Zirconia: A Proven, Durable Ceramic for Esthetic Restorations
Abstract
In dental ceramics, zirconia has proven to be a durable, reliable framework material capable of inhibiting crack growth and preventing catastrophic failure. Zirconia, which is the oxidized form of zirconium, can exist in several phases, depending on temperature. Used in combination with CAD/CAM technology, zirconia enables the fabrication of esthetic all-ceramic restorations in all areas of the mouth.
Computer-aided design/computer-aided manufacturing (CAD/CAM) has enabled materials to be used in dental applications that typically could not be fabricated by conventional means. One of the most significant of these materials is yttria partially stabilized tetragonal zirconia. Zirconia (ZrO2) is the oxidized form of zirconium (Zr) just as alumina (Al2O3) is an oxide of aluminum (Al).
Zirconia Properties
The most widely used all-ceramic material is what is commonly referred to in dentistry as “pure zirconia.” While not exactly pure, machinable zirconia is predominately zirconia with stabilizing components such as yttria and minor components such as alumina, which are important for long-term stability. Zirconia may exist primarily in the tetragonal phase at room temperature by adding components such as calcia (CaO), magnesia (MgO), yttria (Y2O3), and ceria (CeO2).1
The addition of 3wt% to 5wt% yttria produces a partially stabilized zirconia. Although stabilized at room temperature, the tetragonal zirconia phase may change under stress to monoclinic phase with a subsequent 3% volumetric increase. This dimensional change diverts energy from crack formation and can stop a crack from progressing further. This is called transformation toughening. This material characteristic helps zirconia frameworks resist catastrophic failure. Even though a crack may exist in the material, the phase change prevents it from proceeding throughout the restoration. Adding higher amounts of stabilizing materials produces the cubic phase of zirconia at room temperature, which replicates the appearance, though not the expense, of real diamonds.1
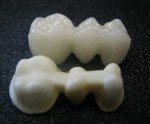
Fully dense zirconia is extremely difficult to machine, taking up to two hours just to fabricate a single unit. Therefore, most restorations with zirconia frameworks are fabricated by machining a porous or partially fired block of zirconia. The framework is milled oversize and then fired from approximately 1,350°C to 1,500°C to fully densify the zirconia, producing a translucent material and strength values from 900 MPa to 1,200 MPa.2,3 A large amount of sintering shrinkage, typically 20% to 30%, occurs in transitioning from the milled framework to the dense framework (Figure 1). The different firing temperatures are related to the particle size of the powder in the block—generally, the smaller the initial size, the lower the firing temperature. Most blocks have barcodes that communicate the density of the milling block to the computer in order to properly mill the framework oversize. Over- or under-firing may affect the final crystal size, porosity, mechanical properties, and fit.
Issues of Concern
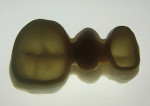
Of the all-ceramic restorations produced in 2010, zirconia-based units represented approximately 50% of that total. As zirconia’s popularity has increased, so, too, has the number of suppliers of machinable blocks. Differences among zirconia ceramics can be found in the level of zirconia purity, grain size, trace elements, as well as stabilizing compounds. Differences also exist among zirconia powders and the processing of those powders into blocks. The high cost of blocks is not necessarily due to the material, but can be attributed to the processing required to produce a reliable, homogeneous block. Poor-quality zirconia blocks may warp and have significant porosity, as seen in Figure 2 and Figure 3. Consequently, clinical outcomes such as fit, translucency, strength, and long-term stability can be significantly affected. Also, zirconia may degrade over time; minor components such as alumina help prevent this degradation.4
One concern that is the subject of research and debate is the issue of chipping of the veneering porcelain. In tests of zirconia itself, the zirconia does not seem to fail. However, there have been numerous reports of veneer chipping. In several published studies, the veneer chipping rate was approximately 15% after 3 to 5 years.5-7 A review of various clinical trials on zirconia restorations showed a wide range of chipping at 1 to 5 years from 5% to 25% for low-fusing porcelains such as Cercon® Ceram (DeguDent, www.degudent.com), Lava™ Ceram (3M ESPE, www.3Mespe.com), and IPS e.max® Ceram (Ivoclar Vivadent, www.ivoclarvivadent.com). Chipping rose to 54% after 1 year for Triceram® (Dentaurum, www.dentaurum.com) porcelain on dense zirconia.8,9 However, others report minor amounts of chipping problems.10,11
It is important to note that there appears to be a correlation with the firing cycle and peak firing temperature of the veneer. Laboratory studies have shown that low-fusing porcelains may be less resistant to cracking than high-fusing porcelains and the porcelain density is lower. Zirconia is a good thermal insulator that prevents heat transfer to the veneer porcelain, stopping it from becoming fully dense whether fast-fired or fired at a low temperature. Also, fast cooling may create stress in the porcelain, which could lead to cracking. Any surface adjustment, such as grinding, sandblasting, and even polishing, can change the phase on the surface of the zirconia and may affect the stability and strength of the zirconia as well as the veneer porcelain.12-14 Another area of interest is the use of zirconia as a full-contour restoration. Although zirconia has a fine microstructure, there are varied reports as to its abrasion properties, and there are no published clinical studies on the longevity of full-contour zirconia and the wear of opposing dentition. There are also no international standards for CAD/CAM and CAD/CAM materials such as zirconia. It is important to keep in mind that zirconia blocks approved by the US Food and Drug Administration (FDA) generally are not required to demonstrate any specific mechanical or functional properties. The blocks are approved in a process in which they are shown to be equivalent to a previous block on the market that has demonstrated biocompatibility and clinical success, so-called “510K.” The zirconia blocks to date are approved for frameworks, but not specifically for other applications such as full-contour crowns, bridges, or large implant-supported substructures.
Some research shows that zirconia is abrasive to the opposing dentition. Studies using natural teeth have shown that zirconia causes excessive wear of the tooth structure. This is particularly true if an as-machined surface is simply glazed. If glazed, the glaze eventually wears off, exposing a rough surface that then accelerates tooth wear. If the zirconia is not polished to a mirror finish then the zirconia may cause excessive wear.15 Other studies in progress demonstrate that polished zirconia is “wear-kind.”16 Recent studies using an enamel substitute, steatite (soapstone), tend to show that highly polished zirconia is wear-kind.17 If producing zirconia crowns, the surfaces must be highly polished and remain intact. If the occlusion needs to be adjusted and the clinician cannot achieve a high polish, the restoration should be sent back to the lab for finishing.
Excessive or coarse grinding can cause cracks, which penetrate into the zirconia substructure, causing transformation that may initially hold these cracks closed. However, over time with exposure to the oral environment and reversal of the transformation stress, the cracks can begin to propagate. Furthermore, many of the “high-translucency zirconias” have low amounts of alumina, which is an important stabilizer. Lower amounts of alumina may cause excessive transformation, cracking, and tooth abrasion7 ; however, clinical trials are expected to provide a more definitive answer.
Milled Framework and Milled Veneer Restorations
A possible solution to the veneer chipping problem as well as a method for quickly fabricating the veneer for frameworks involves the use of a milled veneer fused or bonded to a zirconia framework. There are currently three products available for clinical use. Two of them, Vita Rapid Layer Technology (RLT) (Vident, www.vident.com) and IPS e.max® CAD-on (Ivoclar Vivadent), may be used for single units and three-unit posterior bridges. The third product, the Lava™ Digital Veneering System (DVS) (3M ESPE), is presently limited to single units.
The milled veneer approach involves using the Sirona inLab® system (Sirona Dental Systems, www.sirona.com) to produce a “split file”; two files are created when fabricating a full-contour bridge. One file is used to mill the zirconia framework, and the second file mills a corresponding full-contour veneer from a fully dense block. In the case of RLT, a Triluxe (Vident) multicolor block is milled to produce the veneer. This milled veneer is then bonded onto the zirconia framework using composite resin cement (Figure 4 and Figure 5). This milling concept holds forth the possibility of completely eliminating physical casts for three-unit bridges. Combining the CEREC® (Sirona Dental Systems) digital impression of the preparation, opposing teeth, and bite registration may produce a virtual model in which the proper occlusal contacts are generated in the milled veneer. Thus, no physical articulation is needed to develop the correct occlusal contacts.
This split-file software technique is also used by Ivoclar Vivadent to produce zirconia frameworks veneered with machined e.max CAD blocks. The CAD-on technique combines a substructure milled from IPS e.max ZirCAD zirconia and a veneer milled from IPS e.max CAD lithium disilicate. The IPS e.max CAD is crystallized after machining using a furnace. The two components are joined using IPS e.max CAD Crystall/Connect fusion glass, which melts during a firing cycle. The molten glass then bonds the framework and veneer together.
The third milled veneer system, DVS from 3M ESPE, is designed for single-unit restorations. In this case, the zirconia coping is milled and a data file to produce the milled veneer is made. The veneer, however, is milled from a porous porcelain block. The veneer is attached to the zirconia with a special dentin fusing powder, and the combined components are fired to densify the porcelain veneer and fuse it to the zirconia.
Zirconia frameworks are most often luted to the tooth structure using glass-ionomer cement; however, several primers may be used to create a bond to zirconia. Glass ionomers bond weakly if at all.7 Zirconia bonding might improve retention to the tooth structure, and these resin cements are not soluble. However, there is a debatable issue with respect to sandblasting the zirconia. Most manufacturers recommend against the use of sandblasting on the veneer surface but allow low-pressure (25 psi to 50 psi, 2/3 bar) small-particle alumina (25 µm to 50 µm) sandblasting of the interior as the final step before cementing the restoration. This may help clean the internal surface and provide some mechanical retention for nonbonded cement. Some studies have demonstrated a potential problem in sandblasting the internal surface with respect to crack growth, while others have shown an improvement in properties.18,19
Zirconia Abutments
Zirconia blocks have been developed (by Sirona Dental Systems) to produce custom abutments for a variety of implant systems. An intermediate element (TiBase, Sirona) is used between the implant and zirconia. One end of the TiBase unit screws into the implant, and the milled zirconia abutment is bonded to the abutment portion of the TiBase. A partially fired zirconia block is milled to produce a custom abutment.
If cone beam data is merged with intraoral scan data, then an accurate custom abutment can be designed and milled. After sintering the zirconia, the abutment is bonded to the TiBase using composite resin cement, such as RelyX™ Unicem (3M ESPE) or Multilink® Automix (Ivoclar Vivadent). Initial studies show the bond of the zirconia to the titanium using composite to be sufficient to withstand intraoral stresses.
Conclusion
Zirconia has become one of the dominant types of ceramic used for a variety of CAD/CAM restoration classes, including framework/hand veneer, framework/milled veneer, full-contour fixed prosthodontics, implant abutments, and large implant-supported substructures. Although zirconia is currently the strongest dental ceramic available, fabrication variations and finishing procedures can affect the longevity of this material as well as the veneer porcelain. Having a good working relationship with a dental laboratory is strongly recommended.
References
1. Swain MV, Hannink RHJ. Metastability of the martensitic transformation in a 12 mol% ceria-zirconia alloy: II, grinding studies. Journal of the American Ceramic Society. 1989;72(8):1358-1364.
2. Swain MV. Limitation of maximum strength of zirconia-toughened ceramics by transformation toughening increment. Journal of the American Ceramic Society. 1985;68(4):C97-C99.
3. Xu HHK, Jahanmir S, Ives LK. Effect of grinding on strength of tetragonal zirconia and zirconia-toughened alumina. Machining Science and Technology. 1997;1(1):49-66.
4. Lawson S. Environmental degradation of zirconia ceramics. J Eur Ceram Soc. 1995;15(6):485-502.
5. Sailer I, Fehér A, Filser F, et al. Prospective clinical study of zirconia posterior fixed partial dentures: 3-year follow-up. Quintessence Int. 2006;37(9):685-693.
6. Vult von Steyern P, Carlson P, Nilner K. All-ceramic fixed partial dentures designed according to the DC-Zirkon technique. A 2-year clinical study. J Oral Rehabil. 2005;32(3):180-187.
7. Sailer I, Fehér A, Filser F, et al. Five-year clinical results of zirconia frameworks for posterior fixed partial dentures. Int J Prosthodont. 2007;20(4):383-388.
8. Al-Amleh B, Lyons K, Swain M. Clinical trials in zirconia: a systematic review. J Oral Rehabil. 2010;37(8):641-652.
9. Rothbrust, F, Keutschegger W, Kraxner S, et al. Microstructural effects on the clinical performance of anatomical zirconia [abstract]. J Dent Res. 2011;90(spec iss A). Abstract 3668.
10. Blatz M, Mante F, Chiche G, et al. Clinical survival of posterior zirconia crowns in private practice [abstract]. J Dent Res. 2010;89(spec iss B). Abstract 2110.
11. Nathanson D, Chu S, Yamamoto H, et al. Performance of zirconia based crowns and FPDs in prosthodontic practice [abstract]. J Dent Res. 2010;89(spec iss B). Abstract 2115.
12. Fahmi M, Pober R, Giordano R. Effect of surface treatment on porcelain bond strength to zirconia [abstract]. J Dent Res. 2007;86(spec iss A). Abstract 1571.
13. Fahmi M, Pober R, Giordano R. Thermal shock of porcelain veneered zirconia with various surface treatment [abstract]. J Dent Res. 2008;87(spec iss B). Abstract 0092.
14. Arrejaie A, Giordano R, Pober R. Mechanical properties of Y-TZP/porcelain interface with multiple surface treatments [abstract]. J Dent Res. 2010;89(spec iss A). Abstract 1571.
15. Shah S, Michelson C, Beck P, et al. Wear of enamel on polished and glazed zirconia [abstract]. J Dent Res. 2010;89(spec iss A). Abstract 227.
16. Sorensen JA, Sultan EA, Sorensen PN. Three-body wear of enamel against full crown ceramics [abstract]. J Dent Res. 2011;90(spec iss A). Abstract 1652.
17. Geis-Gerstorfer J, Schille C. Influence of surface treatment on wear of solid zirconia (LAVA) [abstract]. J Dent Res. 2011;90(spec iss A). Abstract 3056.
18. Bulot D, Sadan A, Burgess JO, Blatz MB. Bond strength of a self-adhesive universal resin cement to lava zirconia after two surface treatments [abstract]. J Dent Res. 2003;82(spec iss A). Abstract 0578.
19. Papanagiotou HP, Morgano SM, Giordano RA, Pober R. In vitro evaluation of low-temperature aging effects and finishing procedures on the flexural strength and structural stability of Y-TZP dental ceramics. J Prosthet Dent. 2006;96(3):154-164.
About the Author
Russell Giordano II, DMD, CAGS, DMSc
Associate Professor
Director of Biomaterials
Boston University Goldman School of Dental Medicine
Boston, Massachusetts