Periodontal Management and Restoration of an Amelogenesis Imperfecta Patient: A Case Report
Robert A. Horowitz, DDS; D.K. Gautam, BDS, MDS; Suneet Karol, BDS, MDS; and Bindiya Kumari, BDS, MDS
Abstract
This report describes the treatment of a young male patient diagnosed with amelogenesis imperfecta (AI), a hereditary disorder that affects the enamel of both primary and permanent dentition. For management and rehabilitation, it is crucial to determine the type of AI—hypoplastic, hypomaturation, or hypocalcified. As with this patient, who presented with tricho-dento osseous syndrome, patients may present with associated expression of a syndrome (partial or full) and secondary changes in the periodontium. AI is a serious problem; therefore extensive treatment using a multidisciplinary approach must be instituted, especially if the patient is syndrome-associated.
Amelogenesis imperfecta (AI) is a hereditary disorder that affects the enamel of both primary and permanent dentition.1 Developmental disturbances of the dentition encompass a broad range of disorders and affect its shape, size, structure, growth and functions.2 Pediatric patients with any of these developmental disturbances present with challenging esthetic, functional, and psychological needs.3
AI may be associated with other dental and skeletal defects such as root resorption, attrition, delayed eruption, impaction, pulp stones, dens in dente, and anterior open bite. The condition exhibits hyperemic and edematous gingiva. To date, 40 papers regarding the condition have been published; among them, 28 patients were hyperemic. This condition contributes to mouth breathing and reluctance of the patient to brush because of sensitive teeth. Along with poor oral hygiene, factors such as patient noncompliance affect the prognosis of prosthetic treatment.4 Esthetic crowns are sometimes used to compensate for soft enamel after thorough oral prophylaxis. Composite restorations may be provided for patients with minimal occlusal defects and minimal tooth destruction.2
Case Report
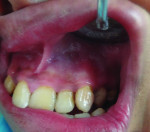
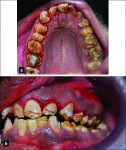
A 15-year-old male with chipped, yellow-brown, snow-capped teeth and generalized desquamation of the gingiva visited the outpatient Department of Periodontology HP Government Dental College (HPGDC) hospital, Shimla, India. The patient had difficulty chewing due to hypersensitivity and a burning sensation in his mouth. The case is depicted in Figure 1 through Figure 9.
Diagnosis
Based on history, clinical examination, and radiographic examination—as described below—the patient was diagnosed with AI hypomaturation type (IID), autosomal dominant.
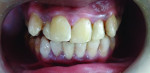
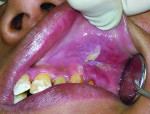
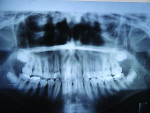
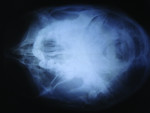
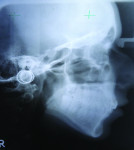
The patient exhibited the typical characteristics of AI both clinically and radiographically. The clinical examination showed that both primary and permanent dentition were affected, with snow-capped anterior teeth (Figure 4), yellowish brown posterior teeth (Figure 7), and an anterior open bite (Figure 4). The gingiva of the patient described was hyperemic and edeomatous, and the labial gingiva of the maxillary arch was desquamating, leaving behind denuded surfaces (Figure 3). Radiographic findings included: a lateral cephalometric view showing an obtuse mandibular angle (Figure 8 and Figure 9); a panorex showing enamel and dentin with similar densities (Figure 5); and a submentovertex view of the base of skull showing no osteosclerosis (Figure 6). The patient history revealed that while parents lacked the characteristic, one of the patient’s two teenage sisters presented with similar manifestations.
Also considered—and ruled out—in making the determination were differential diagnosis conditions including dental fluorosis, molar incisor hypomineralization (MIH), chronological enamel hypoplasia, and extrinsive discoloration of the enamel.5
Periodontal Treatment
Despite the discomfort—especially the tooth sensitivity—that led to the patient’s poor hygiene, the existing bone loss and desquamating gingivitis necessitated extensive oral prophylaxis.4
The oral health of the patient was maintained after conventional periodontal therapy. Antisensitivity tooth paste (stannous fluoride 0.4%) was applied all over the dentition prior to the scaling procedure, and careful hand scaling was performed. Frequent chlorhexidine (0.2%) oral rinses and metronidazole (25%) gel application was completed in the deeper pockets. The patient was kept on antisensitivity tooth paste (stannous fluoride 0.4%) to be used with an ultra-soft tooth brush twice daily after meals and an antisensitivity mouthwash (potassium nitrate) to be used twice daily 30 minutes after brushing. The patient was also prescribed ibuprofen 400 mg tid for 1 week and vitamin D and vitamin A for 1 month. In response to the periodontal therapy, signs of desquamative gingivitis abated and the tissue normalized, but there was only partial relief of the tooth hypersensitivity.
Prosthetic Treatment
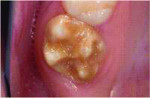
All carious teeth (molars, Class I cavities) were restored with glass-ionomer cement (GIC) restorative material, and full composite veneers were placed to block the exposed dentinal tubules. This treatment adequately addressed the functional and esthetic aspects of the patient’s problems.
Discussion
AI is a group of clinically and genetically heterogeneous disorders caused by malformed protein synthesis in the enamel; ameloblastin, enamelin, tuftelin, and amelogenin occur due to mutations in the AMELX, ENAM, MMP-20 and KLK-4 genes.2,3,6,7 The cause of mutation can be adenoviruses.8 The incidence of AI varies widely from 1 in 700 people in northern Sweden to 1 in 1,400 people in the United States.1-4 Some syndromes are mild, while others are devastating—characteristics include osteosclerosis of the base of skull, kinky hair, and brittle nails.1 The diversity of enamel malformations observed is believed to reflect differences in the timing and duration of the occurance of the disruption during the process of enamel formation. Only teeth actively forming enamel at the time of illness are affected.9 AI may be associated with other dental and skeletal defects like root resorption, attrition, delayed eruption, impaction, pulp stones, dens in dente, anterior open bite. Among 15 cases of delayed eruption, nine cases of pulp stones have been reported. According to Seow pulp stones are found as a result of external local irritation due to thin enamel.1,2
Witkop (1988)10 classified AI, which relies on phenotype and pedigree.1 AI classifications are shown in Table 1.
The formation of enamel is a multistep process, and problems may arise during any of the steps: elaboration of enamel; matrix mineralization; and maturation.1 As a result of problems at any point, the enamel may be of poor quality, which contributes to enamel chipping, flaking and reduced occluso-vertical dimensions. The underlying dentin may be exposed, making the patient hypersensitive to hot and cold. The alveolar bone may also be hypoplastic, and the alveolus may not completely develop.1,6
Treatment Guidelines
To maximize treatment success, the authors recommend the following guidelines:
• Careful patient management makes it essential to classify patients according to AI type based on history, clinical examination, radiographic examination, and genetics (if possible). A differential diagnosis should be made to rule out other similar clinical conditions.
• Association with a syndrome must be ruled out before any active intervention is initiated.
• Patient’s age, psychology, and access to interdisciplinary dental care need to be taken into consideration before finalizing the treatment approach.
• Early-morning appointments are advised to allay the patient’s apprehension.
• A brief course of antihypersensitivity medications and anti-inflammatory drugs is advised.
• After taking sensitivity control measures, comprehensive full-mouth hand scaling should be performed—possibly under local anesthesia. Care should be taken to prevent soft enamel from chipping and to decrease the chances of gouging exposed dentin.
• Subgingival curettage can be performed in deep pockets, taking care that soft cementum not be removed excessively.
• Complete restoration and full-mouth rehabilitation with esthetic crowns may be done in severe cases associated with the KLK-4 gene, which affects enamel-bonding properties.
• In cases of other gene-associated AI, occlusal glass-ionomer cement restorations and composite veneers can be provided.
• Regular patient reevaluation must be done in order to enhance treatment outcome and patients compliance.
The Future
In the future, genetic testing will be available to identify the specific mutation that causes inherited diseases in a given family. Mutation analysis, protein expression, and western blotting are among the diagnostic tools used to determine the exact type of AI.11 Recombinant enamel proteins are currently being tested with respect to their effect on crystal growth, as the KLK-4 gene mutation leads to the formation of enamel with poor bonding characteristics. Optimal restorative adhesive dentistry in these patients requires a significantly improved understanding of the process of tooth development.9
Genetic Counseling
It is clear that genetic heterogeneity may complicate discussion of Mendelian inheritance and classification,5 but with increasing access to molecular identification, families can discuss likely risk for AI and future problems.12 These conditions are often embarrassing, distressing, and lead to social exclusion and ridicule. Sensitive interviews and early supportive interventions are essential.5
Conclusions
AI is a serious problem that can result in reduced oral health–related quality of life and cause psychological problems. This condition—especially when syndrome-associated—requires extensive dental and periodontal treatment and benefits from a multidisciplinary approach. While planning management, the patient’s age, severity of the disorder, and access to appropriate dental care should be taken into consideration.13
About the Authors
Robert A. Horowitz, DDS
Clinical Assistant Professor, Departments of Implant Dentistry and Periodontics, Oral Surgery, New York University College of Dentistry, New York, New York, USA
D.K Gautam, BDS, MDS
Professor and Head, Department of Periodontology, Himachal Dental College, Sundernagar, India
Suneet Karol, BDS, MDS
Senior Lecturer, Department of Periodontology, HP Government Dental College, Shimla, India
Bindiya Kumari, BDS, MDS
Junior resident, Department of Periodontology and Implantology, HP Government Dental College, Shimla, India
References
1. Neville B, Damn DD, Allen CM, Bouquot J. Oral and Maxillofacial Pathology. 3rd ed. St. Louis, MI: Saunders Elsevier; 2008.
2. Simmer JP, Fincham AG. Molecular mechanisms of dental enamel formation. Crit Rev Oral Biol Med. 1995;6(2):84-108.
3. Simmer JP, Hu JC. Expression, structure, and function of enamel proteinases. Connect Tissue Res. 2002;43(2-3):441-449.
4. Canger EM, Celenk P, Yenísey M, Odyakmaz SZ. Amelogenesis imperfecta, hypoplastic type associated with some dental abnormalities: a case report. Braz Dent J. 2010;21(2):170-174.
5. Crawford PJM, Aldred M, Bloch-Zupan A. Amelogenesis imperfecta. Orphanet J Rare Dis. 2007;2:17.
6. Rajpar MH, Harley K, Laing C, et al. Mutation of genes encoding the enamel-specific protein, enamelin, causes autosomal-dominant amelogenesis imperfecta. Hum Mol Genet. 2001;10(16):1673-1677.
7. Wright JT. The molecular etiologies and associated phenotypes of amelogenesis imperfecta. Am J Med Genet A. 2006;140(23):2547-2555.
8. Borovjagin AV, Dong J, Passineau MJ, et al. Adenovirus gene transfer to amelogenesis imperfecta ameloblast-like cells. PLoS One. 2011;6(10):e24281.
9. Simmer JP, Hu JC. Dental enamel formation and its impact on clinical dentistry. J Dent Educ. 2001;65(9):896-905.
10. Witkop CJ Jr. Amelogenesis imperfecta, dentinogenesis imperfecta and dentin dysplasia revisited: problems in classification. J Oral Pathol. 1988;17(9-10):547-553.
11. Ozdemir D, Hart PS, Ryu OH, et al. MMP20 active-site mutation in hypomaturation amelogenesis imperfecta. J Dent Res. 2005;84(11):1031-1035.
12. Sockalingam S. Dental rehabilitation of amelogenesis imperfecta using thermoformed templates. J Indian Soc Pedod Prev Dent. 2011;29(1):53-56.
13. Santos MC, Hart PS, Ramaswami M, et al. Exclusion of known gene for enamel development in two Brazilian families with amelogenesis imperfecta. Head Face Med. 2007;3:8.