Immediate Temporization of Immediate Implants in the Esthetic Zone: Case Reports Evaluating Survival and Bone Maintenance
Barry P. Levin, DMD
Abstract: The literature supporting immediate temporization in the partially edentulous dentition is limited to a few controlled studies and case reports. Complicating issues for these situations include immediate placement, bone and/or soft-tissue deficiency, and demanding esthetic expectations. When challenged with extracting hopeless teeth, replacing with implants, and providing an immediate fixed restoration, the clinician should respect biologic and biomechanical principals. This study followed 30 consecutively placed implants in the esthetic zone, inserted at time of tooth extraction, and immediately temporized and augmented with bone grafting and resorbable guided bone regeneration. Implant survival, adverse events (eg, implant failure and infection), and esthetic outcomes were evaluated. In this study, the esthetic zone is defined as the dentition spanning maxillary or mandibular first bicuspids. All implants osseointegrated and were ready for definitive restorative therapy by 12 weeks. No adverse events, such as infection, persistent inflammation, or abutment screw loosening, occurred. Radiographic bone levels were documented. Clinical technique and rationale is also emphasized.
Patients receiving implants today are often unaware of the original loading protocols of 3 to 6 months of load-free healing prior to restorative therapy.1,2 Often, when patients require extraction of an anterior tooth, they do not accept even the shortest period of partial edentulism. Traditionally, these situations were addressed with either removable or provisional restorations. When these solutions were unsatisfactory, implant therapy was often rejected in lieu of traditional fixed partial dentures or resin-bonded bridges.
Immediate temporization was originally indicated for the completely edentulous arch. Investigators have demonstrated relatively similar implant survival rates compared with traditional loading protocols.3,4 Several guidelines derived from these studies and case reports have evolved. The provisional restoration must remain until osseointegration occurs. Meticulous occlusal schemes that minimize nonaxial forces are recommended. Most importantly, the implant must achieve primary stability.5 This may necessitate undersizing osteotomy diameters and using self-tapping, screw-shaped implants. Many investigators advocate a minimal insertion torque to serve as an indicator of primary stability. In a human study, Drago6 recommended an insertion torque of 30 ncm. Neugebauer7 found predictable osseointegration in the animal model when 35 ncm was required for implant placment.6,7 When fixtures are deemed stable within the osteotomy site, connection of a provisional splinted prosthesis in the edentulous arch can be performed with favorable expectations.
In recent years, the option of immediate placement performed in conjunction with temporization has become popular. Barone et al8 demonstrated the validity of this procedure, and Degidi et al9 presented a large number of immediately temporized implants with a 5-year success rate of 97.2%.
Since immediate implantation was first described,10,11 many clinicians and investigators have contributed to the high success rates that dentists enjoy. One controversy in immediate implant therapy is the behavior of the socket walls following tooth removal and the impact on the soft-tissue dimensions, which directly affect long-term esthetic outcomes. In a dog study, Araujo et al12 demonstrated that implant placement into an extraction socket did not prevent loss of the buccal-lingual dimensions of the ridge. In a study of 18 patients who received 21 implants, Botticelli et al13 showed when measures are not taken to counteract physiologic bone remodeling after extraction, more than 50% of the buccal bone dimensions are lost in the 4 months after immediate implantation. The researchers performed a follow-up study at 5 years and found all 21 implants were functioning. Five sites exhibited soft-tissue recession, exposing the buccal margin of the restoration. Four of these areas were in the mandibular arch. Both animal and human trials have shown implant placement alone into extraction sockets does not prevent bone loss and risk of esthetic failure. The implementation or simultaneous regenerative therapy not only provide bone support for osseointegration but also for long-term support of soft tissues has been well documented. In the canine model, Schwarz et al14 demonstrated the efficacy of combined guided bone regeneration (GBR) and bone replacement graft therapy. This study emphasized the usefulness of a bone graft material to support a resorbable membrane and act as an osteoconductive scaffold. In a human study comparing three different membranes in conjunction with a bone replacement graft to simultaneously augment peri-implant defects, Moses et al15 found significant defect resolution with combined therapy. They concluded that premature exposure of the barrier membrane, depending on membrane composition, may result in impaired bone healing. Resorbable membranes with a collagen cross-linked native metabolite demonstrated resistance to bacterial collagenolytic breakdown when exposed to the oral cavity, whereas the noncross-linked membrane led to impaired bone regeneration when exposed. In a historical perspective, Gelb16 and Shanaman17 advocated the use of bone allograft for support of nonresorbable expanded polytetrafluoroethylene (e-PTFE) barrier membranes to facilitate prosthetically driven implant placement. In a comparative study, Gher et al18 demonstrated crestal bone apposition when an allograft was combined with membrane therapy compared to slight crestal resorption when a barrier was used alone in immediate implant therapy. The evidence suggesting the use of a graft material to provide an osteoconductive scaffold for GBR support cannot be ignored. Cammack et al19 demonstrated the efficacy of human bone allograft in ridge and sinus augmentations, leading to more than 40% vital bone substitution. More recently, Levin20 demonstrated transmucosal healing of immediate implants simultaneously grafted with human bone allograft and a resorbable collagen membrane.
In recent years, implant design has evolved, with the purpose of maintaining crestal bone. Implant surface, thread design, and prosthetic connection all can be used to retain the height and thickness of bone around the neck of the implant. It is theorized that bone maintenance is critical for the support of soft tissue and long-term esthetic success. Most dental implant manufacturers produce implants with a rough surface. A novel, chemically modified surface (SLActive®, Straumann, https://www.straumann.us) has demonstrated rapid osseointegration.21 This titanium surface has shown the potential to spontaneously heal experimentally created dehiscence-type defects in the dog model.22
The location of the abutment-to-implant interface is another area of investigation regarding bone preservation. Two-piece implants, designed to be inserted to the bone crestal level, have historically shown crestal bone loss of approximately 1.5 mm to 2.0 mm.23,24 Hermann et al25 demonstrated in the dog model that the biologic remodeling of crestal bone to a two-piece implant inserted to bone level is about 2.0 mm. One related factor is the inflammatory infiltrate located at this junction,26 which, when absent, is not an issue without concomitant bone loss.27 The esthetic concerns of some clinicians, when considering a tissue-level implant in the anterior portion of the dentition, are that exposure of the restorative margins may be more likely. This is especially relevant in the patients with a thin, scalloped periodontal biotype. Bone-level implants were designed to circumvent these concerns. One modification of this implant, in comparison to traditional bone-level implants, is a horizontal offset of this microgap, or junction. In a recent animal study, Jung et al28 investigated the effects of this horizontal offset on crestal bone. This study showed the first bone-to-implant contact exists 0.26 mm to 0.44 mm apical to the implant shoulder after 6 months of functional loading. This is a significant difference compared with what clinicians have accepted as predictable—1.5 mm to 2.0 mm of crestal remodeling around traditional bone-level implants.
Purpose
This study evaluated several parameters. First, there was an implant survival of consecutively placed immediate implants that received immediately delivered provisional restorations without occlusal contact. Second, marginal bone levels on radiographic evaluation after delivery of the final loaded prosthesis were recorded and compared to those at time of implant insertion. Long-term follow-up of all parameters will be discussed in future research.
Methods and Materials
This is a retrospective study in which 23 consecutively treated patients received 30 bone-level implants to support either a single-unit or short-span immediately temporized restoration in the esthetic zone. A total of 19 single crowns and five splinted restorations were delivered at the time of implant placement.
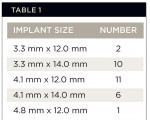
This zone was identified as any partially edentulous span from the first bicuspid to first bicuspid in the maxillary or mandibular arch. The distribution of implant locations is demonstrated in Figure 1 and Figure 2. The implant diameters were either 3.3 mm or 4.1 mm, with the exception of one 4.8-mm wide implant. The implant lengths were either 12.0 mm or 14.0 mm, depending on the anatomic requirement necessary for these fixtures to achieve primary stability at placement. Implant dimensions are seen in Table 1.
All implant sites were bordered by natural teeth on their mesial and distal aspects, with the exception of multiple implant placements where implants were adjacent to pontic sites or adjacent fixtures. All implants were screw-shaped threaded implants with a modified titanium surface (SLActive) and designed to be inserted at the bone crest (Straumann® bone level implants, Straumann) with an internal prosthetic connection (regular cross-fit or narrow cross-fit). Minimum insertion torque was 40 ncm. All prostheses, either single-crown or short-span bridge, were screw-retained. No restorations were cement-retained. Early in the study, final abutments were used to directly veneer tooth-colored material (bis-acryl or composite resin) to create one-piece restorations. When temporary abutments became available, they were modified to create mechanical retention of the same restorative material for direct veneering and fabrication of one-piece, screw-retained restorations.
Following fixation of the provisional restorations, the residual extraction socket space or horizontal defect distance (HDD) and labial plates of bone, regardless of the presence of fenestrations and/or dehiscences, were grafted with particulate bone replacement grafts. This graft consisted of either mineralized freeze-dried bone allograft (FDBA) or a biphasic calcium phosphate graft (BoneCeramic, Straumann). The distribution of graft material for each case is listed in Table 2. A resorbable collagen membrane (RCM) was always adapted over the graft and secured around the provisional restoration. Membrane composition is viewed in Table 3. Primary coverage of the barrier was always achieved with a coronally advanced flap, to compensate for minimal postoperative tissue shrinkage.
All provisional restorations were relieved of occlusal contacts in centric occlusion and excursive movements. These restorations remained in situ for at least 6 to 12 weeks prior to any modifications or commencement of definitive restorative therapy. Definitive restorations consisted of cement-retained crowns or bridges, retained by either custom or premachined abutments, torqued to 35 ncm. All final restorations were in full occlusal contact.
Case 1
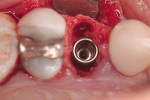
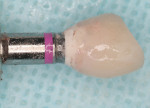
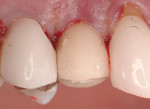
A 69-year-old male presented with loose crowns on teeth Nos. 7 to 9 (Figure 3). The restorative dentist determined these teeth could not be restored due to secondary caries within the prepared post channels (Figure 4 and Figure 5). The patient was unwilling to wear a removable, provisional removable partial denture (RPD) during osseointegration. Occlusal analysis revealed adequate protection for an immediately temporized splinted restoration, free of occlusal contacts in all excursive movements. The teeth were carefully extracted (Figure 2), following flap reflection, preserving the thin labial plate of bone. With the aid of a surgical guide, two 4.1-mm x 12.0-mm bone-level implants with a modified SLA surface (SLActive) (Figure 6). The insertion torque of both implants was 50 ncm. A composite graft consisting of FDBA (LifeNet Health, https://www.accesslifenethealth.org), calcium sulfate (Lifecore Biomedical, LLC, https://www.lifecore.com), and approximately 5% tetracycline HCl was used to obdurate all residual extraction sockets and over the facial bone to a level of about 2.0-mm thickness to compensate for loss of the bundle bone postextraction (Figure 7). A screw-retained provisional fixed partial denture (FPD) was fabricated on modified abutments with bis-acryl and flowable composite resin materials (Figure 8). The restoration, free of all occlusal contacts, was tightened to 15 ncm. A resorbable collagen membrane (RCM) (Cytoplast®, Osteogenics Biomedical, https://www.osteogenics.com) was secured around the provisional restoration (Figure 9). The facial flap was coronally repositioned with resorbable sutures (Figure 10 and Figure 11). The patient was advised to avoid any mastication with the anterior dentition and maintain a soft diet for at least 1 month. Postoperative medications included antibiotic therapy, amoxicillin 500 mg three times daily for 10 days, low-dose methylprednisolone (Medrol Dosepak, Pfizer, https://www.pfizer.com), non-steroidal anti-inflammatory medication for 400 mg for 3 days (Lodine, Wyeth Pharmaceuticals, https://www.wyeth.com), and chlorhexidine gluconate b.i.d. for 2 weeks.
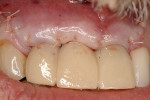
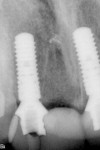
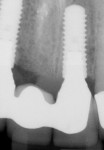
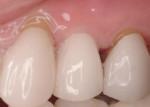
At 12 weeks following surgery, the provisional bridge was removed for the first time and fixture-level impressions were taken. Two computer-aided design/computer-aided manufacturing (CAD/CAM) zirconia abutments and a CAD/CAM zirconia three-unit bridge (Etkon™, Straumann), along with a zirconia crown on tooth No. 10 were fabricated (Figure 11, 12, 13).
Case 2
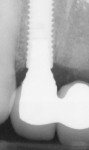
A 43-year-old female was enrolled in a periodontal maintenance program in which she alternated quarterly between her general dentist and periodontist for hygiene. She presented on an emergency basis with a fracture maxillary first bicuspid (Figure 14). Having received previous implant therapy to replace a mandibular second molar, she was hesitant to be partially edentulous in an esthetically sensitive area. Immediate provisionalization was chosen. Following careful tooth extraction, a bone-level implant with a SLActive surface was placed at an insertion torque of 50 ncm (Figure 15). A screw-retained temporary crown was fabricated on a PEEK (polyether-ether-ketone) Plastic Temporary Abutments (BioHorizons®, https://www.biohorizons.com) (Figure 16). Augmentation of the residual extraction socket was accomplished with a biphasic, calcium phosphate (BoneCeramic) graft (Figure 17) and collagen GBR membrane (Cytoplast). The membrane was adapted around a screw-retained provisional restoration (Figure 18) out of occlusal contact (Figure 19, 20, Figure 21). After 12 weeks, the patient’s general dentist performed traditional restorative therapy. A cement-retained porcelain-fused-to-metal (PFM) crown was delivered onto a cast, custom abutment into full occlusal contact (Figure 22 and Figure 23).
Results
All 30 implants were osseointegrated. No adverse events, such as graft failure, infection, prolonged discomfort, or component failure during provisionalization, occurred. Depending on soft-tissue maturation, all implants were determined “ready for final impressions” between 6 and 12 weeks following surgery. All implants were restored with cement-retained restorations. All final abutment screws were tightened to 32 ncm, according to the manufacturer’s recommendation. There were no reports of implant mobility or discomfort on abutment screw tightening. Therefore, the implant survival rate at time of restoration was 100%.
Another parameter in this study was proximal bone levels at the period of final restoration compared to time of placement. Bone maintenance was considered present when radiographic evidence of bone at or above the bone level of the fixture was observed. This may hold clinical importance relative to the presence or absence of the interproximal papilla, position of facial marginal mucosa, and subsequent esthetic outcome. The placement all 30 implants were in what the author classifies as the “esthetic zone;” this of primary importance to the success of implant therapy. Of the 30 implants, five were noted as having radiographic bone level apical to the coronal bevel of the fixture. Although none of the five implants demonstrated bone levels apical to the first thread, the “bone maintenance” criteria was not met according to the author’s definition. These five implants would be considered successful according to the original criteria of Albrektsson;2 the patients had no esthetic complaints. This would mean that 25 out of 30 bone-level implants did achieve “bone maintenance” at the time of restoration. Bone maintenance was achieved for approximately 83% of the implants in this study. These implants were placed into extraction sockets, devoid of intimate contact with marginal bone circumferentially. Both bone grafting and application of a resorbable membrane were used for not only providing circumferential bone for osseointegration but also soft-tissue maintenance. In a submerged healing environment, Gher et al18 demonstrated bone apposition with combined GBR and bone grafting compared with slight bone resorption when membrane therapy alone was used. In the present study, submerged healing was not possible. The immediate placement of a provisional restoration resulted in transmucosal or nonsubmerged healing. The use of combination therapy plus exploitation of the osteophilic implant surface and novel prosthetic attachment was employed with the expectation of crestal bone maintenance. This was evident in 83% of the cases.
The bone grafts (either mineralized bone allograft or Straumann BoneBeramic) were selected due to their osteoconductive properties and ability to maintain space under the membrane. Both grafts have been shown to be slowly replaced by host bone capable of osseointegration. The graft volume was determined by what was necessary to recreate ideal bony contours and support the esthetic soft-tissue frame. No predetermined volume was selected for the procedures. The graft volume was determined by the necessary amount required to achieve the desired bony contours. The bone graft was not responsible for any additional stability of the implants. This was achieved exclusively by the mechanical interlocking of the implant threads and initial bone–to-implant contact. The collagen barriers were selected based on their ability to provide cell-exclusionary properties (epithelial and fibroblast occlusivity) and preference to cells derived from the bony walls of the extraction sites. The membranes were adapted to cover the grafts and the peripheral 3 mm to 4 mm of the surrounding defect/socket walls.
The time from final restoration to evaluation was 3 months to 18 months. The average loading time was 9.1 months. Since final restoration, no biologic or mechanical complications or failures have been reported.
Discussion
The technique for simultaneous bone augmentation is well supported in the literature. When evaluating the stability of the facial and proximal mucosal margins in “early” placement situations in the esthetic zone, Buser et al29 demonstrated 12-month stability. His method, using the same implant design as this study, employed simultaneous bone grafting and membrane application. Although the graft materials and membrane composition varied, the technique and principles were similar. The thickness of the labial plate of bone following extraction is of primary concern. Augmentation of the bony plate at time of implant placement, with a graft of low substitution rate, preserves the thickness of the bone responsible for support of the peri-implant mucosa. In their review of the literature concerning immediate implant with immediate provisionalization, De Rouck et al30 recommended obturation of the void between the implant and buccal socket wall with a bone filler of low substitution rate. This step is recommended to counter the loss of bundle bone, viewed as inevitable following tooth extraction—the consequence of such bone loss directly related to marginal recession. Another variable that may contribute to the stability of the mucosal margin is the design of the implant-abutment connection. In a study by Buser et al,29 as in this one, the implants employed have a horizontal offset or platform switch. In the research by Buser et al, the implants showed a mean bone loss of 0.18 mm at 12 months. It is theorized, with increasing evidence supporting the concept, that the overall dimensions of the peri-implant biologic width remain stable However, by shifting the microgap from the bone crest, the vertical component of the biologic width may be minimized. The decrease in vertical bone remodeling results in less apical migration of the mucosal margin (ie, recession).
The present study evaluated the outcome of implants placed into extraction sockets at the time of tooth extraction or immediate placement. The technique of flapless surgery, especially when placing immediate implants, has gained significant popularity in recent years.31,32 The “minimally invasive” nature of these procedures make them attractive to patients and untrained dentists who wish to perform implant surgical procedures. This therapy does not allow for significant, if any, augmentation of the thin labial plate of bone following extraction. What may appear as highly esthetic in the short term may become problematic and more difficult to correct over time. The predictable loss of facial bone usually leads to marginal recession of the implant mucosa and esthetic compromise. Chen et al33 evaluated the stability of the marginal mucosa following flapless implant surgery. They evaluated 85 single-tooth implants placed in the maxillary central and lateral incisor positions. The authors reported a significant number of sites lost proximal and facial soft-tissue height and cautioned against implementation of this type of implant procedure in the esthetically sensitive areas. Many of the esthetic compromises associated with flapless surgery are related to the physiologic remodeling of the underlying bone. In their prospective study, Covani34 et al found less predictable bone augmentation around implants placed without flap elevation when compared with those augmented with conventional flap reflection. These implants were placed into extraction sockets with significant destruction of the facial bone. The amount of regeneration was significantly greater when combination therapy consisting of bone xenograft application and collagen membrane placement was performed with flap reflection. Both groups in this study achieved osseointegration and similar implant stability quotients.
Van de Velde et al35 demonstrated the high incidence of implant mal-positioning and damage to the bony walls that go undetected with flapless surgery. The researchers noted that not only was globally off-axis placement common, but there was a high incidence of perforation of the buccal and palatal osteotomy walls.
The long-term esthetic ramifications of the damage to the osseous recipient bed are speculated to be significant. In the present study, the simultaneous augmentation of the hard tissues is performed to not only give the surgeon the opportunity to visualize any osseous dehiscences and/or fenestrations but to also counter the predictable loss of bundle bone following tooth removal. The defects are certainly easier to treat earlier rather than after completion of restorative therapy, which could be catastrophic to the esthetic outcomes of these cases.
This study evaluated the success of the procedure in terms of confirming osseointegration and whether the implants will be acceptable in a timely manner for final restorative therapy (an estimated 3 months) without any adverse events, such as implant failure, component complications, and esthetic disasters. This is comparable with standard, transmucosal healing. The author intends to revisit these 30 implants in 3 years and 5 years to provide follow-up data on implant survival, success, bone levels, and esthetic satisfaction.
Conclusions
This study demonstrated that immediate implant placement and immediate nonloaded restoration in the esthetic zone is a predictable treatment if selection criteria are respected. All sites receiving immediate implants were augmented with a bone replacement graft and resorbable membrane. All provisional restorations were screw-retained and free of occlusal contact. Proximal bone levels coronal to traditionally accepted levels were achieved 83% of the time, and implant survival was 100%. All patients completed treatment and achieved esthetically satisfactory and functional results without unforeseen delays. At approximately 9 months of loading, no complications or failures were noted. Immediate placement and immediate provisionalization can be considered a predictable therapy.
References
1. Brånemark PI. Osseointegration and its experimental background. J Prosthet Dent. 1983;50(3):399-410.
2. Albrektssson T, Brånemark PI, Hansson HA, et al. Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop Scand. 1981;52(2):155-170.
3. Tarnow DP, Emtiaz S, Classi A. Immediate loading of threaded implants at stage 1 surgery in edentulous arches: ten consecutive case reports with 1-5 year data. Int J Oral Maxillofac Implants. 1997;12:319-324.
4. Van de Velde T, Collaert B, De Bruyn H. Immediate loading in the completely edentulous mandible: technical procedure and clinical results up to 3 years of functional loading. Clin Oral Implants Res. 2007;18(3):295-303.
5. Gapski R, Wang HL, Mascarenhas P, et al. Critical review of immediate loading. Clin Oral Implants Res. 2003;14(5):515-527.
6. Drago CJ, Lazzara RJ. Immediate occlusal loading of Osseotite implants in mandibular edentulous patients: a prospective observational report with 18-month data. J Prosthodont. 2006;15(3):187-194.
7. Neugebauer J, Traini T, Thams U, et al. Peri-implant bone organization under immediate loading state. Circularly polarized light analysis: a minipig study. J Periodontol. 2006;77(2):152-160.
8. Barone A, Rispoli L, Vozza I, et al. Immediate restoration of single implants placed immediately after tooth extraction. J Periodontol. 2006;77(11):1914-1920.
9. Degidi M, Piattelli A, Gehrke P, et al. Five-year outcome of 111 immediate nonfunctional single restorations. J Oral Implantol. 2006;32(6):277-285.
10. Schulte W, Heimke G. The Tübingen immediate implant. Die Quintessenz. 1976;27:17-23.
11. Schulte W, Kleineikenscheidt H, Linder K, et al. The Tübingen immediate implant in clinical studies. Dtsch Zahnarztl Z. 1978;33(5):348-359.
12. Araújo MG, Sukekava F, Wennström JL, et al. Ridge alterations following implant placement in fresh extraction socket: an experimental study in the dog. J Clin Periodontol. 2005;32(6):645-652.
13. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
14. Schwarz F, Herten M, Ferrari D, et al. Guided bone regeneration at dehiscence-type defect using biphasic hydroxyapatite + beta tricalcium phosphate (Bone Ceramic) or a collagen-coated natural bone mineral (BioOss Collagen): an immunohistochemical study in dogs. Int J Oral Maxillofac Surg. 2007;36(12):1198-1206.
15. Moses O, Pitaru S, Artzi Z, et al. Healing of dehiscence-type defects in implants placed together with different barrier membranes: a comparative clinical study. Clin Oral Implants Res. 2005;16(2):210-219.
16. Gelb DA. Immediate implant surgery: three-year retrospective evaluation of 50 consecutive cases. Int J Maxillofac Implants 1993;8(4):388-399.
17. Shanaman RH. The use of guided tissue regeneration to facilitate ideal prosthetic placement of implants. Int J Periodontics Restorative Dent. 1992;12(4):257-265.
18. Gher ME, Quintero G, Assad D, et al. Bone grafting and guided bone regeneration for immediate dental implants in humans. J Periodontol. 1994;65(9):881-891.
19. Cammack GV 2nd, Nevins M, Clem DS 3rd, et al. Histologic evaluation of mineralized and demineralized freeze-dried bone allograft for ridge and sinus augmentations. Int J Periodontics Restorative Dent. 2005;25(3):231-237.
20. Levin BP. Single stage implant placement and simultaneous bone augmentation. Implant Realities. 2008;1:24-27.
21. Buser D, Broggini N, Wieland M, et al. Enhanced bone apposition to a chemically modified SLA titanium surface. J Dent Res. 2004;83(7):529-533.
22. Schwarz F, Herten M, Sager M, et al. Bone regeneration in dehiscence-type defects at chemically modified SLActive) and conventional SLA titanium implants: a pilot study in dogs. J Clin Periodontol. 2007;34(1):78-86.
23. Albrektsson T, Zarb G, Worthington P, et al. The long-term efficacy of currently used dental implants: a review and proposed criteria of success. Int J Oral Maxillofac Implants. 1986;1(1):11-25.
24. Jemt T, Lekholm U. Single implants and buccal bone grafts in the anterior maxilla: measurements of buccal crestal contours in a 6-year prospective clinical study. Clin Implant Dental Relat Res. 2005;7(3):127-135.
25. Hermann JS, Cochran DL. Nummikoski PV, et al. Crestal bone changes around titanium implants. A radiographic evaluation of unloaded nonsubmerged and submerged implants in the canine mandible. J Periodontol. 1997;68(11):1117-1130.
26. Broggini N, McManus LM, Hermann JS, et al. Persistent acute inflammation at the implant-abutment interface. J Dent Res. 2003;82(3):232-237.
27. Broggini N, McManus LM, Hermann JS, et al. Peri-implant inflammation defined by the implant-abutment interface. J Dent Res. 2006;85(5):473-478.
28. Jung RE, Jones AA, Higginbottom FL, et al. The influence of non-matching implant and abutment diameters on radiographic crestal bone levels in dogs. J Periodontol. 2008;79(2):260-270.
29. Buser D, Halbritter S, Hart C, et al. Early implant placement with simultaneous guided bone regeneration following single-tooth extraction in the esthetic zone: 12-month results of a prospective study with 20 consecutive patients. J Periodontol. 2009;80(1):152-162.
30. De Rouck T, Collys K, Cosyn J. Single-tooth replacement in the anterior maxilla by means of immediate implantation and provisionalization: a review. Int J Oral Maxillofac Implants. 2008;23(5):897-904.
31. Becker W, Goldstein M, Becker BE, et al. Minimally invasive flapless implant surgery: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S21-S27.
32. Schwartz-Arad D, Chaushu G. Immediate implant placement: a procedure without incisions. J Periodontol. 1998;69(7):743-750.
33. Chen ST, Darby IB, Reynolds EC, et al. Immediate implant placement postextraction without flap elevation. J Periodontol. 2009;80(1):163-172.
34. Covani U, Cornellini R, Barone A. Buccal bone augmentation around immediate implants with and without flap elevation: a modified approach. Int J Oral Maxillofac Implants. 2008;23(5):841-846.
35. Van de Velde T, Glor F, De Bruyn H. A model study on flapless implant placement by clinicians with a different experience level in implant surgery. Clin Oral Implants Res. 2008;19(1):66-72.
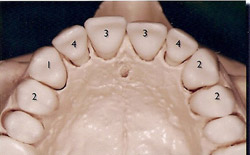 Figure 1 Implant distribution in maxilla. | 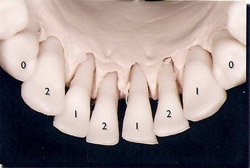 Figure 2 Implant distribution in mandible. | ||||||
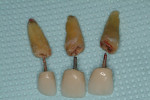
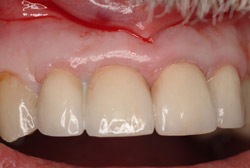 Figure 3 Preoperative situation. | 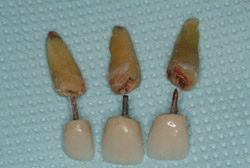 Figure 4 Extracted teeth. | ||||||
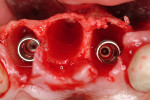
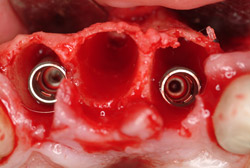 Figure 5 Implant placement. | 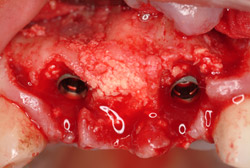 Figure 6 Bone allograft. | ||||||
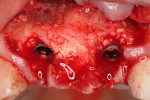
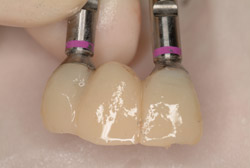 Figure 7 Screw-retained provisional bridge. | 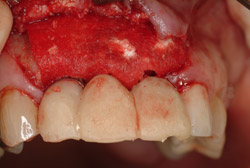 Figure 8 Collagen GBR. | ||||||
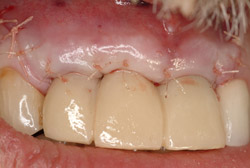 Figure 9 Flap adaptation. | 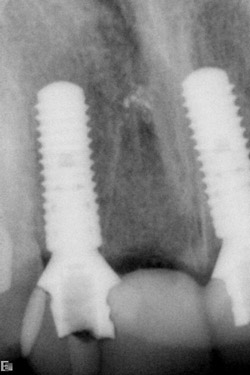 Figure 10 Postoperative PA. | ||||||
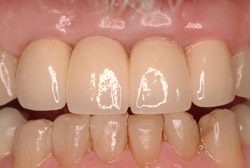 Figure 11 Completed case | |||||||
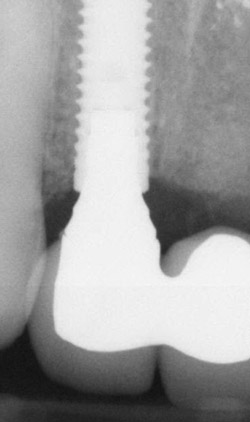 Figure 12 | 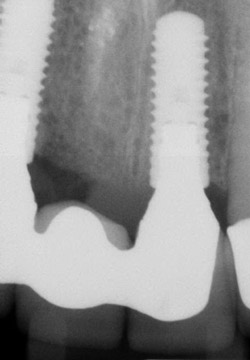 Figure 13 | ||||||
| Figure 12 and Figure 13 Final FPD PAs. | |||||||
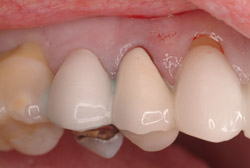 Figure 14 Preoperative situation. | 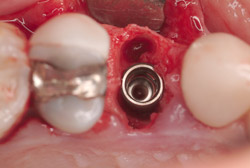 Figure 15 Immediate placement. | ||||||
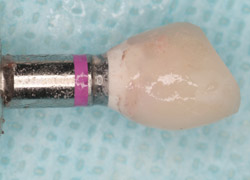 Figure 16 Screw-retained provisional crown. |  Figure 17 Straumann BoneCeramic | ||||||
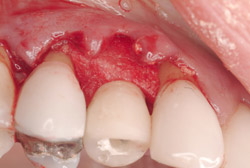 Figure 18 Collagen GBR. | 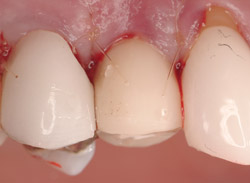 Figure 19 Flap adaptation. | ||||||
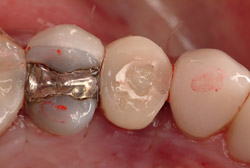 Figure 20 Provisional crown out of occlusal contact. | 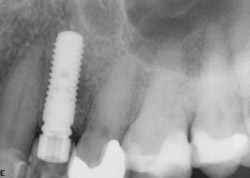 Figure 21 Postoperative PA. | ||||||
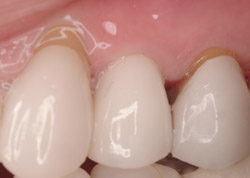 Figure 22 Final crown. | 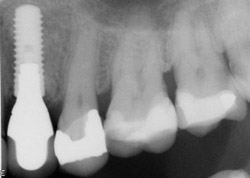 Figure 23 Completed case PA. | ||||||
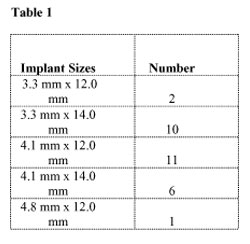 Table 1 | 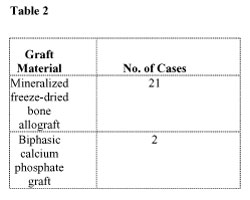 Table 2 | ||||||
 Table 3 | 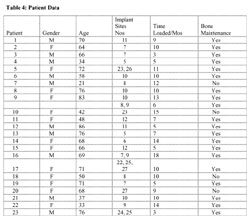 Table 4 | ||||||
| |||||||