The Dual-Zone Therapeutic Concept of Managing Immediate Implant Placement and Provisional Restoration in Anterior Extraction Sockets
Abstract
Improvements in implant designs have helped advance successful immediate anterior implant placement into fresh extraction sockets. Clinical techniques described in this case enable practitioners to achieve predictable esthetic success using a method that limits the amount of buccal contour change of the extraction site ridge and potentially enhances the thickness of the peri-implant soft tissues coronal to the implant-abutment interface. This approach involves atraumatic tooth removal without flap elevation, and placing a bone graft into the residual gap around an immediate fresh-socket anterior implant with a screw-retained provisional restoration acting as a prosthetic socket seal device.
For more than 2 decades, the clinical protocol for immediate anterior implant placement into fresh extraction sockets has evolved from a two-stage protocol with full-thickness flaps1,2 to a one-stage protocol3—often flapless4,5 and sometimes with an immediate provisional restoration placed at the same appointment without compromising implant survival rates.6-8 This evolution was facilitated by improvements in the macro- and micro-geometry of implant designs and their respective restorative components as well as validation from animal9,10 and human11,12 clinical research.
The ongoing challenge for clinicians utilizing immediate anterior implant placement protocols today is no longer just achieving osseointegration, the rates of which are extremely high.13,14 Instead, the challenge is improving on protocols that allow for less traumatic, more time-efficient and yet highly predictable esthetic treatment outcomes in the more demanding anterior region.15-18 The main determinants to achieving long-term esthetic predictability in this environment are related to understanding and managing a complex combination of clinical and biologic factors, as follows:
1. diagnosis and classification of the extraction site in the esthetic zone
2. the natural biologic modeling and remodeling of extraction sockets
3. the relationship of underlying bone to the overlying soft-tissue profile and their stability
4. the relationship of labial and vertical soft-tissue thickness and height around implant-supported restorations
5. site-specific surgical protocols that minimize negative dimensional alteration of the site
6. restorative protocols and materials that optimize healing as well as soft-tissue stability and color
Diagnosis and Classification
Since not all extraction sites are the same, Salama and Salama19 as well as Elian et al20 suggested extraction site classification schemes that were meant to be utilized in developing site-specific treatment designs based on gingival margin level as well as the presence or absence of the labial and interproximal bone surrounding the compromised tooth to be extracted. Tarnow et al21 and Salama et al22 demonstrated the direct relationship of the position of the interproximal bone surrounding a tooth or implant, respectively, to the location and shape of the overlying papilla. Where deficiencies existed in any of the criteria listed above, they would be classified as Type II or Type III sites, depending on severity.23 For such compromised sites, surgical augmentation protocols24,25 and/or orthodontic site development19 would be required to augment and reconstruct the lost hard and soft tissues prior to26,27 or at the time of implant placement.28,29 However, whenever the extraction socket is not compromised—with an esthetically acceptable mid-facial gingival margin position and a completely intact labial plate and interproximal bone levels—a Type I classification is rendered, demanding an emphasis on hard- and soft-tissue preservation protocols.
When a tooth is extracted, the dimensional change of height and width of the alveolar ridge will occur. The reduction of the height of the bony wall is more pronounced at the buccal than lingual aspect of the extraction socket.30 This correlates to the amount of vertical loss of bone height, which is also more pronounced on the buccal wall. It has been well documented that major contour changes of the alveolar process take place during the first 6 months following tooth extraction. This results in the reduction of the width of the alveolar ridge and the vertical height.31-34 This reduction interferes with the placement of dental implants and influences the success of the prosthesis with regard to esthetics. If the implant is not placed more to the palatal aspect and apical in the extraction socket to assure the proper emergence profile, the optimal esthetic outcome may be compromised.24,35 This buccal bone resorption caused by excessive buccal placement of an implant into a fresh extraction socket may exaggerate these complications.17,36 These esthetic complications are emphasized in patients with a thin periodontal phenotype, and an additional surgical intervention may be required to remediate the surrounding soft-tissue architecture before, during, or after implant placement.28,29
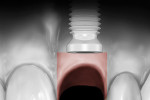
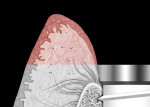
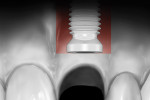
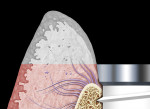
The dual zone is divided into two regions: the tissue zone and bone zone. The tissue zone is the labial-palatal dimensional change of the vertical region of tissue defined as ranging from the free gingival margin (0 mm) to the labial crest of bone mid-facially (Figure 1 and Figure 2). The bone zone is the tissue apical to the osseous crest (Figure 3 and Figure 4).37 After tooth removal, implant placement, bone grafting, and provisional restoration, the contour of the ridge can change. This article provides a clinical example of a case that illustrates the use of these techniques to minimize contour change associated with immediate anterior implants.
Surgical- and Restorative-Specific Tissue Preservation Protocols
The most critical surgical strategy for immediate implant placement and provisional restoration is atraumatic tooth removal without flap elevation—especially in the esthetic zone, where the buccal bone plate and soft tissues are thinnest in buccal-palatal dimension.12 The rationale for this procedure is to maintain the remaining blood supply from the periosteum and endosteum for maximum healing potential; the periodontal ligament is the third blood supply that is eliminated during tooth removal. If necessary, any of the anterior six teeth can be sectioned bucco-lingually with a surgical-length bur to achieve atraumatic removal without trauma to the buccal plate. The implant should be placed palatally and 3 mm to 4 mm apical to the free gingival margin (FGM) to optimize esthetics. Autogenous, allograft, xenograft, and synthetic bone replacements, substitutes, and/or materials can be used in the gap to graft the bone and tissue zones, respectively. The graft material helps serve as a scaffold to maintain hard- and soft-tissue volume as well as blood clot for initial healing. A recent article by Araújo, Linder, and Lindhe suggested that a xenograft material can be used in the gap and the particles can be incorporated into the soft-tissue profile without any inflammatory reaction.38 These incorporated particles provide substance to increase the soft-tissue profile.
The provisional restoration subsequently can act as a “prosthetic socket-sealing” device39 to protect, contain, and maintain the blood clot and bone-graft material during the healing phase of treatment. Table 1 outlines the treatment sequence and accompanying procedure for this clinical technique.
Case Presentation
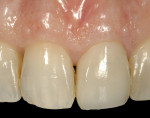
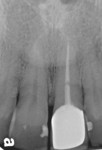
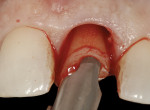
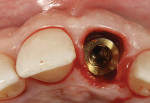
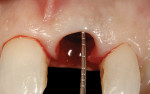
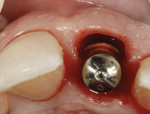
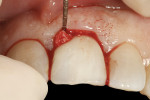
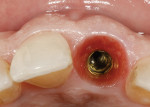
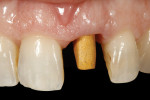
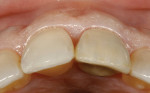
A 48-year-old Caucasian female presented with an oblique root fracture of tooth No. 9 (Figure 5). The patient complained of slight swelling toward the palatal rugae area adjacent to the tooth. The periapical radiograph revealed a horizontal-oblique line coincident with the apical extent of the post-core restoration (Figure 6). An irreversible hydrocolloid impression (Jeltrate® Alginate Impression Material, DENTSPLY Caulk, www.caulk.com) was made and an acrylic “egg-shell” was fabricated using the powder-liquid or Nealon technique.40 Excess material was trimmed accordingly to create the finalized provisional egg shell prior to relining (Figure 7). Sharp dissection of the supracrestal fibers was performed with a 15c scalpel blade, and the tooth was removed atraumatically with extraction forceps (Figure 8 and Figure 9). Due to the fracture, sectioning of the tooth was not needed. After socket debridement with a surgical excavator, a 5-mm-diameter threaded-design, and textured-surface implant (OSSEOTITE® Certain® FOSS, Biomet 3i, www.biomet3i.com) was placed toward the palatal aspect of the extraction socket to a depth of 3 mm from the FGM equivalent to the mid-facial osseous crest (Figure 10 and Figure 11). It is important to note that flap elevation was not necessary for tooth extraction or immediate implant placement, and the buccal bone plate was intact after tooth removal.
The appropriate screw-retained polyether-ether-ketone (PEEK) provisional abutment (PreFormance®, Biomet 3i) was placed using the platform-switch concept during the provisional restoration phase, which was continued through to the definitive restoration (Figure 12). The acrylic egg shell was then filled with an autopolymerizing resin (Super T, American Consolidated Manufacturing Co.), seated spatially onto the abutment in the proper facial and vertical positions, and allowed to set prior to removal (Figure 13). It is critical to place enough acrylic resin material to secure the abutment mechanically and capture the subgingival contour of the soft-tissue profile; however, this must be done without engaging the undercut regions of the proximal contact areas, as doing so would make provisional removal extremely difficult and could potentially be harmful to the implant. Therefore, a minimum torque value of 30 to 35 Ncm should be achieved upon implant placement to allow immediate provisional restoration.
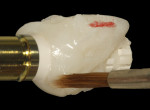
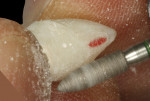
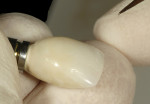
The relined provisional restoration was then disinfected and placed onto a laboratory analog/replica, the proximal contact areas were marked with a red wax pencil, and the subgingival contours were re-established with resin using a sable-haired brush (Figure 14). The lingual contour of the provisional restoration was shaped with a heatless stone using a straight mandrel on a low-speed handpiece. The marked proximal contact areas, which define the buccal extent of acrylic resin removal, must be respected. Enough resin must be left palatally to keep the provisional restoration intact, stable, and secure to the PEEK abutment. Appropriate laboratory diamond burs were employed to trim the final shape/contour of the provisional restoration (Figure 15). After polishing with flour of pumice, extrinsic acrylic colorants were applied to the provisional restoration surface; it should be noted that the surface colorants should be applied only to the supragingival area of the provisional restoration (Figure 16). The provisional restoration was re-placed onto the implant and assessed clinically using the following criteria: 1) the color and form of the tooth replaced must be correct; 2) the subgingival contours must be supportive of the soft-tissue profile; and 3) the tooth must be in infraocclusion in maximum intercuspal position and lateral excursive movements (Figure 17).
It is paramount that the provisional restoration be fabricated first, prior to the placement of the bone graft material, since the graft material must remain intact and uncontaminated during the procedure. The best way to achieve this is to remove the newly fabricated provisional restoration precisely before bone graft placement.
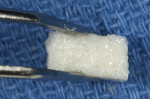
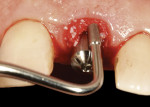
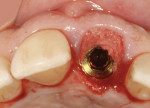
To accomplish this, a flat-contoured healing abutment was placed onto the implant after the provisional restoration was removed (Figure 18). A xenograft bone graft material (Bio-Oss®, Geistlich Pharma AG, www.geistlich.com) was used in this specific instance to graft the bone and tissue zones (Figure 19). The abutment must be tall and narrow enough to allow the bone graft to be placed and packed against it with an amalgam-condensing instrument up to the most coronal aspect of the FGM, thereby maximizing the amount of graft material that can be placed into the tissue zone (Figure 20). The flat-profiled healing abutment was then removed, leaving the bone graft material undisturbed (Figure 21), and the provisional restoration was successively re-placed onto the implant, tightened with finger pressure. The extruded excess bone graft material was removed to the FGM with a periodontal probe (Figure 22), and a posttreatment radiograph was taken. A periodontal curette could also have been used to accomplish this procedure.
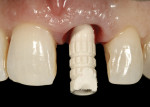
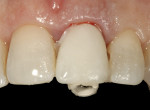
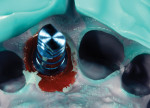
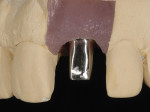
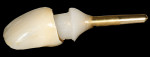
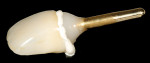
The patient was instructed not to toothbrush the area for 3 days, and a postoperative examination was performed 1 week later. One- and 3-month follow-up visits were performed. At 5 months, a periapical digital film was taken to verify radiographic healing post-immediate implant and provisional restoration treatment (Figure 23). The provisional restoration was removed for the first time, and a healthy implant gingival sulcus was noted (Figure 24 and Figure 25). Subsequently, an implant-level transfer coping was attached for an open-tray impression, and pattern resin was used to capture the subgingival soft-tissue profile (Figure 26). A smaller diameter transfer coping (5 mm to 4 mm) was used to create a platform-switched restoration. The dental laboratory fabricated a soft-tissue cast that allowed a screw-retained gold alloy abutment for a cement-retained crown restoration to be constructed (Figure 27 and Figure 28). In order to control the correct shade of the gingival tissues and thereby eliminate the “gray effect,”41 the gold alloy abutment was gold plated (Dentsply® Gold Plating Solution, DENTSPLY) (Figure 29). The gold-plated custom abutment was inserted intraorally, and a duplicate cementing-die replica was fabricated using a bis-acryl material (Luxatemp® Ultra, DMG America, www.dmg-dental.com) prior to crown insertion (Figure 30).42 The fabrication of the cementing-die replica was done by lubricating the internal aspect of the crown, expressing the bis-acryl material into the crown, placing a metal die pin into the unset material, and then removing the die from the crown after polymerization (Figure 31). The crown could then be filled with a cementing medium (TempBond® NE™, Kerr Dental, www.kerrdental.com) and seated on the die replica, and the excess cement removed prior to intraoral seating of the restoration (Figure 32).
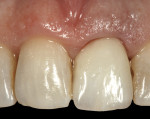
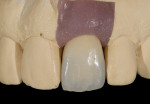
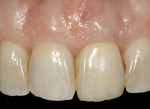
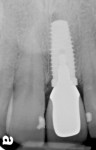
The clinical strategy and benefit of this technique is to allow the indirect removal of excess cement around the final crown. This clinical step is extremely important to avoid the potential for peri-implantitis due to residual cement left in the implant gingival sulcus. The definitive crown restoration can, thus, be cemented with confidence, respecting periodontal health (Figure 33). From a three-quarter occlusal intraoral view, there was little to no clinical indication of a contour difference between the untreated tooth No. 8 versus the treated tooth No. 9 (Figure 34). The 6-month posttreatment periapical radiograph showed good bone levels around the platform-switched implant tooth No. 9 (Figure 35).
Summary
To achieve predictable esthetic success with immediate placement of implants into fresh anterior extraction sockets, the critical clinical keys that must be respected are: 1) atraumatic tooth removal without flap elevation; and 2) placement of a bone graft into the bone and tissue zones in the residual gap around an immediate fresh-socket implant, with a screw-retained provisional restoration acting as a prosthetic socket seal device. These clinical steps are helpful to limit the amount of buccal contour change of the extraction site ridge and potentially enhance the thickness of the peri-implant soft tissues coronal to the implant-abutment interface.
Acknowledgments
The authors would like to thank Mr. Adam Mieleszko, CDT, and Mr. Luis Santiago for the laboratory support in the fabrication of the restoration shown.
References
1. Schulte W, Kleineikenscheidt H, Lindner K, et al. Animal experiments on the question of healing around the Tubingen immediate implant (German). Dtsch Zahnarztl Z. 1978;33(5):326-331.
2. Becker W, Goldstein M, Becker BE, Sennerby L. Minimally invasive flapless implant surgery: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S21-S27.
3. Buser D, Mericske-Stern R, Bernard JP, et al. Long-term evaluation of non-submerged ITI implants. Part 1: 8-year life table analysis of a prospective multi-center study with 2359 implants. Clin Oral Implants Res. 1997;8(3):161-172.
4. Becker W, Wikesjö U, Sennerby L, et al. Histologic evaluation of implants following flapless and flapped surgery: a study in canines. J Periodontol. 2006;77(10):1717-1722.
5. Wöhrle PS. Single-tooth replacement in the aesthetic zone with immediate provisionalization: fourteen consecutive case reports. Pract Periodontics Aesthet Dent. 1998;10(9):1107-1114.
6. Kan JY, Rungcharassaeng K, Lozada J. Immediate placement and provisionalization of maxillary anterior single implants: 1-year prospective study. Int J Oral Maxillofac Implants. 2003;18(1):31-39.
7. De Rouck T, Collys K, Wyn I, Cosyn J. Instant provisionalization of immediate single-tooth implants is essential to optimize esthetic treatment outcome. Clin Oral Implants Res. 2009;20(6):566-570.
8. Block MS, Mercante DE, Lirette D, et al. Prospective evaluation of immediate and delayed provisional single tooth restorations. J Oral Maxillofac Surg. 2009;67(11 suppl):89-107.
9. Araújo MG, Sukekava F, Wennström JL, Lindhe J. Ridge alterations following implant placement in fresh extraction sockets: an experimental study in the dog. J Clin Periodontol. 2005;32(6):645-652.
10. Araújo MG, Wennström JL, Lindhe J. Modeling of the buccal and lingual bone walls of fresh extraction sites following implant installation. Clin Oral Implants Res. 2006;17(6):606-614.
11. Cooper LF, Raes F, Reside GJ, et al. Comparison of radiographic and clinical outcomes following immediate provisionalization of single-tooth dental implants placed in healed alveolar ridges and extraction sockets. Int J Oral Maxillofac Implants. 2010;25(6):1222-1232.
12. Sanz M, Cecchinato D, Ferrus J, et al. A prospective, randomized-controlled clinical trial to evaluate bone preservation using implants with different geometry placed into extraction sockets in the maxilla. Clin Oral Implants Res. 2010;21(1):13-21.
13. Schnitman PA, Wöhrle PS, Rubenstein JE, et al. Ten-year results for Brånemark implants immediately loaded with fixed prostheses at implant placement. Int J Oral Maxillofac Implants. 1997;12(4):495-503.
14. Tarnow DP, Emtiaz S, Classi A. Immediate loading of threaded implants at stage 1 surgery in edentulous arches: ten consecutive case reports with 1- to 5-year data. Int J Oral Maxillofac Implants. 1997;12(3):319-324.
15. Tarnow DP, Chu SJ. Human histologic verification of osseointegration of an immediate implant placed into a fresh extraction socket with excessive gap distance without primary flap closure, graft, or membrane: a case report. Int J Periodontics Restorative Dent. 2011;31(5):515-521.
16. Chen ST, Darby IB, Reynolds EC. A prospective clinical study of non-submerged immediate implants: clinical outcomes and esthetic results. Clin Oral Implants Res. 2007;18(5):552-562.
17. Chen ST, Buser D. Clinical and esthetic outcomes of implants placed in postextraction sites. Int J Oral Maxillofac Implants. 2009;24 Supp:186-217.
18. Kan JY, Morimoto T, Rungcharassaeng K, et al. Gingival biotype assessment in the esthetic zone: visual versus direct measurement. Int J Periodontics Restorative Dent. 2010;30(3):237-243.
19. Salama H, Salama M. The role of orthodontic extrusive remodeling in the enhancement of soft and hard tissue profiles prior to implant placement: a systematic approach to the management of extraction site defects. Int J Periodontics Restorative Dent. 1993;13(4):312-333.
20. Elian N, Cho SC, Froum S, et al. A simplified socket classification and repair technique. Pract Proced Aesthet Dent. 2007;19(2):99-104.
21. Tarnow DP, Magner AW, Fletcher P. The effect of the distance from the contact point to the crest of bone on the presence or absence of the interproximal dental papilla. J Periodontol. 1992;63(12):995-996.
22. Salama H, Salama MA, Garber D, Adar P. The interproximal height of bone: a guidepost to predictable aesthetic strategies and soft tissue contours in anterior tooth replacement. Pract Periodontics Aesthet Dent. 1998;10(9):1131-1141.
23. Funato A, Salama MA, Ishikawa T, et al. Timing, positioning, and sequential staging in esthetic implant therapy: a four-dimensional perspective. Int J Periodontics Restorative Dent. 2007;27(4):313-323.
24. Buser D, Ruskin J, Higginbottom F, et al. Osseointegration of titanium implants in bone regenerated in membrane-protected defects: a histologic study in the canine mandible. Int J Oral Maxillofac Implants. 1995;10(6):666-681.
25. Wang HL, Boyapati L. “PASS” principles for predictable bone regeneration. Implant Dent. 2006;15(1):8-17.
26. Buser D, Dula K, Belser UC, et al. Localized ridge augmentation using guided bone regeneration. II. Surgical procedure in the mandible. Int J Periodontics Restorative Dent. 1995;15(1):10-29.
27. Buser D, Dula K, Hess D, et al. Localized ridge augmentation with autografts and barrier membranes. Periodontol 2000.1999;19(1):151-163.
28. Grunder U. Crestal ridge width changes when placing implants at the time of tooth extraction with and without soft tissue augmentation after a healing period of 6 months: report of 24 consecutive cases. Int J Periodontics Restorative Dent. 2011;31(1):9-17.
29. Kan JY, Rungcharassaeng K, Lozada JL, Zimmerman G. Facial gingival tissue stability following immediate placement and provisionalization of maxillary anterior single implants: a 2- to 8-year follow-up. Int J Oral Maxillofac Implants. 2011;26(1):179-187.
30. Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
31. Araújo MG, Lindhe J. Ridge alterations following tooth extraction with and without flap elevation: an experimental study in the dog. Clin Oral Implants Res. 2009;20(6):545-549.
32. Iasella JM, Greenwell H, Miller RL, et al. Ridge preservation with freeze-dried bone allograft and a collagen membrane compared to extraction alone for implant site development: a clinical and histologic study in humans. J Periodontal. 2003;74(7):990-999.
33. Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23(4):313-323.
34. Becker W, Goldstein M. Immediate implant placement: treatment planning and surgical steps for successful outcome. Periodontol 2000. 2008;47(1):79-89.
35. Caneva M, Salata LA, de Souza SS, et al. Influence of implant positioning in extraction sockets on osseointegration: histomorphometric analyses in dogs. Clin Oral Implant Res. 2010;21(1):43-49.
36. Caneva M, Salata LA, de Souza SS, et al. Hard tissue formation adjacent to implants of various size and configuration immediately placed into extraction sockets: an experimental study in dogs. Clin Oral Implant Res. 2010;21(9):885-890.
37. Tarnow DT, Salama MA, Garber D, et al. The dual-zone therapeutic concept of managing immediate implant placement into anterior extraction sockets: Part 1 – Labial-palatal changes in ridge dimension with and without provisional restoration and/or bone grafting: a retrospective cross-sectional cohort multicenter study. Int J Periodontics Restorative Dent. Manuscript submitted for publication.
38. Araújo MG, Linder E, Lindhe J. Bio-Oss collagen in the buccal gap at immediate implants: a 6-month study in the dog. Clin Oral Implants Res. 2011;22(1):1-8.
39. Trimpou G, Weigl P, Krebs M, et al. Rationale for esthetic tissue preservation of a fresh extraction socket by an implant treatment concept simulating a tooth replantation. Dent Traumatol. 2010;26(1):105-111.
40. Nealon FH. Acrylic restorations by the operative nonpressure procedure. J Prosthet Dent. 1952;2(4):513-527.
41. Ishikawa-Nagai S, Da Silva JD, Weber HP, Park SE. Optical phenomenon of peri-implant soft tissue. Part II. Preferred implant neck color to improve soft tissue esthetics. Clin Oral Implants Res. 2007;18(5):575-580.
42. Wadhwani C, Piñeyro A. Technique for controlling the cement for an implant crown. J Prosthet Dent. 2009;102(1):57-58.
Have a comment or question on this topic? Join the Implant Community today to interact with your colleagues at dentalaegis.com/go/cced183
About the Authors
Stephen J. Chu, DMD, MSD, CDT
Clinical Associate Professor
Director of Esthetic Education
Columbia University College of Dental Medicine
New York, New York
Maurice A. Salama, DMD
Private Practice
Atlanta, Georgia
Henry Salama, DMD
Private Practice, Atlanta, Georgia
David A. Garber, DDS, BDS
Private Practice
Atlanta, Georgia
Hanae Saito, DDS, MS
Postgraduate Periodontology Resident,
New York University College of Dentistry
New York, New York
Guido O. Sarnachiaro, DDS
Clinical Assistant Professor
Department of Prosthodontics
Columbia University School of Dentistry
New York, New York
Clinical Assistant Professor
Department of Periodontology-Oral Implantology
Temple University School of Dentistry
Philadelphia, Pennsylvania
Private Practice
New York, New York
Dennis P. Tarnow, DDS
Clinical Professor,
Director of Implant Education,
Columbia University College of Dental Medicine,
New York, New York