydrochloride ointment in moderate to severe chronic adult periodontitis: a randomized, double-blind, vehicle-controlled, multicenter study. J Periodontol. 1993;64(7):637-644.
20. van Steenberghe D, Rosling B, Soder P, et al. A 15-month evaluation of the effects of repeated susbgingival minocycline in chronic adult periodontitis. J Periodontol. 1999;70(6):657-667.
21. Stoller NH, Johnson LR, Trapnell S, et al. The pharmacokinetic profile of a biodegradable controlled-release delivery system containing doxycycline compared to systemically delivered doxycycline in gingival crevicular fluid, saliva, and serum. J Periodontol. 1998;69(10):1085-1091.
22. Sedlacek MJ, Walker C. Antibiotic resistance in an in vitro subgingival biofilm model. Oral Microbiol Immunol. 2007;22(5):333-339.
About the Authors
Clay B. Walker, PhD
Oral Biology, University of Florida
Gainesville, Florida
Kenneth C. Godowski, MSc
Tolmar Inc
Fort Collins, Colorado
Steven Garrett, DDS
Tolmar Inc, Fort Collins
Colorado
Craig Wesselman, MS
Tolmar Inc
, Colorado


![Figure 1 Figure 1 Total counts (mean ± standard error [SE]) for anaerobic bacteria <em>P gingivalis</em> and <em>T forsythia</em> recovered at each sampling period.](/media/thumbnail/1830/150x150)