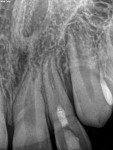
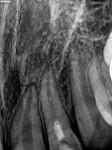
Regenerative Endodontics
Tissue engineering in clinical practice
James Baker, DMD | Brooke Blicher, DMD | Rebekah Lucier Pryles, DMD
Pulpal necrosis in immature permanent teeth poses unique challenges to the dental practitioner. Managing these pathologies effectively is essential to preventing premature tooth loss and the devastating physical and psychological consequences that can follow. The importance of retention is underscored by our inability to place implants in young patients until growth is complete.
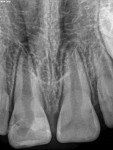
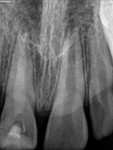
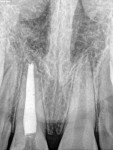
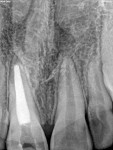
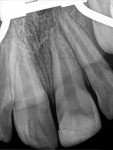
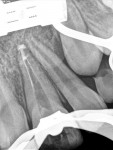
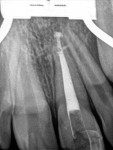
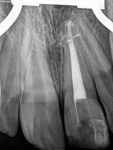
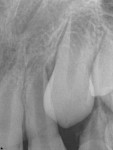
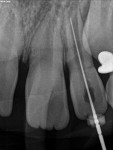
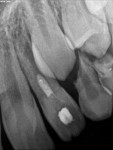
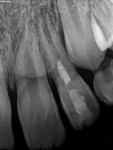
Unfortunately, traditional non-surgical root canal therapy (NSRCT) is an unfavorable treatment option for immature teeth because their thin dentinal walls prohibit further instrumentation and their open apices prohibit obturation with gutta percha. Consequently, endodontic treatment for these teeth histor-ically necessitated barrier creation at the apex to properly contain obturation material. This process is referred to as “apexification.” Barriers are created either with long-term intracanal calcium hydroxide1 (Figure 1 through Figure 4) or, more recently, with immediate apical placement of a mineral trioxide aggregate (MTA) plug2 (Figure 5 through Figure 8). Calcium hydroxide has fallen out of favor for use in apexification procedures due to the length of time required for treatment and its potential to weaken the dentinal walls.3 Although both techniques offer similar treatment predictability, MTA apexification is preferable due to shorter treatment times and decreased risk of fracture.4 Despite similar outcomes, the thin dentinal walls remaining after both of these apexification methodologies results in an uncertain long-term prognosis. To address these problems, significant clinical interest has emerged in the development of techniques to promote root maturation.
These techniques, or “regenerative endo-dontic procedures (REPs),” began with seminal case reports in the early 2000s that described the revascularization of necrotic, immature teeth.5,6 The protocols utilized included disinfection with antimicrobial agents, mechanical stimulation of intracanal bleeding, and radiographic monitoring for continued root development. Reported cases demonstrated resolution of apical pathology, increases in canal wall thickness, and continued apical closure. These initial findings served as the impetus to further study the factors involved in restoring the vitality of the pulp-dentin complex in immature, permanent teeth with pulpal necrosis.
REPs utilize principles of tissue engineering, including the need for stem cells, a scaffold, and signaling molecules to promote redevelopment of the pulp-dentin complex.7 Wounding the apical papilla stimulates the release of mesenchymal stem cells, and growth factors from dentin promote their migration into the pulp, differentiation, and proliferation. In the majority of REPs, fibrin networks in the blood clot and the dentinal walls serve as the biologically active scaffold to promote cell growth and differentiation, thereby permitting replacement by regenerative tissue. Additional efforts have been made to incorporate materials such as platelet rich plasma and platelet rich fibrin as adjunctive scaffolds. Although standardized clinical protocols are in development, room for improvement exists. The current recommendations and evidence-based rationale are summarized below.
Case Selection
Proper diagnosis and case selection is the foundation of endodontic therapy, including REPs. Currently, REPs are only indicated for immature necrotic teeth (Figure 9); however, some reported cases show potential applications for mature teeth.8 Additional treatment planning considerations include the proposed coronal restoration and patient’s ability to follow up. If a post is needed to retain the coronal restoration, apexification may prove to be a more favorable treatment choice. Furthermore, because close monitoring is necessary to ensure the success of REPs, any concerns regarding potential follow-up issues may also render apexification more predictable.9
Clinical Protocols
First Visit
Like in NSRCT, pulp space disinfection is essential to achieving positive outcomes in REPs. Unlike in NSRCT, instrumentation must be minimal to avoid damaging the thin dentinal walls (Figure 10). Furthermore, the irrigants and medicaments used for disinfection must also minimize toxicity to mesenchymal stem cells to maintain their pluripotency. Disinfectants utilized in REP include sodium hypochlorite (NaOCl), antibiotic pastes, and calcium hydroxide. These are often used in conjunction with ethylenediaminetetraacetic acid (EDTA).10 Chlorhexidine, which was used previously in REPs, is cytotoxic to mesenchymal stem cells and no longer recommended for this type of disinfection.11
Extensive research demonstrates that high concentrations of NaOCl are detrimental to stem cell proliferation and denature both crucial growth factors and cell signaling molecules. For this reason, NaOCl should be diluted to a concentration of 1.5% when used for REPs.12 Copious, gentle irrigation with 1.5% NaOCl is recommended for 5 minutes. Following irrigation, intracanal medicaments, such as calcium hydroxide or an antibiotic paste, can be used to further disinfect root canal spaces. Initial protocols utilized a triple antibiotic paste that contained ciprofloxacin, metronidazole, and minocycline. Concerns regarding minocycline-mediated dentinal staining and subsequent coronal staining resulted in the creation of a double antibiotic paste, which only contains ciprofloxacin and metronidazole. Initially, antibiotic pastes were employed at a higher concentration of 1 gm/mL; however, this high concentration was found to be detrimental to stem cell survival. Therefore, to minimize the impact on stem cell survival while maintaining antimicrobial efficacy, the concentration was reduced to 1 mg/ml.13 Unlike the antibiotic pastes, calcium hydroxide causes no detrimental effects on stem cell survival, regardless of the concentration used.13 Consequently, more modern REP protocols employ calcium hydroxide. Medicaments are left in the canal for 2 to 4 weeks, and reapplication should be considered at subsequent treatment visits if signs and symptoms remain.10
Subsequent Visit
Following proper disinfection, the stage is set for the completion of REP treatment. Anesthesia is achieved without a vasoconstrictor to maximize blood flow into the canal space. The canal space should be re-irrigated with saline, followed by a 17% EDTA solution to release critically important growth factors from the dentin. These growth factors potentiate stem cell migration, differentiation, and proliferation, leading to the eventual deposition of reparative tissue into the root canal space.14
Next, the canal space past the apical for-amen is over-instrumented in order to stimulate bleeding from the apical tissues. As previously discussed, the clot formed contains both mesenchymal stem cells and the fibrin scaffold necessary for regeneration. This cell introduction response is referred to as “homing” and involves the recruitment of stem cells into the root canal space, followed by their subsequent differentiation. Following overinstrumentation and blood clot formation or the placement of an alternative scaffold such as platelet rich plasma or platelet rich fibrin, the material should be controlled at a level that will allow for the addition of 3 to 4 mm of restorative material.10
In order to facilitate placement of a res-toration over the clot or scaffold, a resorbable matrix can be placed over the blood clot along with a capping material. MTA is generally used, however, alternative bioceramic ma-terials should be used if coronal discoloration is a concern. Depending on the clinical circumstances, the access opening should be sealed with a clinically acceptable restorative material, such as a resin-modified glass ionomer, composite resin, or amalgam (Figure 11).10
Clinical Outcomes
REPs are evaluated by their ability to eliminate signs and symptoms of infection, increase root wall thickness and initiate root end closure, and reproduce a positive response to pulp sensitivity testing (Figure 12 through Figure14). Although no long-term studies evaluating the functional survival of teeth following REPs currently exist, short-term studies indicate that infection is eliminated in 75% to 100% of cases. These success rates are comparable with those achieved using MTA apexification; however, reported results range considerably due to variation in the inclusion/exclusion criteria and clinical protocols as well as differing definitions of success. In addition to eliminating infection, the data suggests that REPs are better at increasing both root length and dentinal wall thickness when compared with MTA apexification. As with infection resolution, the amount of root development varies greatly from case to case. Approximately 60% of all published cases of REPs report return of positive pulp sensitivity following treatment.15 This suggests that not only has vascularized tissue returned to the dental pulp space, but also neuronal tissue. Because these neurons provide early detection of noxious stimuli and help to shield the pulp from injuries, their presence may enhance immune responses. Their return is a sign that the REP may have recreated an immunocompetent tissue capable of protecting the pulp from potential future injury.
Instead of resulting in “true regeneration,” histologic assessment reveals that current REPs provide more of a “guided endodontic repair,” producing mineralizing odontoblast-like cells, rather than true odontoblasts. Consequently, the tissue is not representative of a normal pulp-dentin complex.16 Although REPs cannot yet fully restore pulp to its native condition, from a clinical perspective, the published outcomes can be classified as successful beyond what is achievable with other treatment options.
Summary
Regenerative endodontics represents a shift from a materials-based treatment approach to a biologically-based means to manage and potentially reverse the progress of endodontic disease. Although current clinical protocols fail to achieve true regeneration of the pulp-dentin complex, they do stimulate the development of reparative tissue. Despite its limitations, this repair process resolves infection, potentiates root development, and often results in a return of pulp vitality to previously necrotic immature, permanent teeth. Not only does this minimize the risks associated with apexification, but it also provides acceptable outcomes for teeth with poor prognoses and few alternative treatment options. Continued advancements in translational science will hopefully lead to improved protocols with more predictable results and ultimately, the application of tissue engineering to truly regenerate the lost pulp-dentin complex in necrotic teeth.
References
1. Cvek M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha. A retrospective clinical study. Endod Dent Traumatol. 1992;8(2):45-55.
2. Mente J, Hage N, Pfefferle T, et al. Mineral trioxide aggregate apical plugs in teeth with open apical foramina: a retrospective analysis of treatment outcome. J Endod. 2009;35(10):1354-1358.
3. Andreasen JO, Farik B, Munksgaard EC. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent Traumatol. 2002;18
(3):134-137.
4. Lin JC, Lu JX, Zeng Q, Zhao W, Li WQ, Ling JQ. Comparison of mineral trioxide aggregate and calcium hydroxide for apexification of immature permanent teeth: A systematic review and meta-analysis. J Formos Med Assoc. 2016;115(7):523-530.
5. Iwaya SI, Ikawa M, Kubota M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol. 2001;17
(4):185-187.
6. Banchs F, Trope M. Revascularization of immature permanent teeth with apical periodontitis: new treatment protocol? J Endod. 2004;30(4):196-200.
7. Nakashima M, Akamine A. The application of tissue engineering to regeneration of pulp and dentin in endodontics. J Endod. 2005;31(10):711-718.
8. Saoud TM, Mistry S, Kahler B, Sigurdsson A, Lin LM. Regenerative endodontic procedures for traumatized teeth after horizontal root fracture, avulsion, and perforating root resorption. J Endod. 2016;42(10):1476-1482.
9. Law, AS. Considerations for regeneration procedures. J Endod. 2013;39(3 Suppl):S44-56.
10. American Association of Endodontists. Clinical Considerations for a Regenerative Procedure 6-8-16 page. https://www.aae.org/uploadedfiles/publications_and_research/research/currentregenerativeendodonticconsiderations.pdf. Updated June 8, 2016. Accessed September 8, 2017.
11. Trevino EG, Patwardhan AN, Henry MA, et al. Effect of irrigants on the survival of human stem cells of the apical papilla in a platelet-rich plasma scaffold in human root tips. J Endod. 2011;37(8):1109-1115.
12. Martin DE, Henry MA, Almeida JFA, et al. Concentration-dependent effect of sodium hypo-chlorite on stem cells of apical papilla survival and differentiation. J Endod. 2014;40(1):51-55.
13. Ruparel NB, Teixeira FB, Ferraz CC, et al. Direct effect of intracanal medicaments on survival of stem cells of the apical papilla. J Endod. 2012;38(10):
1372-1375.
14. Galler KM, D’Souza RN, Federlin M, et al. Dentin conditioning codetermines cell fate in regenerative endodontics. J Endod. 2011;37(11):1536-1541.
15. Diogenes A, Ruparel NB. Regenerative endodontic procedures: clinical outcomes. Dent Clin North Am. 2017;
61(1):111-125.
16. Simon SR, Tomson PL, Berdal A. Regenerative endodontics: regeneration or repair? J Endod. 2014;
40(4 Suppl):S70-75.
About the Authors
James Baker, DMD
General Practice Resident
Danbury Hospital
Danbury, CT
Brooke Blicher, DMD
Private Practice
White River Junction, Vermont
Assistant Clinical Professor
Department of Endodontics
Tufts University School of
Dental Medicine
Boston, Massachusetts
Clinical Instructor
Department of Restorative Dentistry and Biomaterials Science
Harvard School of Dental Medicine
Boston, Massachusetts
Rebekah Lucier
Pryles, DMD
Private Practice
White River Junction, Vermont
Assistant Clinical Professor
Department of Endodontics
Tufts University School of
Dental Medicine
Boston, Massachusetts