Clinical Implications of Peri-Implantitis and Peri-Mucositis in the Contemporary Dental Practice
A management protocol combining implant surface decontamination and direct bone re-apposition
The use of dental implants has become a common treatment modality in the management of partially and fully edentulous cases.1-3 Their long-term success rates have been reported to be very high; however, the implant process is not immune to complications that can arise from poor surgical or prosthetic management, material failure, and improper short-/long-term maintenance leading to peri-implant breakdown of both the hard and soft tissue surrounding the dental implant(s).4,5 The peri-implant breakdown of these tissues is characterized by an inflammatory reaction and presents in two forms: peri-mucositis and peri-implantitis.4,5
Differentiation and Incidence
Peri-mucositis has been described as a disease where the presence of inflammation is confined to the soft tissues, with no loss of the supporting bone around the dental implant.4-6 It is characterized by bleeding upon probing, suppuration, and clinical probing depths less than or equal to 4 mm.5 In contrast, peri-implantitis presents as a disease where the inflammatory process includes the soft tissues as well as bone loss around the implant.4-6
Both Fransson and colleagues and Roos-Jansaker and colleagues noted that peri-mucositis was present in 48% of implants followed to 14 years.7,8 Varying rates of prevalence of peri-implantitis have been reported; following the diagnosis criteria previously outlined with the addition of radiographic evidence of the supporting alveolar bone, the rate has been estimated to be between 11% and 47% of patients.9,10 Understanding that peri-mucositis and peri-implantitis may occur in almost 50% of cases in general and specialty practices poses a significant dilemma to the dental practitioner in how to manage these affected patients appropriately.
Disease Etiology
It is generally accepted that peri-mucositis is a precurser to peri-implantitis, much like gingivitis is a precursor to periodontitis. It has also been described that peri-implant diseases have been associated with Gram-negative anaerobic bacteria similar to those around natural teeth in patients with advanced chronic periodontitis.11
Once bacteria adhere to titanium surfaces, a biofilm forms that is integral to the formation of peri-implant diseases.6,12-14 Any success in the treatment of peri-implant diseases must include the elimination of the biofilm from the implant surface.15 This is most effective in the reversal of peri-mucositis cases. Peri-implantitis lesions, much like severe periodontitis lesions, are more complex infections. Overwhelming bacterial insult with a decreased host-immune response results in the significant bone loss that can be observed in peri-mucositis cases. As previously stated, Gram-negative anaerobes are abundant in peri-implantitis lesions,26 in addition to Staphylococcus aureus pathogens in the early peri-implant site.17,18 Other organisms present in 60% of peri-implantitis cases include Prevotella intermedia, Porphyromonas gingivalis, Aggregatibacter, Treponema denticola, Fusobacterium nucleatum, Prevotella nigrescens, and Peptosteptococcus micros.19,20 Other markers are consistent between severe periodontitis and peri-implantitis, such as interleukin (IL)-1, IL-6, IL-8, IL-12, alkaline phosphates, and elastase activity.21-23
With this in mind, the main goal in treating peri-implantitis cases is similar to that of peri-mucositis cases, namely removal of the biofilm present from the implant surfaces, in addition to sterilization and decontamination of the affected implant surface. Risk factors for peri-mucositis and peri-implantitis development include similar risk factors to those of gingivitis and periodontitis. Table 124-30 outlines some of these risk factors.
Management of the aforementioned risk factors in acceptable, clinically proven modalities may lead to a decrease in the development of peri-mucositis or peri-implantitis cases. Additionally, incorporating these risk factors into the treatment planning process may also lead to a better management of patients more susceptible to the development of the peri-implant diseases, and potentially other treatment options may be recommended.
All dental implants must pass through the peri-mucosal layer of the soft tissue into the underlying bone. There is a biologic seal, a peri-implant mucosal seal, which prevents the introduction of harmful substances, such as bacterial plaque, toxins, and debris, which can initiate tissue and cell injury.2,3
Once the biologic seal is broken down, signs of peri-mucositis and/or peri-implantitis can be noticed. One of the best treatments for the reversal of these lesions is an early diagnosis of the problem, followed by immediate correction of the causative agent.
Establishing a Diagnosis
Diagnosis can be accomplished by conventional means similar to that of establishing the progression of periodontal disease. It is important to use light pressure during periodontal probing of the implant sulcular region, as the peri-implant mucosal seal is extremely delicate, and to take periapical radiographs to establish a baseline once the implant is put into final functional load. Dental-specific computed tomography scans are useful, as many peri-implantitis lesions can occur buccally or lingually, which may not be visible in periapical views.16
Registering probing depths in follow-up clinical exams that show bleeding suggests the presence of soft-tissue inflammation, and any suppuration suggests the presence of bacterial pathogens. These signs are indicative of peri-implant breakdown and necessitate additional radiographs and further evaluation and/or treatment.16
Further evaluation can include additional radiographs, stability measurements of the integrity of the implant, bacterial culturing, and inflammatory markers, which are all useful tools in establishing a diagnosis of peri-mucositis or peri-implantitis, and which can aid in implementing clinical protocols for the reversal or stabilization of the inflammatory disease process.
Treatment Options
Once the diagnosis of peri-mucositis or peri-implantitis is made, there are both nonsurgical and surgical treatment options to consider. Removal of any debris and the biofilm is essential in the attempted resolution of these lesions. Nonsurgical management of these types of lesions has proven to be more beneficial in treating peri-implant mucositis lesions than peri-implantitis lesions. Nonsurgical methods include carbon fiber curettes, titanium curettes, ultrasonic devices, local drug delivery, antiseptics, and laser therapy. Schar and colleagues31 showed that nonsurgical methods can be useful in the reduction of mucosal inflammation for up to 6 months, and that complete resolution of inflammation was not routinely achieved. Mombelli and colleagues15 stated that it is clear that local or systemic therapy of peri-implantitis can have a positive effect on clinical and microbiologic parameters, but the results so far point to limits of nonsurgical therapy in the resolution of deep peri-implantitis lesions.31
Surgical treatment of peri-implantitis provides better access to the peri-implant region and can allow for further therapies to change the peri-implant tissues and allow for the regeneration of bone. Numerous authors have presented case reports and case report series in regards to various methods of surgical treatment for peri-implantitis type lesions.32-42 Case reports vary in methods: implant surface mechanical debridement, removal of exposed and contaminated implant surface enhancements, localized drug delivery, localized delivery of antimicrobials to the implant surfaces, and laser therapy, followed by various bone grafting techniques. Mombelli15 concluded that surgical access by a full-thickness flap can allow for a thorough cleaning of the contaminated implant surface that, combined with systemic antibiotics and appropriate surgical methods and introduction of a bone substitute, may be advantageous in the stabilization of the peri-implant defect.
The introduction of laser therapy to aid in the stabilization and decontamination of the exposed and affected implant surface has demonstrated favorable initial results.32,43-45 Carbon dioxide (CO2) and Nd:YAG lasers are the most widely used lasers in oral surgery; however, they can increase temperatures enough to cause important structural damage to both implants and peri-implant bone.46,47 Nevertheless, Kreisler and colleagues48 showed that the CO2 laser could be used with low-power intensity to avoid thermal damage to the implant surface. The Er:YAG laser has been used in oral implant surgery because it provides precise sectioning of both hard and soft tissues with a negligible thermal effect and it neither alters the implant seal nor affects osseointegration. The Er,Cr:YSGG laser has been shown to minimize postoperative pain while decreasing prosthetic healing time in stage II implant therapy49 and provide minimal collateral thermal effects at the implant-bone interface.50,51 The following case reports will provide additional follow-up to a procedure described to treat peri-implantitis defects, with the introduction of the Er,Cr:YSGG laser as part of the clinical therapy for the correction of moderate to advanced peri-implantitis. The following protocol (Table 245) has been in effect for more than 3 years by the author for the correction of these types of cases.
Case 1
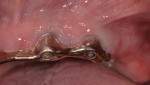
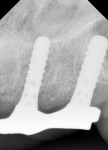
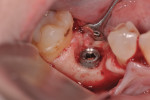
A 66-year-old nonsmoking man presented for implant reconstruction of his mandibular arch. Several years prior, implants had been placed into his maxillary arch after immediate removal of his remaining dentition. The four maxillary implants were restored by an implant-supported, bar-removable denture prosthesis. At the consultation visit, the patient presented with probing depths greater than 8 mm on two of the four maxillary implants, suppuration, and bleeding upon probing (Figure 1). Radiographically, large vertical defects, which contained circumferential type bone defects, were noted (Figure 2). The patient elected to undergo treatment to attempt to obtain resolution of the clinical signs and symptoms of peri-implantitis and to regenerate the supporting bone around the dental implants.
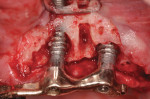
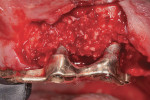
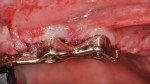
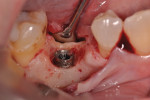
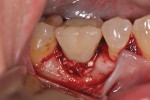
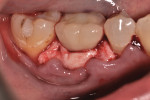
After administration of preoperative oral antibiotics (Augmentin 875 mg, 20 tablets, 1 tablet every 12 hours) started the day prior to the surgical procedure, the patient presented for treatment of the peri-implantitis lesions. Administration of an appropriate local anesthetic was given, followed by elevation of full-thickness muco-periosteal flaps. The initial pre-corrected peri-implantitis lesions, with resultant peri-implant tissue damage, can be seen in Figure 3. Following the debridement/antiseptic chemical treatment/laser decontamination (Er,Cr:YSGG, Biolase, www.biolase.com), the large peri-implant defects requiring regeneration were evident (Figure 4). A platelet-rich-plasma (PRP)-enhanced allogenic mineralized cancellous graft complex (1- to 2-mm particle size) was then placed around and into the peri-implant defects (Figure 5), followed by a PRP bioactive membrane placed over the graft complex seated. The site was then closed with 5.0 Monocryl® sutures (Ethicon, www.ethicon.com) in a continuous sling suturing method (Figure 6). The patient then completed the course of the systemic antibiotics prescribed, plus chlorhexidine rinses two times daily for 14 days post-surgery. The patient was then reinstructed on oral hygiene for the area, the bar clasps were replaced in the definitive prosthesis, and his bite was adjusted. A comparison of the 1.5-year postoperative periapical radiograph (Figure 7) with that taken upon presentation (Figure 2) shows the resolution of the peri-implant bone defects presentment. Probing depths at the 1.5-year postoperative visit were reduced to less than 4 mm in the area of the implants repaired.
Case 2
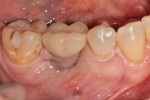
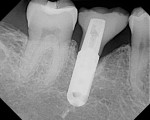
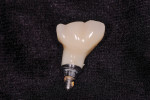
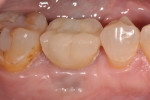
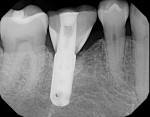
A 62-year-old nonsmoking woman presented for repair of advanced probing depths around an implant placed and restored approximately 6 years prior (Figure 8). The peri-implant tissues at the implant in the mandibular right first molar position lacked attached keratinized gingival tissue, had probing depth greater than 10 mm in the peri-implant area, and the area bled easily upon probing. The mesial and distal vertical defects present are apparent in the pretreatment periapical radiograph (Figure 9). The patient consented to undergoing the clinical therapy outlined for the treatment of peri-implantitis lesions in Table 2. After administration of the systemic antibiotic and administration of an appropriate local anesthetic, the implant crown was removed (Figure 10). Note the shape of the apical portion of the prosthesis, and how the natural emergence profile that should be present in an implant restoration was potentially a contributing factor in this case, making the patient unable to appropriately debride the sulcular area of the pre-existing implant crown.
After full-thickness flap elevation, the pre-existing peri-implant bone defect can be seen as a circumferential type intraosseous defect (Figure 11). Following the outlined mechanical debridement/chemical detoxification/decontamination protocol, the exposed implant surface was appropriately treated prior to bone regeneration techniques (Figure 12). Before replacing the implant crown, the porcelain surface of the apical third of the crown was etched, and composite material was added and contoured to provide for a more appropriate contour to support a more cleansable intrasulcular environment. The PRP/graft complex was then seated, and a PRP bioactive membrane was placed over the graft complex (Figure 13) prior to placement of a PRP-enhanced acellular dermal matrix graft (LifeNet, www.lifenethealth.org) to increase the connective tissue thickness on the facial of the implant surface (Figure 14). Closure was then accomplished by a continuous sling suturing technique with 5.0 Monocryl suture. The patient was instructed to continue the systemic Augmentin and to use chlorhexidine rinses two times daily for 2 weeks postoperatively. A 1-year post-repair clinical photo (Figure 15) shows the excellent soft tissue health present, and the stable zone of attached tissue present. Comparing the 1-year post-repair periapical radiograph (Figure 16) with the pre-repair radiograph (Figure 9), the bone fill on the mesial and distal surfaces of the implant fixture is evident.
Discussion
Clinicians are bound to run into the incidence of peri-mucositis and/or peri-implantitis cases in their recall systems, and the early diagnosis and treatment of these types of lesions can provide a favorable outcome before the disease reaches its terminal stage. Peri-mucositis cases carry a more favorable outcome if the etiology can be eliminated, and conservative nonsurgical approaches to remove the biofilm have been shown in the literature to provide a favorable outcome.15
On the other hand, peri-implantitis type lesions are much more difficult to completely eradicate, and nonsurgical protocols have been shown not to provide a positive resolution to the peri-implantitis type lesions.15 Mainly because flap exposure provides access to the exposed implant surface, surgical therapies allow for more effective debridement, decontamination, and detoxification of the affected implant surfaces in the peri-implantitis lesion. With the variety of implants available to the clinician today, there are some reports of different outcomes for treatment with specific surfaces versus other implant surfaces,46 though a complete discussion of implant selection is beyond the scope of this article.
The protocol for the treatment of peri-implantitis type lesions outlined here not only involves mechanical debridement, chemical detoxification, and the use a Cr,Er:YSGG laser to decontaminate the infected implant surface, but also allows for the direct re-apposition of bone. The author has observed resolution using this protocol more than 50 sites and recommends more extensive clinical studies to validate the results obtained on a larger scale and substantiate the clinical protocol used here.
Conclusion
Dental implants have become an integral and successful staple in treatment plans for patients on a routine basis. As is the case with natural teeth, localized infections can develop around implants that can be multi-factorial in origin. The incidence of peri-mucositis and peri-implantitis in the maintenance aspect of general and specialty practices presents a significant problem in regards to the long-term success rates of both the surgical and restorative aspect of patient care and should be addressed with an evidence-based treatment protocol.
About the Author
Paul S. Petrungaro, DDS, MS
President, Petrungaro Periodontics & Aesthetic Implantology
Speaker and Educator
Chicago, Illinois
References
1. Adell R, Eriksson B, Lekhollm U, et al. Long-term follow-up study of osseointegrated implants in the treatment of totally edentulous jaws. Int J Oral Maxillofac Implants. 1990;5(4):347-359.
2. Babbush CA. Dental Implants: The Art and Science. 2nd ed. Philadelphia, PA: WB. Saunders Co.; 2001:201-216.
3. Misch CE. Contemporary Implant Dentistry. 2nd ed. St. Louis, MO: Mosby-Year Book Inc.:1993.
4. Lidhe J, Meyle J; Group D of European Workshop on Periodontology. Peri-implant diseases: Consensus report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008;35(suppl 8):282-285.
5. Sanz M, Chapple IL; Working Group 4 of the VII European Workshop on Periodontology. Clinical research on peri-implant diseases: consensus report of Working Group 4. J Clin Periodontol. 2012;39(suppl 12):202-206.
6. Mombelli A, Lang NP. The diagnosis and treatment of peri-implantitis. Periodontol 2000. 1998;17:63-76.
7. Fransson C, Lekholm U, Jemt T, Berglundh T. Prevalence of subjects with progressive bone loss at implants. Clin Oral Implants Res. 2005;16(4):440-446.
8. Roos-Jansåker AM, Lindahl C, Renvert H, Renvert S. Nine-to fourteen-year follow-up of implant treatment. Part II: presence of peri-implant lesions. J Clin Periodontol. 2006;33(4):290-295.
9. Koldsland OC, Scheie A, Aass AM. Prevalence of peri-implantitis related to severity of the disease with different degrees of bone loss. J Periodontol. 2010;81(2):231-238.
10. Froum SJ, Rosen PS. A proposed classification for peri-implantitis. Int J Periodontics Restorative Dent. 2012;32(5):533-540.
11. Mombelli A, Van Oosten MAC, Schurch E, Lang NP. The microbiota associated with successful or failing osseointegrated titanium implants. Oral Microbiol Immunol. 1987;2:145-151.
12. Salvi GE, Aglietta M, Eick S, et al. Reversibility of experimental peri-implant mucositis compared with experimental gingivitis in humans. Clin Oral Implants Res. 2012;23(2):182-190.
13. Augthun M, Conrads G. Microbial findings of deep peri-implant bone defects. Int J Oral Maxillofac Implants. 1997;12(1):106-112.
14. Salcetti JM, Moriarty JD, Cooper LF, et al. The clinical, microbial, and host response characteristics of the failing implant. Int J Oral Maxillofac Implants.1997;12(1):32-42.
15. Mombelli A, Moëne R, Décaillet F. Surgical treatments of peri-implantitis. Eur J Oral Implantol. 2012;5(suppl d):S61-S70.
16. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnosis and clinical implications. J Periodontol. 2013;84(4):436-443.
17. Heitz-Mayfield LJ, Lang NP. Comparative biology of chronic and aggressive periodontitis vs. peri-implantitis. Periodontol 2000. 2010;53:167-181.
18. Leonhardt A, Renvert S, Dahlén G. Microbial findings at failing implants. Clin Oral Implants Res. 1999;10(5):339-345.
19. Sánchez-Gárces MA, Gay-Escoda C. Periimplantitis. Med Oral Patol Oral Cir Bucal. 2004;9(suppl 69-74):S63-S69.
20. Mombelli A, Feloutzis A, Brägger U, Lang NP. Treatment of peri-implantitis by local delivery of tetracycline. Clinical, microbiological and radiological results. Clin Oral Implants Res. 2001;12(4):287-294.
21. Duarte PM, de Mendonça AC, Máximo MB, et al. Differential cytokine expressions affect the severity of peri-implant disease. Clin Oral Implants Res. 2009;20(5):514-520.
22. Javed F, Al-Hezaimi K, Salameh Z, et al. Proinflammatory cytokines in the crevicular fluid of patients with peri-implantitis. Cytokine. 2011;53(1):8-12.
23. Plagnat D, Giannopoulou C, Carrel A, et al. Elastase, alpha2-macroglobulin and alkaline phosphatase in crevicular fluid from implants with and without periimplantitis. Clin Oral Implants Res. 2002;13(3):227-233.
24. Kozlovsky A, Tal H, Laufer BZ, et al. Impact of implant overloading on the peri-implant bone in inflamed and non-inflamed peri-implant mucosa. Clin Oral Implants Res. 2007;18(5):601-610.
25. Whörle PS. Single-tooth replacement in the aesthetic zone with immediate provisionalization: fourteen consecutive case reports. Pract Periodontics Aesthet Dent. 1998;10(9):1107-1114.
26. Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80(9):1388-1392.
27. Rocchietta I, Nisand D. A review assessing the quality of reporting of risk factor research in implant dentistry using smoking, diabetes and periodontitis and implant loss as an outcome: critical aspects in design and outcome assessment. J Clin Periodontol. 2012;39(suppl 12):S114-S121.
28. Bormann KH, Stühmer C, Z’Graggen M, et al. IL-1 polymorphism and periimplantitis. A literature review. Scheiz Monatsschr Zahnmed. 2010;120(6):510-520.
29. Tengvall P, Elwing H, Sjöqvist L, et al. Interaction between hydrogen peroxide and titanium: a possible role in the biocompatibility of titanium. Biomaterials. 1989;10(2):118-120.
30. Quirynen M, Gijbels F, Jacobs R. An infected jawbone site compromising successful osseointegration. Periodontol 2000. 2003;33(1):129-144.
31. Schär D, Ramseier CA, Eick S, et al. Anti-infective therapy of peri-implantitis with adjunctive local drug delivery or photodynamic therapy: six-month outcomes of a prospective randomized clinical trial. Clin Oral Implants Res. 2013;24(1):104-110.
32. Azzeh MM. Er,Cr:YSGG laser-assisted surgical treatment of peri-implantitis with 1-year reentry and 18-month follow-up. J Periodontol. 2008;79(10):2000-2005.
33. Büchter A, Kleinheinz J, Meyer U, Joos U. Treatment of severe peri-implant bone loss using autogenous bone and a bioabsorbable polymer that delivered doxycycline (Atridox). Br J Oral Maxillofac Surg. 2004;42(5):454-456.
34. Gammage DD, Bowman AE, Meffert RM. Clinical management of failing dental implants: four case reports. J Oral Implantol. 1989;15(2):124-131.
35. Hämmerle CH, Fourmousis I, Winkler JR, et al. Successful bone fill in late peri-implant defects using guided tissue regeneration. A short communication. J Periodontol. 1995;66(4):303-308.
36. Lozada JL, James RA, Boskovic M, et al. Surgical repair of peri-implant defects. J Oral Implantol. 1990;16(1):42-46.
37. Mellonig JT, Griffiths G, Mathys E, Spitznagel J Jr. Treatment of the failing implant: case reports. Int J Periodontics Restorative Dent. 1995;15(4):384-395.
38. Muller E, González YM, Andreana S. Treatment of peri-implantitis: longitudinal clinical and microbiological findings–a case report. Implant Dent. 1999;8(3):247-254.
39. Santos VR, Duarte PM. Surgical anti-infective mechanical therapy for peri-implantitis: a clinical report with a 12-month follow-up. Gen Dent. 2009;57(3):230-235; quiz 236-237.
40. Suh JJ, Simon Z, Jeon YS, et al. The use of implantoplasty and guided bone regeneration in the treatment of peri-implantitis: two case reports. Implant Dent. 2003;12(4):277-282.
41. Tinti C, Parma-Benfenati S. Treatment of peri-implant defects with the vertical ridge augmentation procedure: a patient report. Int J Oral Maxillofac Implants. 2001;16(4):572-577.
42. Tözüm TF, Keçeli HG. Treatment of peri-implant defect with modified sandwich bone augmentation. Case report and follow-up. NY State Dent J. 2008;74(4):52-57.
43. Schwarz F, Sahm N, Iglhaut G, Becker J. Impact of the method of surface debridement and decontamination on the clinical outcome following combined surgical therapy of peri-implantitis: a randomized controlled clinical study. J Clin Periodontol. 2011;38(3):276-284.
44. Arnabat-Domínguez J, Bragado-Novel M, España-Tost AJ, et al. Advantages and esthetic results of erbium, chromium:yttrium-scandium-gallium-garnet laser application in second-stage implant surgery in patients with insufficient gingival attachment: a report of three cases. Lasers Med Sci. 2010;25(3):459-464.
45. Petrungaro PS. A clinical therapy for the treatment of peri-implantitis. Inside Dentistry. 2013;5(9):94-100.
46. Swift JQ, Jenny JE, Hargreaves KM. Heat generation in hydroxyapatite-coated implants as a result of CO2 laser application. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79(4):410-415.
47. Romanos GE, Everts H, Nentwig GH. Effects of diode and Nd:YAG laser irradiation on titanium discs: a scanning electron microscope examination. J Periodontol. 2000;71(5):810-815.
48. Kreisler M, Götz H, Duschner H. Effect of Nd:YAG, Ho:YAG, Er:YAG, CO2, and GaAIAs laser irradiation on surface properties of endosseous dental implants. Int J Oral Maxillofac Implants. 2002;17(2):202-211.
49. Arnabat-Domínguez J, España-Tost AJ, Berini-Aytés L, Gay-Escoda C. Erbium:YAG laser application in the second phase of implant surgery: a pilot study in 20 patients. Int J Oral Maxillofac Implants. 2003;18(1):104-112.
50. Revilla-Gutiérrez V, Arnabat-Domínguez J, España-Tost AJ, Gay-Escoda C. Aplicaciones de los láseres de Er:YAG y de Er,Cr:YSGG en Odontología. RCOE. 2004;5(9):551-562.
51. Eversole LR, Rizoiu IM. Preliminary investigations on the utility of an erbium, chromium YSGG laser. J Calif Dent Assoc. 1995;23(12):41-47.