Practice-Based Evaluation of a New Oral Hygiene Product
Good oral hygiene involves the regular removal of dental plaque, biofilm, and debris from the teeth, tongue, dental implants, and restorations. A toothbrush and dentifrice are commonly used for this purpose. Professional dental visits are important to remove any accumulation of plaque and calculus not removed by brushing.
There are many dental hygiene aids that clinicians can recommend to their patients. Manufacturers are continually updating consumer dental products by adding more features to capture users’ attention and hopefully enhance product use and compliance. It is therefore incumbent on dental professionals to stay current on what is commercially available and, as needed, recommend specific products best suited to their patients’ needs.
Ultreo™ (Ultreo, Inc, Redmond, WA) (Figure 1) is the first power toothbrush to combine sonic bristle motion and ultrasound for enhanced plaque removal and improved oral health. As clinicians who recommend power toothbrushes, an evaluation of Ultreo was needed before recommending the new power toothbrush to patients. More importantly, a test of its ability to remove plaque and maintain the patient’s oral hygiene between visits had to be established. The following report describes a small-scale evaluation of Ultreo that enabled the clinicians’ dental personnel to decide whether to recommend this product to patients in their practice.
SUBJECT SELECTION AND ORAL HYGIENE RECOMMENDATIONS
Three subjects were selected who were on 3- or 4-month recalls. Subject 1 was a 62-year-old woman with 26 periodontal pockets 5 mm or greater in depth. She had adequate oral hygiene, but tended to build up calculus between visits. Subject 2 was a 67-year-old woman with four periodontal pockets 5 mm or greater in depth. She also had adequate oral hygiene, but tended to build up moderate extrinsic stain because of a dietary intake of green tea, blueberries, and fruit juices. Subject 3 was a 61-year-old man with 37 periodontal pockets 5 mm or greater in depth. He tended to present with moderate levels of plaque at recall visits. All three subjects were presently using another power toothbrush.
At their baseline visits, the subjects’ overall oral health was recorded using standard office periodontal charting. Full-mouth periodontal probing was performed. After baseline data was recorded, the subjects were shown how to best use the power toothbrush’s features, including using light pressure of the bristles, using a pea-sized amount of toothpaste, and keeping fluid around the bristles and waveguide while brushing. The subjects were asked to use the Ultreo twice a day for 2 minutes each time, using their normal toothpaste. At subsequent visits the subjects brought the Ultreo with them. Data was recorded in a similar manner as at the baseline visit. The subjects were asked whether they noted any adverse reactions to the brush. Oral soft and hard tissue and restorations were examined for any adverse events. A questionnaire was completed after each use period to record user reaction to the toothbrush.
OBSERVATIONS
The subjects were observed over a 3-month period. During this period they returned to the dental office for evaluation as available, with a minimum 2-week period between visits. All three subjects found the brushing experience with the Ultreo to be favorable and continued to use the product during the evaluation period. Evaluation of their brushing technique at subsequent visits indicated some need for improvement (in particular, maintaining bristle contact at the cervical margin of the tooth).
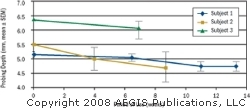
Subject 1 exhibited less bleeding after 6 weeks, and Subject 2 and Subject 3 presented with less plaque after 8 and 7 weeks, respectively. The examiner noted less extrinsic stain on Subject 2 after 9 weeks (Figure 2A and Figure 2B) and less calculus buildup than usual on Subject 1 after 13 weeks. Probing depths of 5 mm or greater at baseline were followed on all of the subjects. The average probing depth for these pockets was calculated at each subsequent visit. Results are presented in Figure 3. All three subjects demonstrated a reduction in probing depth during their respective use periods.
No adverse events were observed or reported by the patients during the evaluations that could be attributed to the toothbrush. Two occurrences of sensitivity were noted by the subjects, but both were transient in nature and nonexistent at subsequent visits. The questionnaires revealed that all three subjects felt that the toothbrush was cleaning their teeth well and that it felt comfortable in their mouths. When asked what they liked most about the toothbrush, the subjects responded favorably to the brush handle and design, the feeling of very clean teeth, and the appearance of less calculus buildup. Subjects’ least favorite aspect of the toothbrush included the size of the brush head (two felt it was too large, one felt it was too small).
CONCLUSION
When evaluating new technology, it is important to evaluate controlled clinical studies as well as in-office patient experiences. This is particularly true regarding products that subjects need to use by themselves in their home. Favorable changes were observed in the oral health of all three subjects after using the Ultreo. All three subjects liked the product and appreciated its clinical performance. Because compliance is important to achieving good oral hygiene results, the authors feel that user acceptance of a product is a critical metric when evaluating a product.
This article was written by Donald P. Callan,DDS; and Millicient Wren, RDH. The authors maintain a private practice in Little Rock, Arkansas.
For more information, contact:
Ultreo, Inc
Phone: 877-485-8736
Web site: www.ultreo.com
E-mail: info@ultreo.com
DISCLAIMER
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry. The preceding is not a warranty, endorsement, or approval for theaforementioned products or services or their effectiveness, quality, or safety on the part of Inside Dentistry or AEGIS Communications. The publisher disclaims responsibility for any injury to persons or propertyresulting from any ideas or products referred to in the preceding material.
 |
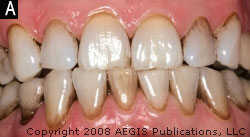 |
| Figure 1 The Ultreo power toothbrush. | Figure 2A Subject 2, extrinsic stain at the baseline visit. |
 |
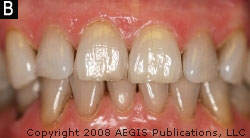 |
| Figure 3 Reduction in probing depth over the evaluation period. | Figure 2B Subject 2, extrinsic stain after 9 weeks of using the Ultreo. |



