Recent Advances in Dental Adhesives - PART I
Mark A. Latta DMD, MS
ABSTRACT: The ability of dental professionals to bond resin materials to enamel and dentin has changed the concepts of cavity preparation, orthodontic treatment, caries prevention, and cementation of fixed prostheses. Today’s restorative treatment options, such as porcelain laminate veneers and direct resin bonding, rely on new adhesive materials and techniques. Improved marginal sealing around bonded restorations has reduced the frequency of unfavorable postoperative responses. This article will explain the involved biological structures, the mechanisms by which adhesion is achieved, and highlight the evolution of the adhesive materials utilized in such procedures.
Key Words: dental, adhesive, bonding, etch, monomer, hybrid
Dental adhesives are used for a wide range of clinical applications in restorative dentistry. Direct composite resin restorations all require bonding, and indirect resin inlays, onlays, and veneers require bonding and—depending on their design—crowns, bridges, and endodontic posts and cores may require and/or benefit from the use of dental adhesives such as BeautiBond in conjunction with resin luting materials. Bonding to tooth structure is essential for pit-and fissure sealants, some endodontic obturation materials, fixed orthodontic appliances, and dentinal hypersensitivity treatments. Consequently, the advancement of dental adhesion science has had a significant impact on modern clinical restorative dentistry.1
Buonocore is widely considered to be the first person to propose the application of adhesion technology in dentistry.2 His ideas were an extension of industrial techniques used to adhere paint and resin coatings to metal, and his groundbreaking research in 1955 demonstrated for the first time that acid-etching of enamel could provide a surface suitable for bonding with resins. Less widely known, however, is that Swiss chemist Oskar Hagger developed a material composed of glycerolphosphoric acid dimethacrylate in 1949 that was commercialized as Sevritron Cavity Seal.3 It was postulated by Kramer and McLean in 1952 that this material chemically bonded to tooth structure.4This, in reality, was the first “self-etching” adhesive material.
By the mid-1960s, the first commercially available pit-and-fissure sealants and composite resin materials utilizing this new adhesive technology were used clinically. Buonocore theorized that bonding was achieved via micromechanical retention due to resin tags filling the defects created by the etchant.5 By the late 1960s, Buonocore also proposed that bonding to dentin was possible.1 Since then, dental adhesives that provide numerically higher bond strengths and more substantive bonded interfaces to both enamel and dentin have been developed. By the 1980s, etch-and-rinse adhesives had gained widespread acceptability, and Nakabayashi in 1982 proposed a mechanism for bonding to dentin that described the infiltration of resin monomers into the tooth.6 By the 1990s, the concept of the “hybrid layer” was accepted, and both multi-step and single-step adhesives were available.7
Using dental adhesives can promote conservation of tooth structure and enable minimally invasive cavity preparation. For example, acid-etch retained “Maryland” bridges are based on the concept of minimal tooth preparation, by taking advantage of the adhesion of the “wings” of the bridge to abutment teeth. In intracoronal cavity preparations, direct composites resin restorations—unlike amalgam fillings—are believed to reinforce weakened dentin or enamel, reduce marginal staining, reduce microleakage, and may also reduce postoperative sensitivity when used appropriately.8
A classification system, primarily based on marketing considerations, has evolved for dental adhesive systems; it describes the specific adhesive as from a “generation”. In fact, the resin-based adhesive systems developed to date can be categorized as one of two types: 1) etch-and-rinse (total-etch) adhesive systems, available as three-step and two-step systems, and 2) “self-etch” adhesive systems, available as two-step and one-step systems.9 Regardless of the classification, however, all modern systems show some etching and demineralization of the tooth surface depending on the chemistry of the system selected. Both etch-and-rinse and self-etch adhesives produce a unique interface at the juncture of tooth surface and adhesive. Differences in the enamel bonding interface, as compared to the dentin bonding interface, is an important consideration in the selection of either an etch-and-rinse or a self-etching adhesive system. Every adhesive requires manipulation steps, and different techniques are required with each system to enable successful bonding, resulting in technique sensitivity for a given bonding procedure.
Etch-and-Rinse Adhesives
Etch-and-rinse adhesives are available as a three-step system (eg, OptiBond FL; Scotchbond Multi Purpose Plus; All Bond 3;) and two-step systems (eg, OptiBond Solo Plus; Adper Single Bond Plus; Prime&Bond NT; Excite). Etch-and-rinse adhesives produce high and durable adhesion values for both enamel and in the bond to dentin. The generation of consistent and high adhesion values to dentin with these systems, however, requires careful attention to detail in their clinical application. On the other hand, self-etch adhesives generally demonstrate better adhesion to dentin than to enamel. Deficiencies in enamel bonding can be particularly critical in anterior aesthetic restorations. If enamel is the dominant substrate to be bonded to, and particularly if the surface consists of intact enamel, such as with anterior diastemata, an unprepared veneer, or a minimally prepared adhesive bridge, an etch-and-rinse system is preferred.
Mechanisms of Action — Enamel and Dentin
The basic mechanism for enamel and dentin bonding using etch-and-rinse systems consists of the following steps: demineralization of the surface by the acid (ie, the etchant), penetration of the adhesive monomers into the microscopic spaces created by the etchant, and curing of the adhesive monomers to form resin tags that microscopically provide a mechanical bond and seal to dentin and enamel.10 In enamel, macro resin tags are created peripherally around the demineralized surface of the hydroxyapatite crystals, and microtags are formed by resin penetration into demineralized crypts within the crystals. On dentin, the etchant creates a demineralized surface 3 µm to 5 µm in depth and, upon rinsing the acid, the smear layer and other debris are removed from the surface. Failure to remove the smear layer reduces dentin permeability, as that layer acts as a barrier and prevents adhesion to the underlying intact tooth structure. Following acid conditioning, collagen contained in the dentin is exposed. The adhesive system monomers then envelope the exposed collagen and remaining mineral, penetrating to the residual intact mineralized dentin within this microscopic etched surface. This interface is referred to as the “hybrid layer”’ and is observed in three distinct areas: within the dentinal tubules, in the microscopic branches lateral to the tubules, and in the intertubular dentin.11
Three-step etch-and-rinse systems contain an etchant, a primer, and a bonding resin (ie, an adhesive). The primer is used to displace residual moisture, thus creating a surface to which the hydrophobic bonding resin can adhere. The hydrophilic solvent in the primer facilitates penetration of the resin monomers into the tooth surface. The subsequently applied bonding resin (ie, the adhesive) fills the residual space and seals the dentinal tubules. Two-step systems combine the primer and adhesive components, thus simplifying the application process into only one step. Some have suggested that the two-step systems may introduce greater variability into bonding results. There is, however, evidence of excellent clinical performance over the years. One key performance factor that influences the success of etch-and-rinse systems includes the thorough removal of the etchant prior to bonding.12 It is also critical to ensure that the dentin remains moist when the primer and adhesive (or primer-adhesive combination) are applied so that adequate space is retained between the exposed collagen to allow for penetration of the etched surface by the resin monomers. Restorations comprising greater dentin surface area relative to the enamel surface area are more demanding in regard to this consideration. Larger posterior cavities and full-coverage crown preparations with extensive dentin surface areas are particularly subject to postoperative sensitivity if the dentin is dried, the collagen collapses, and inadequate resin penetration is developed.9
There is proven long-term bond stability using etch-and-rinse systems with both enamel and dentin. Most studies have shown bonding to enamel is more consistent, with higher initial bond-strength values and less breakdown at the enamel margin over time than with self-etching adhesive systems.13
Self-etching Adhesives
Self-etch adhesives are available as one-step systems (eg, Xeno IV; Clearfil S3; Adper Easy Bond; G-Bond; BeautiBond) and two-step systems (eg, AdheSE; Adper SE Bond; Clearfil SE Bond). These systems do not require an etch-and-rinse procedure. There is no need to address residual dentin moisture with the clinical application of these systems. The demineralizing process occurs simultaneously with the penetration of the adhesive, thus eliminating the potential for over-etching when treating dentin.9
Mechanisms of Action — Enamel and Dentin
As with etch-and-rinse adhesive systems, the mechanism of adhesion is thought to be a combination of mechanical and/ or chemical retention for both enamel and dentin. A hybrid layer that mimics the hybrid later formed with etch-and-rinse adhesives is produced with these systems, and deep resin tag penetration can also be seen. Self-etching adhesive formulations all contain water to facilitate the acid reaction on the tooth surface, but a great range of pH is observed depending on the specific adhesive system. Mild pH self-etch adhesives have shallower resin tags, and the hydroxyapatite with which they interact is not completely demineralized. It is believed that retention may be partly due to a chemical interaction between the monomer and the hydroxyapatite crystal, especially with the less acidic systems.9
Self-etch adhesives are moisture forgiving, and thus they are particularly useful in difficult-to-isolate areas or with difficult patients. Self-etch adhesives, however, generally contain water in addition to monomers in their formulation. Water is more difficult (less volatile) to remove in the drying step after placement. Thus, it is more critical to ensure that the drying of the adhesive after application is effectively completed. In addition, the presence of water in the formulation requires special attention to shelf stability on the part of the manufacturers, due to the potential for hydrolysis of the formula components. Since self-etch adhesive systems demineralize and penetrate the dentin at the same time, there is no concern over incomplete penetration and residual gaps within demineralized dentin. Depending on the pH of the specific adhesive formula, the removal of the smear layer is often less complete, so the dentinal tubules may not be as open as in the case of the etch-and-rinse systems. This may reduce the risk of postoperative sensitivity, although the restorative technique is perhaps the key to reduced postoperative sensitivity.9
Newer Adhesive Developments
Most recent developments in adhesive materials have focused on the self-etching approach. One-bottle, self-etch systems have now evolved to where only one material is applied to the tooth, with no mixing required. Newer self-etching systems have developed compositions that have led to improvements in the products’ shelf lives, with diminished hydrolysis in the package. As noted above, while the quality of dentin adhesion with these self-etch systems is comparable to that of the etch-and-rinse adhesives, the enamel bonding of these systems has fallen short of values for bond strengths achieved in the laboratory using a phosphoric acid etch system.13
Most self-etching adhesives contain some phosphoric acid ester derivative in a mixture of water and other solvents. This acid component drives the demineralization reaction on enamel and dentin. A higher pH of the system usually promoted dentin adhesion while a lower component promotes enamel penetration. A newly developed formulation from SHOFU Inc. called BeautiBond uses an elegant chemistry approach to maximize adhesion to both substrates. BeautiBond contains a carboxylic acid monomer to promote adhesion to dentin and also a phosphonic acid to promote adhesion to enamel (Figure 1). This unique approach has created a single-step, one-component adhesive system capable of generating a high and durable bond to dentin and enamel (Figures 2 and 3). In vitro testing of BeautiBond has exhibited high bond strengths to dentin and enamel even after extensive thermocycling (Figures 4 and 5).15,16 Scanning electron microscopy shows a durable hybrid layer formed to dentin that withstands thermomechanical stress as well as multi-step self-etching adhesive systems. Scanning electron microscopy and transmission electron microscopy of the hybrid layer interface reflects a complete and stable infiltration of resin into the tooth structure unlike other single component adhesive compositions (Figure 6, 7, 8).17
Summary
Dental adhesives are an essential element of clinical restorative dentistry. Numerous forms of adhesive systems are available and include etch-and-rinse systems (also known as “total-etch” systems) and self-etch systems. Both systems require the etching of the tooth surface, and mechanical and/or chemical retention of the restoration or device upon curing of the adhesive. The clinician’s selection of an adhesive has implications for the technique used as well as for the quality of the bond achieved. To date, enamel bonding is superior with etch-and-rinse adhesive systems, while self-etch systems may be more suitable for restorations with large areas of dentin. Newer formulas employing a combination of adhesive monomers, however, may facilitate a higher degree of performance for the self-etching approach. These newer developments, which have been introduced to simplify and improve the performance of adhesive systems for bonding of both enamel and dentin, may significantly advance the art and science of restorative dentistry.
References
1. Perdigao J, Lopes M. Dentin bonding state of the art 1999. Compend Contin Educ Dent 1999;20(12):1151-1162.
2. Buonocore MG. A simple method of increasing the adhesion of acrylic filling materials to enamel surfaces. J Dent Res 1955:34:549-553.
3. McLean JW. Historical Overview: The pioneers of enamel and dentin bonding. In: Roulet JF, DeGrange M, eds. Adhesion --The Silent Revolution in Dentistry. Carol Stream, IL: Quintessence Publishing, 2000;13-17.
4. Kramer IRH, McLean JW. The response of the human pulp to self-polymerising acrylic. Br Dent J 1952;93:150-153.
5. Buonocore MG, Matsui A, Gwinnett AJ. Penetration of resin dental materials into enamel surfaces with reference to bonding. Arch Oral Biol 1966;13(1):61-70.
6. Nakabayashi N, Kojima K, Masuhara E. The promotion of adhesion by the infiltration of monomers into tooth substrates. J Biomed Mater Res 1982;16:265-273.
7. Kanca J. Improving bond strength through acid etching of dentin and bonding to wet dentin surfaces. J Am Dent Assoc 1992;123(9);183-188.
8. Perdigao J, Geradeli S, Hodges JS. Total-etch versus self-etch adhesive: Effect on postoperative sensitivity. J Am Dent Assoc 2003;134(12):1621-1629.
9. Van Meerbeek B, De Munch J, Yoshida Y, et al. Buonocore Memorial Lecture. Adhesion to enamel and dentin: current status and future challenges. Oper Dent 2003;28:215-235.
10. Van Meerbeek B, Vargas MA Inoue S, et al. Adhesives and cements to promote preservation dentistry. Oper Dent 2001;Suppl6;119-143.
11. Van Meerbeek B, Inskoshi S, Braem M, et al. Morphological aspects of the resin-dentin interdiffusion zone observed with different adhesive systems. J Dent Res 1992;72(2):1530-1540.
12. Perdigao J, Geradeli S. Hodges JS. Total-etch versus self-etch adhesive: Effect on postoperative sensitivity. J Am Dent Assoc 2003;134(12):1621-1629.
13. Brackett WW, Ito S, Nishitani Y, et al. The microtensile bond strength of self-etching adhesives to ground enamel. Oper Dent 2006;31(3):332-327.
14. Soderholm KJ, Guelmann M, Bimstein E. Shear bond strength of one 4th generation and two 7th generation bonding agents when used by operators with different bonding experiences. J Adhes Dent 2005;7:57-64.
15. Kawamoto O, Nagano F, Yasumoto K, Sano H. Micro-tensile bond strength of one and two-step self-etching adhesives using a novel thermocycling method. Poster Presentation, 3rd IAD 2008;Special Issue:126, Abstract PO85.
16. Shinno K, Ichizawa K, Nakatsuka T, et al. Bonding ability of resin-bonding systems containing phosphoric acid adhesive monomer. J Dent Res 2008,Abstract 0394.
17. Uno S, Morigami M, Sugizaki J, Yamada T. SEM and TEM observation of the bonding interface created with an experimental one-bottle one-step resin bonding agent. J Adhes Dent 2008;26(1):30-35.
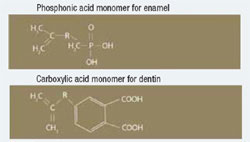 Figure 1 BeautiBond has two monomers: a phosphonic acid monomer for bonding to the enamel and a carboxylic acid monomer to facilitate dentin bonding. | 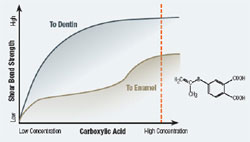 Figure 2 Graph of shear bond strength of BeautiBond’s carboxylic acid monomer to dentin and enamel. | ||||||
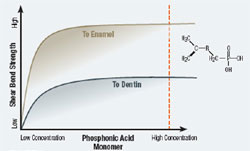 Figure 3 Graph of shear bond strength of BeautiBond’s phosphonic acid monomer to dentin and enamel. | 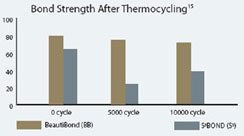 Figure 4 Graphic representation of the microtensile bond strength of BeautiBond and Clearfil S3 Bond to enamel upon thermocycling at baseline, 5,000 cycles, and 10,000 cycles. (Courtesy of Kawamoto C, Sano H, et al of Hokkaido University) | ||||||
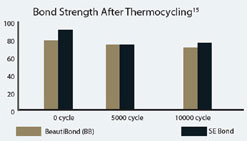 Figure 5 Graphic representation of the microtensile bond strength of BeautiBond and Clearfil SE Bond to dentin upon thermocycling at baseline, 5,000 cycles, and 10,000 cycles. (Courtesy of Kawamoto C, Sano H, et al of Hokkaido University) | 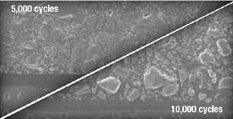 Figure 6 Scanning electron micrograph of BeautiBond at 5,000 cycles and 10,000 cycles. | ||||||
 Figure 7 Scanning electron micrograph of Clearfil S3 at 5,000 cycles and 10,000 cycles. (Courtesy of Kawamoto C, Sano H, et al of Hokkaido University) |  Figure 8 Scanning electron micrograph of Clearfil SE Bond | ||||||
| |||||||