NEWPORT BEACH, CA - Glidewell Laboratories has announced the start of a multiyear, longitudinal clinical study of BruxZir® NOW restorations. The study will be conducted by Technologies in Restoratives and Caries (TRAC) Research, the in-depth research arm of Gordon J. Christensen Clinicians Report® that is helmed by Rella P. Christensen, RDH, PhD.
For the purposes of the clinical study, all BruxZir NOW crowns will be fabricated and cemented at Glidewell Laboratories, with patients being recalled yearly over the next 5 years. At the time of each follow-up appointment, polyvinyl impressions will be taken for the purpose of scanning electron microscope study. In addition, clinical photographs will be captured, and in-house, clinical grading of each restoration will be performed. The first in a series of results is expected for publication in fall 2016.
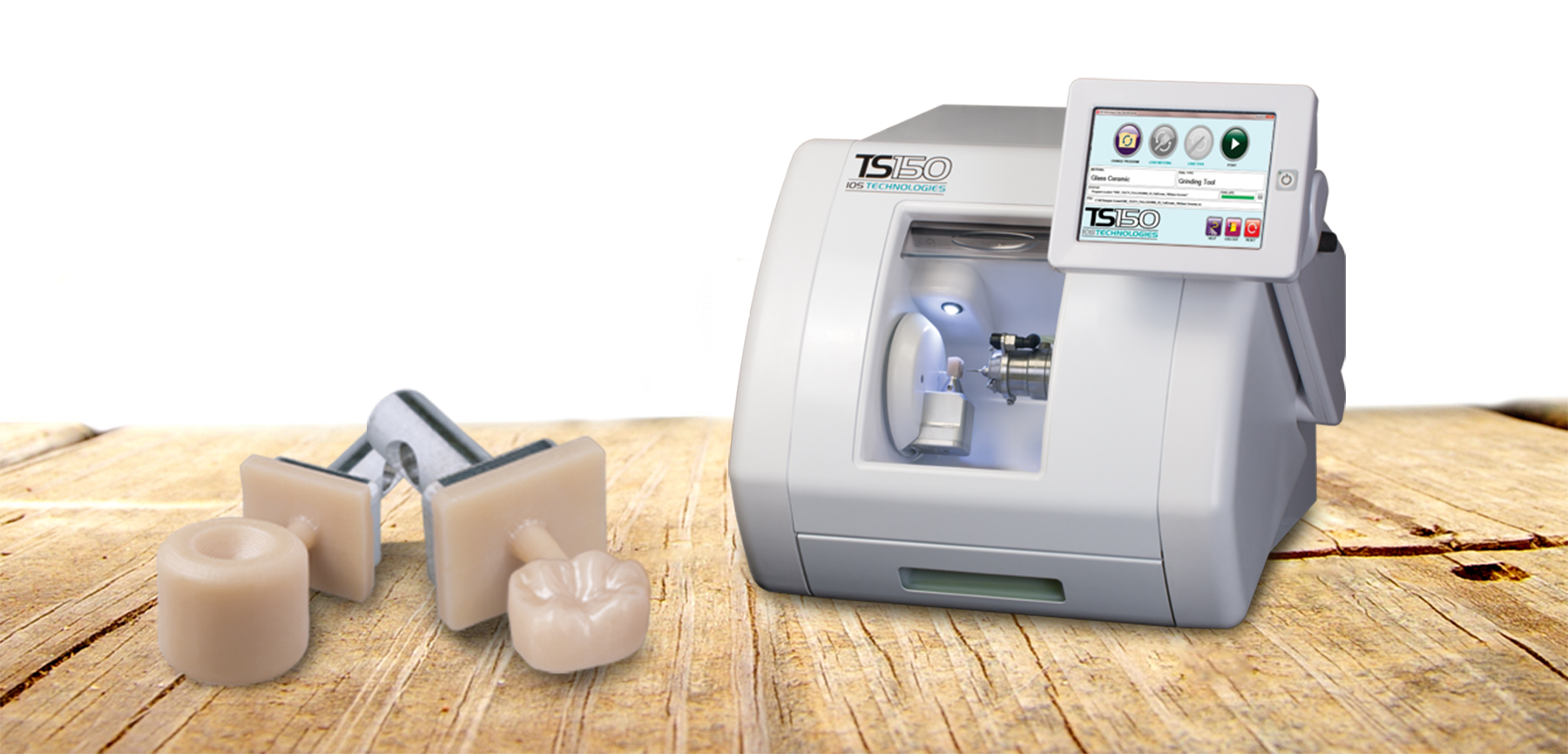
For the first time ever, clinicians can now mill and deliver high-strength, authentic BruxZir Solid Zirconia restoration in a single appointment. Through the use of BruxZir NOW milling blocks and the TS150™ Chairside Milling Solution, restorations can be designed and fabricated right in the dental office. Crowns created through this workflow exhibit flexural strengths of more than 800 MPa, without the need for sintering, staining, or glazing.
Glidewell Laboratories eagerly awaits the results from TRAC Research and CR Foundation®. For more information about BruxZir NOW milling blocks or to view completed studies on BruxZir Solid Zirconia, visit https://www.bruxzir.com.