Guided Implant Surgery with Placement of a Presurgical CAD/CAM Patient-Specific Abutment and Provisional in the Esthetic Zone
Abstract: Parallel use of implant treatment planning software and cone-beam computed tomography (CBCT) can, using certain criteria, consolidate steps and streamline tooth replacement strategies. The authors describe such a case in the esthetic zone whereby flapless extraction and immediate implant placement using CT-guided surgery were performed simultaneously, with placement of a computer-aided design/computer-aided manufactured (CAD/CAM) patient-specific abutment and non-occlusal function provisional in a single visit (supporting the “one-abutment, one-time” concept). An over-retained primary cuspid in a periodontally healthy woman with well-controlled type-2 diabetes was replaced with an implant and CAD/CAM patient-specific abutment in the No. 11 position. A necessary implant-axis angle correction was customized using digital information from a CBCT scan and implant treatment planning software, without the need for site development or a conventional impression. This data integration and streamlined workflow enabled fabrication of a CAD/CAM patient-specific abutment before surgical treatment. The abutment remained in place from implant surgery to the prosthetic phase, with minimal soft-tissue changes, enabling preservation of pink esthetics and expediting treatment. The result was a preserved emergence profile in the presence of high esthetic demands. However, due to slight post-extraction soft-tissue changes, digital reformatting of the abutment was required when the final crown was fabricated, thus limiting the disruption of the biologic width to a one-time occurrence. The importance of case selection for this treatment protocol in the esthetic zone cannot be overemphasized. A thick crestal dentoalveolar bone phenotype (> 1 mm, approaching 2 mm in this case), broad zone of attached and keratinized gingiva (3 mm to 4 mm in this case), adequate peri-implant soft-tissue thickness (> 1 mm in this case), and high primary implant stability (ISQ = 80 in this case) were all critical factors influencing outcome.
Implant treatment planning software has been used successfully for more than two decades.1-4 Such virtual planning modalities offer implant clinicians the ability to map the proposed implant sites relative to the existing bony regional anatomy, and to produce a stereolithographic surgical guide that provides a relatively precise path for optimal osteotomy site preparation in three planes of space.
Assessments of bone quality and quantity are especially critical and often require site development modalities that involve both hard- and soft-tissue regenerative therapy in order to meet acceptable pink and white outcome esthetic scores. This involves a protracted treatment course and increased expense in general. Interestingly, data from prospective clinical studies suggest that more favorable hard- and soft-tissue esthetics can be achieved in maxillary single-tooth replacement cases that do not require pre-implant surgery.5,6
Even if regenerative therapy is not indicated, the typical tooth replacement process requires multiple appointments, including not only diagnostic modeling, but also possible fabrication of a scanning appliance, cone-beam computed tomography (CBCT) scanning, fabrication of a surgical guide, implant placement, and provisionalization, leading to custom abutment fabrication (usually from an implant-level impression), and, ultimately, delivery of the permanent abutment and crown to the implant fixture.
Risk assessment based upon preexisting dentoalveolar bone architecture has received considerable attention in the literature. In a 2011 CBCT study involving nearly 500 patients, Braut et al concluded that 90% of humans have less than 1 mm of facial bone width at the crest and mid-root position of the maxillary anterior natural teeth.7 Nowzari et al (2012) reported similar vulnerability of natural teeth due to suboptimal thickness of supporting facial bone.8 In 2013, Mandelaris et al proposed a new classification system for evaluating dentoalveolar bone phenotype and associated risk via CBCT, to facilitate decision making in interdisciplinary dentofacial therapy (including immediate implant treatment planning applications).9
More importantly, repeated insult from removal and replacement of a prosthesis and abutment can negatively impact the cultivation and preservation of crestal bone maintenance and predispose the patient to crestal bone loss, possibly resulting in structural and esthetic outcome compromises. In 2001, Goldberg et al published a review that emphasizes preservation of biologic width, the importance of an adequate band of attached gingiva around the implant to minimize the possibility of recession, and the placement of a restorative margin no more than 0.5 mm into the healthy sulcus.10 Kan et al also observed that facial sites in the esthetic zone with a thick gingival biotype exhibited less recession over the course of a 2- to 8-year follow-up study of immediate implant placement and provisionalization.11 Finally, in a study examining 63 functional implants, Zigdon and Machtei demonstrated that implants with a wider band of keratinized mucosa (> 1 mm) tended to have less recession. Similarly, implants with thicker mucosa (≥ 1 mm) showed the same trend.12 Table 13,9,11-20 summarizes proposed case-selection criteria that may be useful for immediate tooth replacement as described below.
In cases where conditions of crestal dento-alveolar bone thickness, marrow quality, and peri-implant soft-tissue position and thickness are close to ideal, combining technologies from various sources can enable pre-treatment digital fabrication of a final patient-specific custom abutment by transforming traditional clinical steps into a virtual design scheme with a computer-aided design/computer-aided manufactured (CAD/CAM) workflow.
One-Abutment, One-Time
Recent software advances offer the ability to combine CBCT and optically scanned data from high-quality diagnostic casts to perform CT-guided implant surgery and deliver a patient-specific CAD/CAM-milled abutment and non-occlusal function provisional at the same appointment, with ideal contours to maintain architecture. This abutment can remain in place and become the final abutment (whereby it is never removed, thus the “one-abutment, one-time” designation). Alternatively, it can be replaced with a new, digitally altered patient-specific abutment, thereby disrupting the implant–abutment interface only once, because the dataset used to create the initial abutment can be referenced, modified, and redeveloped all within a digital environment without having to obtain a conventional impression.
Case Report
The authors describe here how the use of implant-planning software with an existing CBCT dataset allowed fabrication of a CAD/CAM, patient-specific abutment that could be secured to the implant fixture on the day of surgery, with the possibility of never removing it if the circumstances allow.
Patient History
A 50-year-old Caucasian woman presented with a chief complaint of requesting implant replacement of a severely resorbed and retained deciduous cuspid (No. H). A review of her medical history revealed well-controlled type-2 diabetes, hypertension, elevated cholesterol, hypothyroidism, anxiety, restless leg syndrome, mild osteoarthritis, gastroesophageal reflux disorder, irritable bowel syndrome, systemic lupus erythematosus, and a low level of vitamin D on a recent blood chemistry panel. Her dental history included good oral hygiene and compliance with semiannual prophylaxis/exam visits.
Treatment Plan
After collaborative workup between the surgical and restorative doctors, the patient decided to proceed with placement of an immediate implant and prefabricated, patient-specific abutment. A CBCT scan was obtained as part of the initial workup. The Digital Imaging Communication in Medicine (DICOMSM) data from this scan were then imported into the SIMPLANT® software (DENTS-PLY Implants, www.dentsplyimplants.com) for virtual surgical planning. This plan incorporated an abutment core file, which is a virtual representation used to produce both a custom ATLANTIS™ abutment (DENTSPLY Implants) and the SIMPLANT Guide.
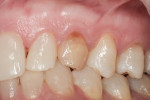
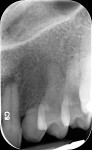
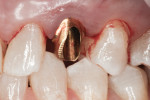
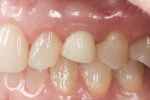
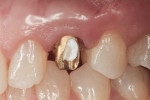
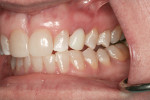
Figure 1 and Figure 2 show the initial clinical presentation and periapical radiograph, respectively, of the proposed implant site. The clinical presentation was categorized as a case-type pattern (Pattern 1) implant candidate revealing normal dental and surgical anatomy.21
Accordingly, to ensure accurate representation of the soft-tissue emergence profile inherent in the tissue around the existing tooth No. H, an irreversible hydrocolloid impression (Ivoclar Accu-Dent® System 2™, Ivoclar Vivadent Inc., www.ivoclarvivadent.com) was made of the maxillary arch, and a diagnostic cast was created. The stone cast was optically scanned and registered to the CBCT hard-tissue data in the SIMPLANT planning software and its accompanying CAD/CAM workflow process.
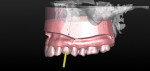
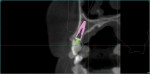
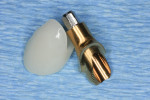
Figure 3 shows the virtual representation of this optical scan, with a virtual replacement tooth generated from the SIMPLANT software. A known angle correction was virtually incorporated into the planned implant path. Figure 4 shows the cross-sectional view of axial inclination and the required existing axis of the planned final tooth position. Figure 5 through Figure 7 show the various views of the patient-specific abutment that was virtually created. The milled patient-specific gold-shaded titanium abutment and polymethylmethacrylate (PMMA) provisional created by the laboratory through the digital workflow process (using the virtual design data) are shown separately (Figure 8) and seated (Figure 9).
Surgical Procedure
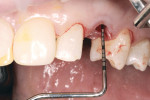
Under intravenous conscious sedation and local anesthesia, a sulcular incision was made with a microsurgical straight blade to remove the supracrestal fibers (Figure 10), followed by uneventful atraumatic extraction of tooth No. H.
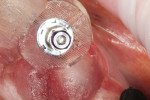
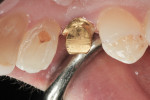
After extraction, complete seating of the surgical guide (Tapered Navigator® SIMPLANT SAFE Guide®, DENTSPLY Implants) was verified (Figure 11). The osteotomy site was then completed through the Tapered Navigator SIMPLANT SAFE Guide CT-guided System using a flapless approach. A 4/3 Biomet 3i T3 Tapered Prevail® implant (Biomet 3i, www.biomet3i.com) was delivered through the Tapered Navigator SIMPLANT SAFE Guide using the appropriate implant mount (Figure 12) for totally guided implant surgery.
The Tapered Navigator SIMPLANT SAFE Guide system has rotational timing, which allows for the implant hex orientation to be positioned at the time of surgery, as it was oriented during the pre-surgical fabrication of the abutment (Figure 12).
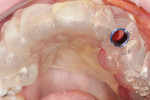
The vertical positioning was confirmed and verified with a UNC-15 mm periodontal probe (Figure 13). An objective analysis was used to substantiate initial implant stability. Figure 14 shows an Osstell® ISQ Smartpeg (Osstell AB, www.osstell.com), which uses resonance frequency to assess implant stability, represented by the implant stability quotient (ISQ). The implant trajectory is also clearly observed by the engagement of the Smartpeg to the implant (Figure 14). The observed ISQ value of 80 for this implant indicates high stability (Figure 15).
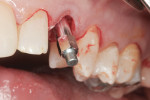
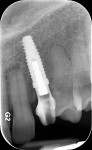
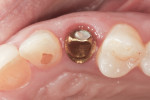
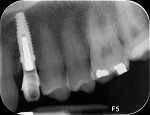
A facial view of the seated patient-specific ATLANTIS abutment, with its appropriately positioned screw-access hole, is pictured in Figure 16, showing maintenance of soft-tissue position. Figure 17 shows the postsurgical radiographic view of the initial seating.
Postoperative Treatment
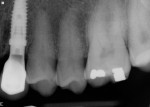
Good initial and mid-phase soft-tissue healing was evident 1 week postoperatively (Figure 18), with excellent preservation of the gingival margin. At 5 weeks, limited gingival inflammation was present due to the patient’s lack of optimal plaque control (Figure 19). At 14 weeks, the peri-implant soft tissue health was improved, and osseointegration was confirmed clinically and radiographically (Figure 20).
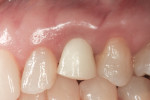
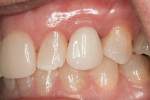
Soft-tissue architecture was well-maintained 14 weeks postoperatively. With the non-occlusal function provisional removed, good peri-implant soft-tissue tone was evident from facial (Figure 21) and palatal aspects (Figure 22). Importantly, the incisal view (Figure 23) showed healthy sulcular epithelium, indicating good biologic-width maturation, which was confirmed radiographically in the osseous topography. The 14-week radiographic view of the implant and abutment (Figure 24) showed good maintenance of crestal bone, with an established biologic-width formation, suggesting complete osseointegation.
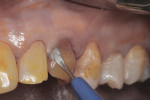
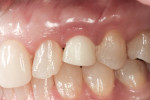
Figure 25 shows slight exposure of the abutment-provisional margin that had occurred on the mesial aspect. A final abutment was designed between the restorative office and the laboratory using a digital workflow to adjust the margin virtually with no disruption of the implant–abutment junction nor to the newly established biologic width. Figure 26 and Figure 27 demonstrate the initial (green) and revised (blue) virtual abutment and associated margin designs. The final ATLANTIS abutment and crown (IPS e.max® Ceram, Ivoclar Vivadent; ArgenZ™ milled zirconia, Argen Corp., www.argen.com) were delivered thereafter (Figure 28 and Figure 29).
Discussion
Braut et al reported in a sample of 498 maxillary anterior cases that the facial bone was ≤ 1 mm thick or missing entirely in 90% of the teeth, as evaluated by CBCT.7 Such data suggest a level of risk that could well preclude the treatment course followed here, and place critical emphasis on preoperative CBCT assessment for appropriate case selection.
The ability to minimize hard- and soft-tissue changes is of paramount importance when performing immediate implant placement in the esthetic zone. Histomorphometric studies in monkeys22 and dogs23 have demonstrated the loss of bundle bone (and its replacement by woven bone) from socket walls undergoing remodeling after extraction. The exclusive association of bundle bone with the periodontal ligament (ie, a dentate anatomic environment) has also been demonstrated.24 Unfortunately, because bundle bone is specific to the natural tooth, some degree of dimensional alteration following tooth loss is inevitable, regardless of the intervention course.
In this case report, the authors present a novel patient-specific CAD/CAM-generated abutment and PMMA provisional made pre-surgically using a fully digital workflow to simplify, expedite, and enhance immediate provisionalization of a single-tooth dental implant, facilitating collaboration among surgical and restorative offices and the dental laboratory.
The case presented offers a simplified and predictable workflow as well as reduced surgical and prosthetic chairtime, and potentially minimizes disruption of the implant–abutment interface (which could compromise biologic width) and, ultimately, preserves crestal bone (ie, “one-abutment, one-time” concept).
The challenge arises in delivering a CAD/CAM abutment at the time of surgery and having that margin be esthetically acceptable as well as periodontally sound for the final prosthesis. This challenge is compounded by the loss of a natural tooth, its associated bundle bone, and the dimensional alterations that will inevitably follow.
In the case described, even with optimal dento-alveolar bone thickness at the crest, keratinized and attached gingival width, and peri-implant soft-tissue thickness, tissue alteration occurred where the mesial margin was visible and required correction by redesigning another abutment to negate a visible margin.
Axial Angulation Correction
Figure 3 and Figure 4 demonstrate the need for a required angle correction. Relying on the SIMPLANT software, expertise in the planning, and expertise from the laboratory technologist in the deliverables, the axial inclination was corrected by approximately 25 degrees in the palatal direction. The SIMPLANT software can incorporate virtual abutments into the design; this capability includes ATLANTIS patient-specific abutments such as was used here. Without this virtual-planning capability, an alternative would be lateral ridge augmentation to physically alter the axial inclination of the implant to accommodate a screw-retained access or utilize an angle correction in a customized final abutment. In the latter scenario, the immediate provisional would require facial access and thus jeopardize esthetics during the interim provisionalization phase.
Micro/Macrogapping Risk
The potential for variations in micro- or macrogapping at the implant–abutment interface is of concern when fabricating abutments that are “reverse-engineered” for implant fixtures. Such adaptation becomes potentially significant when implant components that originate with different manufacturers are combined, as was done in this case.
Interchangeability of implants and abutments from different manufacturers is the subject of increasing clinical focus in regard to gapping at the implant–abutment interface (IAI). A recent study by Zanardi et al using scanning electron microscopy observed increased microgapping when abutments were randomly combined with implant fixtures from different manufacturers (all different from those used in this case), compared with manufacturer-matched components (0.6 µm to 16.9 µm and 0.3 µm to 12.9 µm, respectively).25
The ATLANTIS individualized abutment is designed to be compatible with most major implant systems, including Biomet 3i, which was used here. Similarly, the SIMPLANT software contains internally all of the necessary geometry to effectively index the ATLANTIS abutment, in this case, for use with a Biomet 3i implant. Intraoral scanning may offer future workflow advantages and is an emerging technology that is positioned to interface well with the workflow described here.
Implant Diameter and Platform-Switching
Preservation of crestal bone is critical when the esthetic demand is high, as in this case. Because the diameter of the gold-shaded titanium ATLANTIS abutment used at the implant–abutment interface is compatible with a 4-mm implant that has a 3-mm seating surface (platform-switched), the Biomet 3i T3 Tapered Prevail implant was the optimal choice.
However, if a zirconia ATLANTIS abutment had been contemplated, a wider implant diameter seating surface would have been needed in this case. Given the manufacturing specifications of a patient-specific CAD/CAM zirconia abutment, a 4-mm seating surface (or greater) would have been necessary. A non–platform-switched 4-mm implant would have been counterproductive to maintaining crestal bone levels, as platform switching is known to medialize the biologic width and optimize crestal bone maintenance, compared to non–platform-switched components. In this case, expanding to a 5-mm platform was not an option, given the presenting regional anatomy.
Trammell et al measured both clinical variables and found significantly less bone resorption with platform-switched components, compared with conventional component pairing within the same prosthesis/patient (P < 0.0001).26 Systematic reviews by Al-Nsour et al and Atieh et al concluded that platform switching may preserve bone27,28 and soft-tissue levels.28 A recent study by Canullo et al reported that biologic width reestablishment may have greater impact on bone levels than does platform diameter.29
Furthermore, 3-year follow-up data from an earlier multicenter randomized study by Canullo et al reported statistically significant reductions in bone resorption for platform-switched definitive abutments that were attached to 5-mm implants, compared to provisional abutments that were attached and removed multiple times. The authors suggest that the “one-abutment, one-time” concept (as in this case) may thus further reduce crestal bone resorption.30 From the standpoints of implant survival and soft-tissue health over a 33-month follow-up period, Hartlev et al also reported predictable success using this concept in the esthetic zone.31
A recent split-mouth study by Baffone et al in dogs raises interesting concerns regarding this principle. Experimental defects were prepared in the buccal alveolar walls of one side, while the other side underwent normal healing following bilateral extraction of mandibular premolars and molars and prior to placement of implants with either narrow or wide abutments. In the sites with buccal defects (test sites), the peri-implant soft-tissue adaptation was found to be more coronal on the narrow (ie, less soft-tissue recession) than on the wide abutments. However, in the unaltered ridges (control sites), the wide abutments were associated with less recession.32 While potentially non-generalizable differences exist between this study design and the present case, these results do seem to support the existence of a more favorable attachment scenario when narrower abutments are used, as in this case. While a narrower abutment was used in the present case, careful attention was also taken in the planning of the abutment to minimize overcontouring of the emergence profile and to encourage maintenance of the medialized and coronal biologic width position and resulting crestal bone.
In cases where a modified implant angulation requires a cemented restoration (as in this case), a more expedited approach is favorable, because digital modifications can be made to the laboratory-based data set. Using the CAD/CAM workflow processes, laboratory personnel can implement—virtually—any necessary modifications to the individualized contours of the abutment. This requires that the implant–abutment interface be physically disrupted only once. It also may reduce the costs associated with multiple implant parts, as well as reduce chairtime for the restorative dentist or prosthodontist.
The following caveat remains: In cases where the emergence profile must be developed de novo and multiple modifications will likely be needed, a screw-retained, highly polished provisional should be considered to groom soft tissues leading to an implant-level impression, as opposed to the approach described here. That course offers the greatest certainty of dimensional stability after a 6-month period has elapsed, helping to minimize potential changes in tissue form, with the ultimate objective of sustainable esthetics. In the present case, preservation of a preexisting ideal contour established by a natural tooth was desired. Overall, this is a prosthetic challenge that may or may not have measurable consequences. Of course, a connective tissue graft could also have helped mask post-extraction facial free gingival margin soft-tissue changes, if they occurred and/or thicker soft-tissue dimensions are needed.
The pink esthetic score (PES) assessed by Furhauser et al was found to yield reproducible results as measured by clinicians of various specialties, and thus was deemed useful in objectively assessing outcomes of single-tooth replacement.13 Cutrim et al (2012) also confirmed the utility of the PES in evaluating the health of peri-implant tissues around both screw- and cement-retained single-tooth implants in the anterior maxilla.14 The PES has also demonstrated strong correlations with assessments of both patients33 and clinicians,15,16 as well as reproducible evaluation and intra-examiner agreement.16 The pink esthetic score in the present case was extremely high (13) and, in part, validated the process in proof of principle for the authors.
In preserving gingival architecture and ridge contour, the main difficulty arises from removal of bundle bone associated with supracrestal fibers that must be transected even under the most atraumatic of extraction circumstances. As observed by the authors in this case, some changes in soft-tissue height can occur despite practitioners’ best efforts. Predicting the degree of these changes proved to be the most challenging task.
In dogs, Araújo and Lindhe demonstrated a two-phase loss of alveolar bone after molar hemisection and root removal, which resulted in crestal reduction (due to loss of bundle bone) as well as resorption at the buccal and lingual walls of the extraction site.34 While these results were obtained in an animal model and without implant placement, they do provide a note of caution regarding how post-extraction bone remodeling can impact crestal bone height (and potentially esthetics due to recession), and underscore the importance of having a preexisting thick crestal bone phenotype.
More recently, a randomized clinical trial by Araújo et al evaluated the effects of placing a xenograft material in fresh extraction sockets (incisors, canines, and premolars scheduled for removal). They observed a marked reduction in hard-tissue cross-sectional area in patients in the group that received the xenograft (3% versus 25% for the sockets without xenograft).35 These results suggest that the greater the internal biomechanical support for a fresh extraction site, the more optimal the preservation of alveolar bone; this supports the ridge-preservation efficacy of immediate implant placement—especially into an optimal bone phenotype—whenever possible. In the present case, there was no implant-post extraction alveolus “gap” requiring management. However, the importance of the concept cannot overemphasized.
On the other hand, esthetic parameters have been observed to improve over time after placement of the final restoration. A study by Paul and Held evaluated 31 immediate single-tooth replacements in the esthetic zone with immediate provisionalization that were very similar to this case. Esthetic results, as measured by pink and white esthetic scores, actually improved over time (1.5 to 5 years), and little if any gingival recession was noted.36 Luo et al noted a similar improvement at 3 months after cementation of the final crown.33
Conclusion
In conclusion, this case demonstrates that under certain conditions, a novel tooth-replacement treatment plan can be conceived and executed with successful virtual/digital replacement of steps that traditionally require significant time and effort for both patient and clinician. Here, an initial abutment was digitally designed, produced, and placed without the need for conventional impressioning. Even though a second, modified abutment was required to compensate for a minor soft-tissue change, the final abutment was produced digitally and placed atraumatically, safeguarding an established biologic width and meeting the critical esthetic demands of the case. It is plausible that had these post-extraction soft-tissue alterations been better predicted from the beginning, the need for a second CAD/CAM, patient-specific abutment to compensate would not have been necessary. It is worth noting that because of the workflow efficacy here, the expense of a final ATLANTIS abutment was minimal. The criteria of dento-alveolar bone thickness, peri-implant soft-tissue quality and thickness, good initial implant stability, and high patient compliance should all coexist if such a path is contemplated.
DISCLOSURE
The authors have no financial interest in any of the companies mentioned in this article.
ACKNOWLEDGMENTS
The authors thank the patient for her participation in the study and her collaborative efforts to maximize the treatment outcome; DENTSPLY Implants for support of the study; Mr. Chuck Genco, General Manager, Van Hook Dental Studio, Tempe, AZ, for management of the CAD/CAM workflow and digital data set for customized fabrication of the abutment and provisional used in this case; and Scott A. Saunders, DDS, ELS, CMPP, at Dental and Medical Writing and Editing, LLC (DMWE), Royersford, PA, for professional dental and medical writing and editing services in preparation of the manuscript.
ABOUT THE AUTHORS
George A. Mandelaris, DDS, MS
Assistant Clinical Professor, Department of Oral and Maxillofacial Surgery, Louisiana State University, School of Dentistry, New Orleans, Louisiana; Adjunct Clinical Assistant Professor, Department of Periodontics; University of Illinois, College of Dentistry, Chicago, Illinois; Private Practice, Periodontics and Dental Implant Surgery, Park Ridge and Oakbrook Terrace, Illinois
Scott D. Vlk, DDS
Private Practice, Restorative Dentistry, Yorkville, Illinois
REFERENCES
1. Mandelaris GA, Rosenfeld AL, King SD, Nevins ML. Computer-guided implant dentistry for precise implant placement: combining specialized stereolithographically generated drilling guides and surgical implant instrumentation. Int J Periodontics Restorative Dent. 2010;30(3):275-281.
2. Mandelaris GA, Rosenfeld AL. The expanding influence of computed tomography and the application of computer-guided implantology. Pract Proced Aesthet Dent. 2008;20(5):297-305.
3. Rosenfeld AL, Mandelaris GA, Tardieu PB. Prosthetically directed implant placement using computer software to ensure precise placement and predictable prosthetic outcomes. Part 1: diagnostics, imaging, and collaborative accountability. Int J Periodontics Restorative Dent. 2006;26(3):215-221.
4. Rosenfeld AL, Mandelaris GA, Tardieu PB. Prosthetically directed implant placement using computer software to ensure precise placement and predictable prosthetic outcomes. Part 3: stereolithographic drilling guides that do not require bone exposure and the immediate delivery of teeth. Int J Periodontics Restorative Dent. 2006;26(5):493-499.
5. den Hartog L, Raghoebar GM, Slater JJ, et al. Single-tooth implants with different neck designs: a randomized clinical trial evaluating the aesthetic outcome. Clin Implant Dent Relat Res. 2013;15(3):311-321.
6. Cosyn J, Eghbali A, Hanselaer L, et al. Four modalities of single implant treatment in the anterior maxilla: a clinical, radiographic, and aesthetic evaluation. Clin Implant Dent Relat Res. 2013;15(4):517-530.
7. Braut V, Bornstein MM, Belser U, Buser D. Thickness of the anterior maxillary facial bone wall-a retrospective radiographic study using cone beam computed tomography. Int J Periodontics Restorative Dent. 2011;31(2):125-131.
8. Nowzari H, Molayem S, Chiu CH, et al. Cone beam computed tomographic measurement of maxillary central incisors to determine prevalence of facial alveolar bone width >/=2 mm. Clinical Implant Dent Relat Res. 2012;14(4):595-602.
9. Mandelaris GA, Vence BS, Rosenfeld AL, Forbes DP. A classification system for crestal and radicular dentoalveolar bone phenotypes. Int J Periodontics Restorative Dent. 2013;33(3):289-296.
10. Goldberg PV, Higginbottom FL, Wilson TG. Periodontal considerations in restorative and implant therapy. Periodontol 2000. 2001;25:100-109.
11. Kan JY, Rungcharassaeng K, Lozada JL, Zimmerman G. Facial gingival tissue stability following immediate placement and provisionalization of maxillary anterior single implants: a 2- to 8-year follow-up. Int J Oral Maxillofac Implants. 2011;26(1):179-187.
12. Zigdon H, Machtei EE. The dimensions of keratinized mucosa around implants affect clinical and immunological parameters. Clin Oral Implants Res. 2008;19(4):387-392.
13. Fürhauser R, Florescu D, Benesch T, et al. Evaluation of soft tissue around single-tooth implant crowns: the pink esthetic score. Clin Oral Implants Res. 2005;16(6):639-644.
14. Cutrim ES, Peruzzo DC, Benatti B. Evaluation of soft tissues around single tooth implants in the anterior maxilla restored with cemented and screw-retained crowns. J Oral Implantol. 2012;38(6):700-705.
15. Hartlev J, Kohberg P, Ahlmann S, et al. Patient satisfaction and esthetic outcome after immediate placement and provisionalization of single-tooth implants involving a definitive individual abutment [published online ahead of print September 12, 2013]. Clin Oral Implants Res. 2013. doi: 10.1111/clr.12260.
16. Gehrke P, Lobert M, Dhom G. Reproducibility of the pink esthetic score—rating soft tissue esthetics around single-implant restorations with regard to dental observer specialization. J Esthet Restor Dent. 2008;20(6):375-384.
17. Marquez IC. The role of keratinized tissue and attached gingiva in maintaining periodontal/peri-implant health. Gen Dent. 2004;52(1):74-78.
18. Karthik K, Sivakumar, Sivaraj, Thangaswamy V. Evaluation of implant success: a review of past and present concepts. J Pharm Bioallied Sci. 2013;5(suppl 1):S117-S119.
19. Benic GI, Mir-Mari J, Hämmerle CH. Loading protocols for single-implant crowns: a systematic review and meta-analysis. Int J Oral Maxillofac Implants. 2014;29(suppl):222-238.
20. Gottesman E. Periodontal-restorative collaboration: the basis for interdisciplinary success in partially edentulous patients. Compend Contin Educ Dent. 2012;33(7):478-510.
21. Mecall RA. Computer-guided implant treatment pathway. In: Tardieu P, Rosenfeld A, eds. The Art of Computer-Guided Implantology. Chicago, IL: Quintessence Publishing Co.; 2009.
22. Scala A, Lang NP, Schweikert MT, et al. Sequential healing of open extraction sockets. An experimental study in monkeys. Clin Oral Implants Res. 2014;25(3):288-295.
23. Cioban C, Zăgănescu R, Roman A, et al. Early healing after ridge preservation with a new collagen matrix in dog extraction sockets: preliminary observations. Rom J Morphol Embryol. 2013;54(1):125-130.
24. Ho SP, Kurylo MP, Grandfield K, et al. The plastic nature of the human bone-periodontal ligament-tooth fibrous joint. Bone. 2013;57(2):455-467.
25. Zanardi PR, Costa B, Stegun RC, et al. Connecting accuracy of interchanged prosthetic abutments to different dental implants using scanning electron microscopy. Braz Dent J. 2012;23(5):502-507.
26. Trammell K, Geurs NC, O’Neal SJ, et al. A prospective, randomized, controlled comparison of platform-switched and matched-abutment implants in short-span partial denture situations. Int J Periodontics Restorative Dent. 2009;29(6):599-605.
27. Al-Nsour MM, Chan HL, Wang HL. Effect of the platform-switching technique on preservation of peri-implant marginal bone: a systematic review. Int J Oral Maxillofac Implants. 2012;27(1):138-145.
28. Atieh MA, Ibrahim HM, Atieh AH. Platform switching for marginal bone preservation around dental implants: a systematic review and meta-analysis. J Periodontol. 2010;81(10):1350-1366.
29. Canullo L, Iannello G, Peñarocha M, Garcia B. Impact of implant diameter on bone level changes around platform switched implants: preliminary results of 18 months follow-up a prospective randomized match-paired controlled trial. Clin Oral Implants Res. 2012;23(10):1142-1146.
30. Canullo L, Bignozzi I, Cocchetto R, et al. Immediate positioning of a definitive abutment versus repeated abutment replacements in post-extractive implants: 3-year follow-up of a randomised multicentre clinical trial. Eur J Oral Implantol. 2010;3(4):285-296.
31. Hartlev J, Kohberg P, Ahlmann S, et al. Immediate placement and provisionalization of single-tooth implants involving a definitive individual abutment: a clinical and radiographic retrospective study. Clin Oral Implants Res. 2013;24(6):652-658.
32. Baffone G, Lang NP, Pantani F, et al. Hard and soft tissue changes around implants installed in regular-sized and reduced alveolar bony ridges. An experimental study in dogs [published online ahead of print December 2, 2013]. Clin Oral Implants Res. 2013. doi: 10.1111/clr.12306.
33. Luo Z, Zeng R, Chen Z. Single implants in the esthetic zone: analysis of recent peri-implant soft tissue alterations and patient satisfaction. A photographic study. Int J Oral Maxillofac Implants. 2011;26(3):578-586.
34. Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
35. Araújo MG, da Silva JC, de Mendonça AF, Lindhe J. Ridge alterations following grafting of fresh extraction sockets in man. A randomized clinical trial [published online ahead of print March 12, 2014]. Clin Oral Implants Res. 2014. doi: 10.1111/clr.12366.
36. Paul S, Held U. Immediate supracrestal implant placement with immediate temporization in the anterior dentition: a retrospective study of 31 implants in 26 patients with up to 5.5-years follow-up. Clin Oral Implants Res. 2013;24(6):710-717.