Stable Root Coverage with a Dermal Allograft Enriched with rhPDGF-BB
With a coronally-advanced flap technique, treatment over a large area is possible in one procedure
Treatment of gingival recession is not a new procedure for the periodontist. Methods of treatment have evolved significantly over the past 4 decades, however. Early modalities almost always involved graft-procurement from the palate. These procedures required two surgical sites and the morbidity associated with both areas. Though originally indicated for augmentation of keratinized gingiva,1 modifications of the free-gingival graft also allowed for root coverage.2 The morbidity associated with this technique resulted in the less-invasive subepithelial connective tissue graft. Langer and Calagna reported on the indications and procedural steps necessary for successful mucogingival enhancement with this technique.3
One technique that does not require a secondary graft is the coronally-advanced flap technique. Allen and Miller demonstrated that this technique can achieve significant root coverage for areas of shallow recession.4 In the early 1990s, clinicians began experimenting with combining exogenous materials with coronally-advanced flaps to achieve root coverage. Guided-tissue regeneration (GTR), originally used for treatment of intrabony periodontal defects, proved a useful treatment in areas with existing keratinized mucosa.5
Another alternative to autogenous soft tissue grafts is the use of dermal allografts. Techniques used to cover exposed root surfaces with these biomaterials have proven to be successful in certain applications.6 The advantages of these techniques include negating a second surgical site and the ability to treat large areas in one procedure, since limitations of autogenous tissue volume do not exist. The dermal allograft used in this case report (PerioDerm®, DENSTPLY Implants, www.dentsplyimplants.com) is processed differently than the most commonly used allograft, in that it is not soaked in antibiotics, allowing rapid revascularization. The incorporation of the graft within the surrounding soft tissues results in a more readily substituted graft, eliminating the foul smell commonly associated with exposed dermis, as experienced by the author.
Growth factors are frequently used in oral regenerative procedures. rhPDGF-BB has been shown to enhance periodontal regenerative and mucogingival surgery. The chemotactic and mitogenic properties of rhPDGF-BB recruit mesenchymal stem cells from the surrounding tissues and encourage their proliferation at the site of desired tissue regeneration. The author has found that a prerequisite for the successful use of any growth factor is their effective delivery and timed release. This is accomplished with a matrix capable of carrying them to the site. Collagen is one of the most frequently used matrices for several growth factors.
Accelular dermal allografts are mainly composed of collagen. These materials can augment soft tissue thickness and are not likely to elicit a local or systemic immune response. The combination of rhPDGF-BB and dermal allografts provides a bioactive matrix for corrective mucogingival surgery.
Case Description
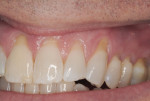
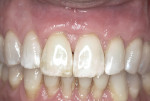
This case report demonstrates stable root coverage over seven maxillary teeth presenting with significant gingival recession. The patient is a healthy 48-year-old man. He has a history of moderate chronic periodontitis, treated with scaling and root planing, as well as regenerative and resective osseous surgery in posterior sextants. Periodontal maintenance is performed quarterly, alternating between periodontal and restorative practices. At a routine maintenance appointment, a significant increase in gingival recession was noted on teeth No. 8 through No. 14 (Figure 1). The patient accepted surgical treatment to address these teeth, as he was aware and displeased with the esthetic and hypersensitive nature of the affected teeth.
Following administration of local anesthesia (4% articaine with epinephrine 1/100,000), horizontal incisions were made at the level of the cementoenamel junctions (CEJs) on teeth No. 8 to No. 14. Vertical releasing incisions at the distobuccal aspect of No. 8 and No. 14 were made. A full-thickness flap was reflected to approximately 3 mm apical to the mucogingival junction. From that point, sharp-dissection to create a partial-thickness flap was performed to allow tension-free coronal repositioning of the gingival margin toward the CEJ of teeth No. 8 through No. 14.
Root-planing with finishing burs and curettes was done to thoroughly clean the roots and slightly reduce convexity to facilitate flap adaptation. Conditioning of the roots was done with 24% EDTA (Straumann® PrefGel®, Straumann, www.straumann.us) for approximately 2 minutes. This step eliminates the smear layer created by instrumentation, permitting open dentinal tubules to contact the overlying graft material.
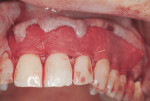
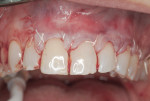
A dermal allograft (PerioDerm) with a thickness of 0.4- to 0.8-mm was hydrated with 0.3% rhPDGF-BB (GEM 21S®, Osteohealth, www.osteohealth.com) for approximately 20 minutes (Figure 2). The graft was trimmed to fit the exposed surgical site at the level of the CEJ of the treated teeth. It was sutured to the interproximal papillae with resorbable sutures (5-0 Monocryl, Ethicon, www.ethicon.com) (Figure 3). The overlying flap was positioned coronal to the CEJs to completely cover the growth factor–enhanced dermal allograft with sling sutures (Figure 4). Light pressure was applied for approximately 3 minutes to minimize the thickness of the blood clot between the graft and the roots as well as between the flap and the graft. A periodontal dressing (CoePak™, GC America, www.gcamerica.com) was applied.
The patient was prescribed amoxicillin for 7 days, a nonsteroidal anti-inflammatory drug (etodolac) for 3 days to minimize inflammation, and a chlorhexidine mouthrinse. Sutures were removed, where loose, at 11 days postoperatively (Figure 5). The patient avoided manual brushing and continued chlorhexidine rinses for 2 additional weeks. At that point, manual brushing with an extra-soft toothbrush using the roll technique was used for a month.
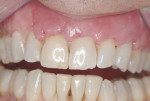
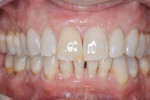
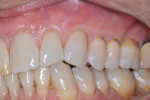
The patient was seen for maintenance every 3 months, resuming his alternating routine between periodontal and restorative offices. Photographs documenting the healing process were taken over the next year and a half. At 6 months after surgery, esthetically pleasing and significant root coverage was documented (Figure 6). At approximately 18 months, all treated teeth demonstrated between approximately 80% to 100% root coverage and a healthy band of keratinized gingiva (Figure 7 and Figure 8). The patient reported to be very pleased with the outcome of the surgical therapy and remains on quarterly maintenance.
Discussion
The use of growth factors to improve the outcomes of surgical procedures in medicine and dentistry is common. In periodontal surgery, rhPDGF-BB has been combined with bone alloplasts and allografts to enhance results in regenerative surgery.7,8
Mucogingival surgery may also benefit from the use of rhPDGF-BB. In a case series, Rubins showed excellent results when a subepithelial connective tissue graft was hydrated with rhPDGF-BB for at least 15 minutes before placement. Their series did not compare the classic procedure without adjunctive growth factors to their technique, so no conclusion can be made regarding how rhPDGF-BB enhanced the outcomes. Singh and Suresh compared a rhPDGF-BB–enriched GTR procedure to GTR alone in the treatment of gingival recession.10 They found significantly greater root coverage at 6 months in the group receiving the combination therapy to GTR alone. This same approach of rhPDGF-BB with beta-TCP plus GTR was tested as an alternative to subepithelial, connective tissue grafts in a comparative case series.11 The authors found comparable results and suggested this growth factor-enhanced procedure as an alternative to autogenous CT graft harvesting for root coverage.
References
1. Sullivan HC, Atkins JH. Freeutogenous gingival grafts. 1. Principles of successful grafting. Periodontics. 1968;6(1):5-13.
2. Miller PD Jr. Root coverage using a free soft tissue autograft following citric acid application. Part 1: Technique. Int J Periodontics Restorative Dent. 1982;2(1):65-70.
3. Langer B, Calagna LJ. The subepithelial connective tissue graft. A new approach to the enhancement of anterior cosmetics. Int J Periodontics Restorative Dent. 1982;2(2):22-33.
4. Allen EP, Miller PD Jr. Coronal positioning of existing gingiva: short term results in the treatment of shallow marginal tissue recession. J Periodontol. 1989;60(6):316-319.
5. Prato GP,Clauser C, Cortellini P. Guided tissue regeneration and a free gingival graft for the management of buccal recession: a case report. Int J Periodontics Restorative Dent. 1993;13(6):486-493.
6. Aichelmann-Reidy ME, Yukna RA, Evans GH, Nasr HF, Mayer ET. Clinical evaluation of acellular allograft dermis for the treatment of human gingival recession. J Periodontol.;2001;72(8):998-1005.
7. McGuire MK, Kao RT, Nevins M, Lynch SE. rhPDGF BB promotes healing of periodontal defects: 24-month clinical and radiographic observations. Int J Periodontics Restorative Dent.;2006;26(3):223-231.
8. Nevins M, Camelo M, Nevins ML, Schenk RK,L ynch SE. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J Periodontol.;2003;74(9):1282-92.
9. Rubins RP, Tolmie PN, Corsig KT, Kerr EN, Kim DM. Subepithelial connective tissue graft with growth factor for the treatment of maxillary gingival recession defects. Int J Periodontics Restorative Dent. 2013;33(1):43-50.
10. Singh P, Suresh DK. Clinical evaluation of GEM 21S® and a collagen membrane with a coronally advanced flap as a root coverage procedure in the treatment of gingival recession defects: A comparative study. J Indian Soc Periodontol. 2012;16(4):577-583. doi: 10.4103/0972-124X.106919.
11. McGuire MK, Scheyer ET. Comparison of recombinant human platelet-derived growth factor-BB plus beta tricalcium phosphate and a collagen membrane to subepithelial connective tissue grafting for the treatment of recession defects: a case series. Int J Periodontics Restorative Dent. 2006;26(2):127-133.
About the Author
Barry P. Levin, DMD
Diplomate, American Board of Periodontology
Clinical Associate Professor,
University of Pennsylvania, Department of Periodontology
Private Practice Limited to Periodontics and Implant Surgery
Elkins Park, Pennsylvania