Revisiting the Hopeless Ridge: Part II—Inductive Orthopedic Allograft Applied to Dental Implant Regeneration
John Lupovici, DDS
Abstract
Part I of “Revisiting the Hopeless Ridge” highlighted the higher complication rates, greater resorption profile, and lower implant success rates associated with autogenous block grafts. The conclusions described in that article were based on a comprehensive literature review, rather than an individual clinician’s experience in clinical practice. Additionally, the idea that such grafts were the gold standard for traditional dental implant-associated bone regeneration was challenged. This article explores the advantageous properties of new commercially available allograft bone in a variety of clinical applications. One such product that combines demineralized bone with lecithin is reviewed, and two case reports using it are presented.
Aloss of alveolar ridge dimensions from resorption in the wake of tooth loss, developmental anomalies, pathology, or trauma may make it difficult, if not impossible, to place implants in a position that will recreate natural function and meet the esthetic needs of patients. In addition to the negative functional and esthetic consequences associated with improper implant positioning, off-axis loads have been associated with biomechanical problems including fractures of the screw, implant, implant collar, prosthesis, and/or porcelain; screw loosening; and poor screw access-hole positioning.1,2
A variety of bone-augmentation strategies have been developed to enable clinicians to rebuild atrophic sites to support optimally positioned implants. Grafting techniques including ridge splitting and expansion,3 guided bone regeneration,4 distraction osteogenesis,5 onlay, inlay, and veneer grafts,6,7 nerve lateralization,8 and sinus augmentation9-11 have been used to treat extraction sockets, horizontally and vertically atrophic ridges, implant-associated defects, and problematic anatomic structures. Autograft, allograft, xenograft, and alloplast augmentation materials all have been used successfully.
The question of which bone-grafting material is optimal remains an open one, however. Ideally, any bone-grafting material should be:
- osteoconductive;
- osteoinductive;
- biocompatible;
- easily shaped to the desired form;
- space-maintaining;
- stable after positioning; and
- easy to use clinically.
Autogenous bone harvested from either extraoral or intraoral sites has been considered the gold standard among the alternatives. Depending on how the bone is obtained, autogenous bone may meet all the requirements listed above. At the same time, harvesting such bone has been associated with complications including morbidity, parasthesia, anesthesia, neurosensory changes to the proximal teeth and tissue, edema, ptosis, incision dehiscence, infection, and even mortality.7,12 High resorption rates for autogenous block grafts also have been reported,13-17 and the survival rates of implants placed in such grafts have fallen into question.18
Allografts
An alternative to autogenous block grafts is allograft material applied in combination with a barrier membrane.19,20 Used in periodontal therapy for more than three decades,21 allograft bone is available commercially and is offered in a variety of shapes and sizes. Its use frees the patient from the need for a second surgical site, with all the attendant discomfort and risks of complication.
Not every type of allograft bone meets all the requirements for an ideal bone-graft material. Freeze-dried bone allograft (FDBA) is biocompatible and osteoconductive; however, it lacks osteoinductivity. Although no significant clinical differences have been found between FDBA and demineralized freeze-dried bone allograft (DFDBA) in primarily intraosseous defects,22-24 DFDBA historically has been considered to be a better choice in sites where regeneration may be more problematic.
Bathing allograft bone in an acidic solution that exposes the bone-morphogenetic proteins theoretically renders it osteoinductive. DFDBA is also osteoconductive and biocompatible. However, when used in dental applications in combination with guided tissue regeneration, clinical results of DFDBA grafts have been widely variable.25-27 Similar variability has been found in attempts to induce new bone formation in animals.28
A number of factors may explain this variation. Although most bone banks follow the American Association of Tissue Banks’ guidelines for procuring, processing, and sterilizing cadaver bone, significant variation nonetheless exists in the way different tissue banks prepare DFDBA. Many banks do not sterilize the bone, but some use ethylene oxide for sterilization, even though this technique has been shown to decrease effectiveness and resorption of the allograft.29 Other sterilization techniques that use high temperatures have been associated with protein denaturation. Some tissue banks irradiate DFDBA, even though one study found that this practice reduced bone induction ability by 40%.30 Particle size also appears to be an important factor in the success of DFDBA at inducing bone formation, with particles in the 125-µm to 1000-µm range possessing a higher osteogenic potential than particles below 125 µm.31
Differences in how commercial bone banks prepare DFDBA do not fully explain the variation in DFDBA osteoinductivity. Clinical variability also has been found among allograft from the same tissue bank.32 With allografts, factors such as donor age33 and other natural variability among humans may also play a role.
Variation also exists in the way allograft materials are delivered, and this can affect their inductive capacity. When Han et al34 compared the osteoinductivity of delipidated demineralized bone to that of demineralized bone mixed with phosphatidylcholine (lecithin), they confirmed that removing lipids from demineralized bone significantly inhibited osteoinduction, but adding purified lecithin appeared to restore the osteoinductive activity and enhance biologic activity above that of a standard demineralized bone preparation. Lecithin is a phospholipid naturally present in cell membranes as well as in many other tissues and organs. It has been found in significant amounts during active bone formation and is believed to play an active role in the biologic calcification that occurs during osteogenesis.35
An established orthopedic graft material recently has been adapted for oral and maxillofacial grafting applications (RegenerOss® Allograft Putty, BIOMET 3i, Palm Beach Gardens, FL). Available in prefilled .5-cc, 1-cc, and 2-cc syringes, the material is a combination of DFDBA suspended in lecithin isolated from soybeans. Because no residual soy proteins have been associated with it, there are no concerns about an immunologic response. Allograft putties that use glycerol as a binding agent pose a concern because of demonstrated levels of toxicity.36 In contrast, lecithin is a phosphatidylcholine, an organic lipid naturally resident in cell membranes.
When incubated at 37° C, RegenerOss Allograft Putty maintains a solid state and remains intact at the graft site for 7 to 14 days, after which it is absorbed by the body with no foreign-body reaction. The consistency of the putty, which is composed of 40% demineralized bone by weight, makes it easy to mix with autogenous or other additional bone-grafting material, should that option be desired. It provides remarkable ease of handling and excellent graft containment after being exuded into the defect site. Because the lecithin carrier is a lipid, it is hydrophobic, offering high resistance to irrigation, blood, and saliva. The demineralized bone used in RegenerOss Allograft Putty is screened thoroughly for HIV, hepatitis, and other infectious agents.
Of great importance, the inductivity of the bone is routinely verified with the SaOS-2 cell-proliferation test. This in vitro laboratory bioassay measures the proliferation of SaOS-2 human osteosarcoma cells in response to incubation with demineralized bone matrix and compares them with positive and negative controls. The results of these assays have demonstrated a correlation coefficient of 0.85 (P < .0005) with implantation of demineralized bone into athymic rat muscle, the previous gold standard for measuring osteoinduction.37
The following clinical cases illustrate the use of RegenerOss Allograft Putty in conjunction with cross-linked resorbable membranes.
Case Reports
Case 1
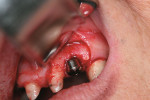
A 48-year-old man presented after experiencing multiple apical abscesses that had recurred during a 3-year period. Intraoral examination indicated the presence of a large apical fistulous tract at the left central incisor (Figure 1). Radiographic examination revealed a post fracture (Figure 2).
The patient was given 1 g of amoxicillin prophylactically before surgery, to be followed by 500 mg three times daily for 7 days. The tooth was extracted atraumatically, yielding evidence of loss of the buccal plate. Subsequent reflection of a full-thickness flap confirmed a loss of the facial plate all the way to the apex (Figure 3). The residual granulomatous tissue was thoroughly debrided from the socket, and approximately 1-cc of RegenerOss Allograft Putty was extruded and adapted to the defect. A 20-mm x 30-mm OsseoGuard® Resorbable Collagen Membrane (BIOMET 3i) was positioned over the graft material. Extensive mesial, distovertical, and periosteal vertical releasing incisions were created in the periosteum, and passive primary closure was obtained. The patient was sent home with a removable transitional partial that provided passive contact with the ridge.
Five-months postsurgery, radiographic examination revealed moderate mineralization with unremarkable soft-tissue healing. A papilla-preserving full-thickness flap was reflected, and a 2-mm trephine was used to extract an 8-mm bone core from the site where an implant was planned for placement. The core was immediately stored in formalin and later sent for histologic review.
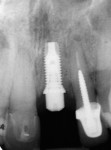
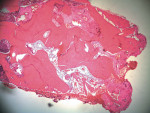
The implant osteotomy was enlarged sequentially under copious irrigation, and a 4/5/4 x 11.5-mm NanoTite™ PREVAIL® Implant (BIOMET 3i) was placed (Figure 4 and Figure 5). The implant successfully osseointegrated and was restored. The results of the histologic examination confirmed new bone formation. The histologic sections (Figure 6) displayed new woven bone tissue in conjunction with the presence of osteoid and osteoblasts. Active remodeling also was evidenced by the encapsulation of the allograft amidst the patient’s bone, with thick reversal lines (Figure 7).
Case 2
The patient was a 64-year-old woman who presented in the wake of a vertical post fracture of her maxillary left lateral incisor. Her only significant medical history was hypertension, controlled through diet.
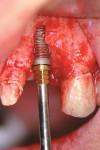
The same antibiotic regime described for Case 1 was administered, and an intrasulcular incision was made with distal release, extending from the left central incisor to the left cuspid. Reflection of a full-thickness flap revealed a complete loss of facial plate. The incisor was extracted atraumatically, preserving the residual alveolar housing. A 4-mm x 13-mm NanoTite™ Certain® Implant (BIOMET 3i) was immediately placed within the residual ridge. Primary stability was achieved, but 13 threads remained exposed on the facial aspect (Figure 8).
RegenerOss Allograft Putty was adapted to the ridge to achieve full coverage of the exposed threads (Figure 9), and an OsseoGuard Membrane was trimmed to fit the contours of the graft site and placed in position. Passive primary closure was obtained through aggressive periosteal release.
Primary closure was maintained through the healing period. Five months after the regeneration and implant placement procedure, a radiograph indicated successful integration (Figure 10). At implant uncovering and healing abutment connection, the site was re-entered, revealing extensive hard-tissue regeneration. The previously exposed threads were completely encased in newly regenerated bone (Figure 11).
Discussion
Osteoinduction is the process by which new bone is formed via the stimulation of osteogenic precursor cells that differentiate and form new bone. Osteoinductive materials, by definition, induce undifferentiated mesenchymal cells to transform into osteoblasts or chondroblasts.38 Historically, the osteoinductive properties of commercially available allograft have been highly variable. Some allografts have been confirmed to lack any inductivity at all. In contrast, the routine verification of the osteoinductivity of RegenerOss Allograft Putty makes it a highly reliable bone-augmentation material.
The ideal application for this material is within self-contained defects, where space-making concerns are not of paramount importance. When attempting to regenerate bone outside the alveolar housing, the author suggests the addition of osteoconductive material to ensure that the desired contours are maintained. Similarly, when using the putty in an extraction socket, the clinician should attempt to achieve primary closure to prevent patient tongue habits from displacing the graft material.
Although excellent volumetric fill and vascularity have been evident upon re-entry of sites grafted with RegenerOss Allograft Putty, the density of the bone has often been judged type 2 to 3 (according to the Lekholm and Zarb scale). The nature of demineralized allograft material may explain the seeming disparity between the excellent histologic results and the apparent soft nature of the new bone. Research by Cammack et al39 found significantly less residual bone in sites augmented with DFDBA, compared with those augmented with the mineralized variety, even though no statistical difference in percentages of new bone formed was found at sites grafted with each material. This suggests that the mineralized material may take longer to resorb, creating the appearance of harder bone at sites grafted with it. Yet, the differences between the two materials appeared to have no bearing on the success or failure of the graft or implant.
Conclusion
DFDBA combined with a lecithin carrier and routinely verified for osteoinductivity provides an excellent alternative to autogenous bone in oral sites where bone augmentation is necessary to ensure ideal implant restorative outcomes. This nontoxic material is commercially available, avoids the morbidity associated with the harvest of autograft material, has excellent handling characteristics, and appears to enhance bone formation, as compared with other allograft bone products. Histologic examination of sites grafted with this material has confirmed the development of well-vascularized bone marrow and newly formed bone. Further investigation is needed to yield additional insight into the osseoinductive capacity achieved when this material is combined with other graft materials.
Disclosure
The author is a consultant for BIOMET 3i.
References
1. Rangert B, Jemt T, Jörneus L. Forces and moments on Brånemark implants. Int J Oral Maxillofac Implants. 1989;4(3):241-247.
2. Khraisat A, Abu-Hammad O, Dar-Odeh N, et al. Abutment screw loosening and bending resistance of external hexagon implant system after lateral cyclic loading. Clin Implant Dent Relat Res. 2004;6(3):157-164.
3. Scipioni A, Bruschi G, Calesini G. The edentulous ridge expansion technique: a five-year study. Int J Periodontics Restorative Dent. 1994;14(5):451-459.
4. Mellonig JT, Nevins M. Guided bone regeneration of bone defects associated with implants: an evidence-based outcome assessment. Int J Periodontics Restorative Dent. 1995;15(2):168-185.
5. Urbani G, Lombardo G, Santi E, et al. Distraction osteogenesis to achieve mandibular vertical bone regeneration: a case report. Int J Periodontics Restorative Dent. 1999;19(4): 321-331.
6. Proussaefs P, Lozada J. The use of intraorally harvested autogenous block grafts for vertical alveolar ridge augmentation: a human study. Int J Periodontics Restorative Dent. 2005;25(4):351-363.
7. Misch CM. Comparison of intraoral donor sites for onlay grafting prior to implant placement. Int J Oral Maxillofac Implants. 1997;12(6):767-776.
8. Jensen J, Reiche-Fischel O, Sindet-Pederson S. Nerve transposition and implant placement in the atrophic posterior mandibular alveolar ridge. J Oral Maxillofac Surg. 1994;52(7): 662-668.
9. Boyne PJ, James RA. Grafting of the maxillary sinus floor with autogenous marrow and bone. J Oral Surg. 1980;38(8):613-616.
10. Tatum H Jr. Maxillary and sinus implant reconstructions. Dent Clin North Am. 1986;30(2): 207-229.
11. Lazzara RJ. The sinus elevation procedure in endosseous implant therapy. Curr Opin Periodontol. 1996;3:178-183.
12. Raghoebar GM, Louwerverse C, Kalk WW, et al. Morbidity of chin bone harvesting. Clin Oral Implants Res. 2001;12(5): 503-507.
13. Misch CM, Misch CE, Resnik RR, et al. Reconstruction of maxillary alveolar defects with mandibular symphysis grafts for dental implants: a preliminary procedural report. Int J Oral Maxillofac Implants. 1992;7(3): 360-366.
14. Raghoebar GM, Batenburg RH, Vissink A, et al. Augmentation of localized defects of the anterior maxillary ridge with autogenous bone before insertion of implants. J Oral Maxillofac Surg. 1996;54(10): 1180-1186.
15. Widmark G, Andersson B, Ivanoff CJ. Mandibular bone graft in the anterior maxilla for single-tooth implants. Presentation of surgical method. Int J Oral Maxillofac Surg. 1997;26(2): 106-109.
16. McAllister BS, Haghighat K. Bone augmentation techniques. J Periodontol. 2007;78(3):377-396.
17. Proussaefs P, Lozada J. The use of intraorally harvested autogenous block grafts for vertical alveolar ridge augmentation: a human study. Int J Periodontics Restorative Dent. 2005;25(4): 351-363.
18. Aghaloo TL, Moy PK. Which hard tissue augmentation techniques are the most successful in furnishing bony support for implant placement? [published erratum appears in: Int J Oral Maxillofac Implants. 2008;23(1):56] Int J Oral Maxillofac Implants. 2007;22(Suppl):49-70.
19. Mellonig JT, Nevins M. Guided bone regeneration of bone defects associated with implants: an evidence-based outcome assessment. Int J Periodontics Restorative Dent. 1995;15(2): 168-185.
20. Simion M, Jovanovic SA, Trisi P, et al. Vertical ridge augmentation around dental implants using a membrane technique and autogenous bone or allografts in humans. Int J Periodontics Restorative Dent. 1998;18(1):8-23.
21. Libin BM, Ward HL, Fishman L. Decalcified, lyophilized bone allografts for use in human periodontal defects. J Periodontol. 1975;46(1): 51-56.
22. Piattelli A, Scarano A, Corigliano M, et al. Comparison of bone regeneration with the use of mineralized and demineralized freeze-dried bone allografts: a histological and histochemical study in man. Biomaterials. 1996;17(11):1127-1131.
23. Rummelhart JM, Mellonig JT, Gray JL, et al. Comparison of freeze-dried bone allograft and demineralized freeze-dried bone allograft in human periodontal osseous defects. J Periodontol. 1989;60(12):655-663.
24. Francis J, Brunsvold M, Prewett A, et al. Clinical evaluation of an allogeneic bone matrix in the treatment of periodontal osseous defects. J Periodontol. 1995;66:1074-1079.
25. Becker W, Schenk R, Higuchi K, et al. Variation in bone regeneration adjacent to implants augmented with barrier membranes alone or with demineralized freeze-dried bone or auto-logous grafts: a study in dogs. Int J Oral Maxillofac Implants. 1995;10(2): 143-154.
26. Smukler H, Landi L, Setayesh R. Histomorphometric evaluation of extraction sockets and deficient alveolar ridges treated with allograft and barrier membrane: a pilot study. Int J Oral Maxillofac Implants. 1999;14(3): 407-416.
27. Parashis A, Andronikaki-Faldami A, Tsiklakis K. Comparison of 2 regenerative procedures—guided tissue regeneration and demineralized freeze-dried bone allograft—in the treatment of intrabony defects: a clinical and radiographic study. J Peridontol. 1998;69(7): 751-758.
28. Becker W, Lynch SE, Lekholm U, et al. A comparison of ePTFE membranes alone or in combination with platelet-derived growth factors and insulin-like growth factor-1 or demineralized freeze-dried bone in promoting bone formation around immediate extraction socket implants. J Periodontol. 1992;63(11):929-940.
29. Aspenberg P, Lindqvist SB. Ethene oxide and bone induction. Controversy remains. Acta Orthop Scand. 1998;69(2):173-176.
30. Zhang Q, Cornu O, Delloye C. Ethylene oxide does not extinguish the osteoinductive capacity of demineralized bone. A reappraisal in rats. Acta Orthop Scan. 1997;68(2):104-108.
31. Shapoff CA, Bowers GM, Levy B, et al. The effect of particle size on the osteogenic activity of composite grafts of allogeneic freeze-dried bone and autogenous marrow. J Periodontol. 1980;51(11): 625-630.
32. Schwarts Z, Mellonig JT, Carnes DL Jr, et al. Ability of commercial demineralized freeze-dried bone allograft to induce new bone formation. J Periodontol. 1996;67(9):918-926.
33. Schwartz Z, Sommers A, Mellonig JT, et al. Ability of commercial demineralized freeze-dried bone allograft to induce new bone formation is dependent on donor age but not gender. J Periodontol. 1996;69(4): 470-478.
34. Han B, Tang B, Nimni ME. Combined effects of phosphatidylcholine and demineralized bone matrix on bone induction. Connect Tissue Res. 2003;44(3-4):160-166.
35. Wuthier RE. Effect of phospholipids on the transformation of amorphous calcium phosphate to hydroxapatite in vitro. Calcif Tissue Res. 1975;19(3): 197-210.
36. Wang JC, Kanim LE, Nagkawa IS, et al. Dose-dependant toxicity of commercially available demineralized bone matrix material. Spine. 2001;26(13): 1429-1435.
37. Adkisson HD, Strauss-Schoenberger J, Gillis M, et al. Rapid quantitative bioassay of osteoinduction. J Orthop Res. 1999;18(3):503-511.
38. Misch CE, Dietsh F. Bone-grafting materials in implant dentistry. Implant Dent. 1993;2(3):158-167.
39. Cammack GV II, Nevins M, Clem DS III, et al. Histologic evaluation of mineralized and demineralized freeze-dried bone allograft for ridge and sinus augmentations. Int J Periodontics Restorative Dent. 2005;25(3):231-237.
About the Author
John Lupovici, DDS
Private Practice
New York, New York
Commack, New York