Faster Onset and More Comfortable Injection with Alkalinized 2% Lidocaine with Epinephrine 1:100,000
ABSTRACT— BACKGROUND: The pH of lidocaine with epinephrine in dental cartridges ranges between 2.9 and 4.4. In this pH range, less than 0.1% of the anesthetic is in the de-ionized or “active” form. The acidity of the anesthetic may delay onset and contribute to injection pain.
OBJECTIVE: The study compared anesthetic latency and injection pain for alkalinized versus non-alkalinized anesthetic in inferior alveolar nerve blocks (IANBs).
METHODS: The study buffered the anesthetic directly in the cartridges using a mixing pen device. The study included 20 participants, each receiving one control and one test IANB injection. The control solution was non-alkalinized 2% lidocaine/epinephrine 1:100,000 at pH 3.85. The test solution was 2% lidocaine/epinephrine 1:100,000 alkalinized to pH 7.31. Latency was measured using endodontic ice confirmed with an electric pulp tester (EPT), and injection pain was measured using a visual analog scale (VAS).
ONSET TIME: With the alkalinized anesthetic, 71% of participants achieved pulpal analgesia in 2 minutes or less. With non-alkalinized anesthetic, 12% achieved pulpal analgesia in 2 minutes or less (P = 0.001). The average time to pulpal analgesia for the non-alkalinized anesthetic was 6:37 (range 0:55 to 13:25). Average time to pulpal analgesia for alkalinized anesthetic was 1:51 (range 0:11 to 6:10) (P = 0.001).
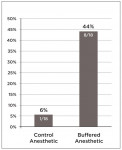
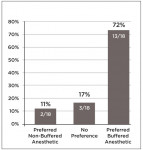
INJECTION PAIN RESULTS: 72% of the participants rated the alkalinized injection as more comfortable, 11% rated the non-alkalinized injection as more comfortable, and 17% reported no preference (P = 0.013). Forty-four percent of the patients receiving alkalinized anesthetic rated the injection pain as zero (“no pain”) on a 100-mm VAS, compared to 6% of the patients who received non-alkalinized anesthetic (P = 0.056).
CONCLUSIONS: Alkalinizing lidocaine with epinephrine toward physiologic pH immediately before injection significantly reduces anesthetic onset time and increases the comfort of the injection.
CLINICAL IMPLICATIONS: Clinicians can begin procedures more quickly and give a more comfortable injection by alkalinizing their lidocaine/epinephrine immediately before delivering the injection.
Local anesthetics represent some of the most widely used drugs in medicine and dentistry. A Canadian study suggests that the average annual usage per individual dentist is about 1,800 cartridges of dental anesthetic,1 or nine cartridges per day for a dentist who practices 200 days per year.
The challenge is that while local anesthetic is the dentist’s primary tool for pain management, its acidity may contribute to lengthy anesthetic waiting periods and cause the “bee sting effect” or burning and stinging during the injection. Many dentists are surprised to learn that the most widely used dental anesthetic solutions are formulated at the pH of lemon juice.
Alkalinizing dental anesthetic (procaine) was first studied by Gros and then written about by Laewen in 1910. Since the introduction of lidocaine in 1948, there have been dozens of studies evaluating whether alkalinization would improve onset time or reduce injection pain.2-36 Occasionally, alkalinization has been studied in combination with warming30,35 or injection speed.28
In December 2010, the Cochrane Collaboration completed a systematic review of 23 peer-reviewed studies on alkalinizing lidocaine and lidocaine with epinephrine, concluding that alkalinizing these two local anesthetics was a safe and effective method of decreasing injection pain and improving patient satisfaction.37
Even so, relatively few dentists are familiar with alkalinization, at least as compared to their counterparts in other areas of medicine.38 One commentator attributes the difference to the fact that, unlike most physicians, dentists use sealed anesthetic cartridges that are not easily alkalinized.39 This may also be the reason that there are just two peer-reviewed studies that have evaluated the efficacy of alkalinized anesthetic in dentistry.40,41
It is unlikely that dentists will move away from the standardized single-dose disposable anesthetic cartridge, raising the question whether, if alkalinization improved dental anesthetic performance, there might be a convenient way to alkalinize the anesthetic cartridges without disrupting the routine delivery of dental care. The alkalinization armamentarium evaluated in this study allows the dental practitioner to alkalinize a cartridge of lidocaine with epinephrine at chairside immediately before loading the syringe and delivering the injection.
Methods and Materials
Study Design
The study used a prospective, randomized, double-blind, crossover design to measure local anesthetic onset time and injection pain among 20 healthy adult volunteers who received an inferior alveolar nerve block (IANB) using alkalinized lidocaine with epinephrine at one appointment, and non-alkalinized lidocaine with epinephrine at another.
Materials and Alkalinization Procedure
For the control solution, the study used 2% lidocaine with epinephrine 1:100,000 with a pH of 3.85 which was provided in 1.8 ml cartridges (DENTSPLY International, www.dentsply.com). As its test solution, the study used cartridges of 2% lidocaine with epinephrine 1:100,000 (DENTSPLY International) alkalinized at a 9:1 ratio with a resulting pH of 7.31 using 8.4% sodium bicarbonate (Onpharma Inc., www.onpharma.com) via an automated compounding pen (Onset®, Onpharma Inc.) (Figure 1). The bicarbonate solution had a pH of 7.55. The cartridges of 2% lidocaine with epinephrine 1:100,000 used for both the test and control injections were from the same manufacturer and lot and were not expired.
In laboratory testing before the study, using an Orion 3-Star pH meter (Thermo Fisher Scientific, www.thermoscientific.com) regularly calibrated with buffer solutions (Oakton pH Buffer Solutions, Oakton Instruments, www.4oakton.com) at pH 4 and 7, an investigator measured the pH of 20 cartridges of 2% lidocaine with epinephrine 1:100,000. The cartridges were from the same lot used for control and test solutions in the study. The pH measured 3.85 ± 0.03 (SD). The investigator measured 20 additional cartridges from the same lot after they had been alkalinized per the process described below. The pH of the alkalinized cartridges was 7.31 ± 0.02 (SD).
During the study, the investigators alkalinized each anesthetic cartridge using the following process: In an operatory out of sight and hearing of any of the other persons who were involved in the study (the dentist, the dental assistant, and the participant), an investigator connected a 1.8-mL cartridge of 2% lidocaine with epinephrine 1:100,000 to a mixing pen that contained a cartridge of 8.4% sodium bicarbonate solution. The mixing pen delivered 0.18 mL of alkalinizing solution into the anesthetic cartridge, and simultaneously transferred 0.18 mL of anesthetic from the cartridge into a reservoir in the pen. The anesthetic cartridge was then immediately removed from the pen and taken into the operatory and delivered to the dental assistant.
Injection Procedures
The dental assistant immediately loaded the cartridge into a standard aspirating dental syringe (Patterson Dental, www.pattersondental.com) and handed it to the dentist, who began the injection using his normal technique for delivering an IANB. For control anesthetic, the only difference was that a cartridge that had not been alkalinized was handed to the dental assistant. There were no identifying marks on the cartridges or other indications that would allow the dentist, the assistant, or the participant to ascertain whether the anesthetic cartridge contained control or alkalinized anesthetic.
No topical anesthetic was used and no other pain management techniques were employed.
The dentist injected the contents of the 1.8-mL anesthetic cartridges over 1 full minute using a 27-gauge long needle (Tyco).
Before the injection was delivered, the dentist had confirmed pulpal vitality on the participants’ right or left lower first molar (the “test tooth”), indicated by sensitivity to cold using Green Hygenic® Endo-Ice® (Coltène-Whaledent, www.coltene.com) confirmed with an electric pulp tester (EPT). The contralateral cuspid was also tested for vitality using the same method (the “contralateral tooth”).
Time to Onset Measurements
Prior to the injection, participants were instructed that, after the injection, they should raise their hand immediately if they began to feel numb or to have a tingling sensation in the lower lip. As soon as the participant gave this notification, the dentist applied Green Endo Ice to a cotton pellet and placed the pellet against the buccal cervical surface of the test tooth and asked the participant to report if there was any sensation. If the participant reported any sensation, the procedure was repeated every 15 seconds until there was loss of sensation. Once sensation to cold was lost, the time to loss of sensation was noted, and then the loss of sensation was confirmed using the EPT. Confirmation was defined in the protocol as the participant reporting no sensation at an 80 reading on the EPT. If confirmed, the onset time noted for the cold test was entered as the time to pulpal analgesia for the test. If the participant continued to have sensation to the EPT, the EPT test was continued at a 30-second interval until sensation was lost, in which case this latter time was entered as the time to pulpal analgesia. Once loss of sensation was determined on the test tooth using the above method, the contralateral tooth was tested using the EPT to ensure that the EPT remained functional.
If a participant did not lose sensation to the Endo Ice test or EPT within 15 minutes after the injection, the injection was deemed to be an anatomical miss, and no data from the injection was used in the analysis. One injection in the study was deemed an anatomical miss.
Injection Pain Measurements
The study also measured injection pain using a 100-mm VAS. Participants were shown the VAS before the injection and instructed on its use by the dentist. At the top of the 100-mm scale, the words “The Worst Pain Imaginable” were written, and at the bottom of the 100-mm scale the words “No Pain” were written. Participants were instructed that after the injection was delivered, they would be asked to point to the place on the scale that represented the amount of pain they experienced, if any. They were also told that in order to more clearly distinguish injection pain from needle insertion pain, once the needle had been placed in the tissue, the dentist would tell the participant that the injection was starting, and that would be the point at which the participant should begin evaluating whether he or she experienced injection pain.
Immediately after completing the injection, the participants were asked to indicate the place on the VAS that represented the level of injection pain experienced, which was marked with an “X.”
Crossover Methodology
Three weeks after Day 1 of testing, Day 2 of testing was completed. Two participants did not complete Day 2. Participants received alkalinized anesthetic on Day 2 if they had received control anesthetic on Day 1, and vice versa. The injection was delivered on the same side of the mouth as in Day 1, and the same tooth was tested in order to evaluate onset time. All testing procedures were otherwise identical.
IRB Approval and Consent
Western IRB, a private independent institutional review board, approved in advance the study protocol and the informed consent document. The objectives and format of the study were presented to the participants in both verbal and written form, and informed consent was obtained.
Participants
The 20 participants in the study ranged in age from 23 to 76 years. The mean age of the participants was 41.6 years. Eighteen participants completed both days of study treatment; two did not. Per the study protocol, no data for the participants who did not complete both days of treatment was used.
The protocol excluded any prospective participant who was pregnant, had known allergies or hypersensitivities to lidocaine or to epinephrine, had any medical condition that represented a contraindication for the use of lidocaine or epinephrine, used within 7 days of treatment any agent or therapy known to affect perception of pain, was missing the mandibular cuspid or mandibular first molar on either side of his or her mouth, or had full-coverage restoration on any of his or her mandibular cuspids or mandibular first molars.
Results
Onset Time
The mean onset time for complete pulpal or “surgical” analgesia for the non-alkalinized local anesthetic was 6 minutes and 37 seconds (Figure 2). The mean onset time for the alkalinized local anesthetic was 1 minute and 51 seconds. Using a paired Wilcoxon ranked-sign test, this difference was determined to be statistically significant, having a P value ≤ 0.001.
The time to achieving surgical analgesia among a minimum of 70% of the participants using the non-alkalinized local anesthetic was 8 minutes and 30 seconds. The time to achieving surgical analgesia among a minimum of 70% of the participants using alkalinized local anesthetic was 1 minute and 57 seconds.
The percentage of participants achieving surgical analgesia at 2 minutes was 11% for the non-alkalinized local anesthetic, while for the alkalinized local anesthetic it was 71%. Using a repeated measures test, this difference was determined to be statistically significant, having a P value ≤ 0.001. Treatment order did not affect the time until pulpal analgesia
(P ≥ 0.328). Figure 2 shows, side-by-side in a butterfly graph, the number of participants that achieved pulpal analgesia at 30-second intervals from zero to 15 minutes, comparing at each time point alkalinized versus non-alkalinized lidocaine with epinephrine.
Injection Pain
Figure 3 shows the number of participants who reported no injection pain. The percentage of participants receiving the alkalinized anesthetic that rated the injection pain to be zero or pain-free (44%) was seven times the percentage of participants rating the control anesthetic to be pain-free (6%). This result was determined to be statistically significant using a repeated measures test (P = 0.004). Treatment order did not affect whether pain was experienced with the test solution (P value = 0.437) or with the control solution (P value = 0.437).
Figure 4 shows the number of participants who rated alkalinized or non-alkalinized anesthetic as the most comfortable, and those who indicated no preference. Of the 18 participants who completed the study, 13 (72%) rated the alkalinized anesthetic as the most comfortable, two (11%) rated the non-alkalinized anesthetic as the most comfortable, and three (17%) indicated no preference. Using the Mehta and Patel version of Fisher’s exact test, it was determined that the differences were statistically significant (P = 0.003). The order of the injections did not affect the pain ratings (P ≥ 0.621).
Discussion
Alkalinizing Local Anesthetic Injections Reduces Latency and Improves Onset Time
Only the de-ionized or free-base form of the local anesthetic, sometimes called the “active form” of the anesthetic,42,43 can freely penetrate the lipid-rich nerve sheath. Once inside the axon, the de-ionized molecule acquires a charge and becomes cationic, capable of blocking the nerve’s sodium channels, and thus inhibiting the action potential of the nerve.14 However, typically, very little of the anesthetic delivered in an injection is in the de-ionized form. According to the Henderson-Hasselbalch equation, the ratio of de-ionized anesthetic molecules to ionized anesthetic molecules at a pH of 3.5 is only one de-ionized molecule for every 25,000 of the ionized (cationic) anesthetic molecules.
Investigators report that the pH of commercially available dental anesthetic cartridges containing 2% lidocaine with epinephrine 1:100,000 ranges from 2.8644 to 4.16,45 or on average, about 3.5. In this study, the pH of the control anesthetic was 3.85.
Normally, the body’s tissue and fluids immediately begin acting upon the injectate and will raise the pH to near physiologic over an extended period of time.46 Ferndandez reported that the in-vitro alkalinization process had its maximum effect about 45 minutes post-injection, when, if practitioners were able to wait this long, the research showed that 95% of dental patients would achieve surgical analgesia.47
Of course, for most dental practices, it would not be practical to wait 45 minutes to start treatment or make a determination that another injection is necessary. Practitioners report that, for blocks, they generally wait 15 minutes to evaluate their patients for numbness and if, after this waiting period, the patient has not achieved analgesia, they will presume a missed block and proceed with a second injection. Studies that evaluate anesthetic success reflect the same wait time before “calling a miss.” Numerous studies48-61 measured anesthetic success or failure using an EPT at 15 minutes, while other studies62-68 measured success or failure at 16 minutes.
Reports that dentists typically wait about 15 minutes after delivering the injection to return to the operatory and determine whether the patient is sufficiently numb to begin the procedure may be evaluated in light of the curve showing the time course of analgesia (Figure 5).
One might expect that regularly returning to the operatory when only one-third of the patients are numb enough to treat would not become the preferred routine, which it has not. At 5 minutes, the percentage of patients that are numb is about one-third, and at 10 minutes, according to the curve in Figure 5, this figure is just approaching 60%. Waiting 15 or 16 minutes (as the aforementioned studies did) means that the practitioner is likely to return and find that about two-thirds of his or her patients are completely numb. Again referring to the curve in Figure 5 showing the time course of analgesia, at about 15 minutes the curve flattens, meaning waiting longer for onset to occur delivers fewer and fewer additional patients that would become numb. Hence, there appears to be practical wisdom in waiting about 15 minutes for patients receiving IANBs to get numb.
The practical question is whether onset time must necessarily be this slow.
It has been theorized that the delay in local anesthetic onset (compared, for instance, to the nearly immediate onset of general anesthesia) is because it takes time for the body to change a sufficient amount of the injected solution from the ionized to the de-ionized form of anesthetic. This involves variables of both anatomy and physiology, which can frustrate and delay the process, although Fernandez showed that if each patient was given 45 minutes, the body’s bicarbonate buffering system would eventually prevail in all but about 5% of dental blocks. These are probably the only true anatomical misses. The hypothesis under investigation here is that if it were possible to raise the pH of the anesthetic immediately before the injection, onset would occur significantly more quickly.
The investigators in this study raised the pH of the anesthetic cartridge to 7.3 just before the cartridge was loaded into the standard dental anesthetic syringe. Using the Onset Mixing Pen (Figure 1) the investigators delivered 0.18 mL of sodium bicarbonate into the anesthetic cartridge while removing the same amount of anesthetic, resulting in a mixed ratio of 9:1. The alkalinized cartridge was then immediately loaded into the syringe and the IANB was delivered. According to Henderson-Hasselbalch, in this study alkalinizing the cartridge dramatically increased the ratio of active anesthetic in the injection, raising it from 11,000:1 to 4:1.
The results shown in Figure 2 demonstrate a noticeable improvement in time to achieving surgical analgesia for the alkalinized anesthetic. More than two-thirds of the participants achieved profound numbness in 2 minutes using the alkalinized cartridges. It took the non-alkalinized anesthetic 9 minutes to achieve this level of success, which is actually better than the time course reflected in other published studies, where standard non-alkalinized lidocaine and articaine took more than 15 minutes to reach the two-thirds success mark.
This study demonstrated a statistically significant improvement; whether achieving a two-thirds success rate in 2 minutes represents a practical clinical improvement versus 9 minutes (or the 15 minutes suggested by the time course of analgesia curve and the associated studies) is a question that may be worth further evaluation elsewhere, as is briefly discussed in the conclusion section below.
Alkalinized Local Anesthetic Injections are More Comfortable
Figure 3 and Figure 4 show that alkalinizing local anesthetic makes a difference in the injection pain experienced by patients, and that patients would prefer alkalinized anesthetic over traditional non-alkalinized anesthetic for blocks.
Pain is a highly subjective sensation and its measurement can be difficult. However, this study used a crossover design to make each participant his or her own control for the purposes of pain evaluation. It used the 100-mm visual analog scale (VAS), which has been accepted by most researchers as a reliable method of measuring pain.
Although the results of this study demonstrate that alkalinizing local anesthetic before injection reduces injection pain, the mechanisms by which alkalinization may attenuate injection pain are not certain,39 nor are the root causes of
injection pain well understood.70 Pain signals may be induced when an injection activates nociceptors in the tissues that sense cold,34 acidity,71 or tissue distention.72 The following discussion traces the primary theories of why alkalinizing local anesthetic makes the injection more comfortable for the patient.
Alkalinized anesthetic may produce less injection pain because it is pH neutral. It has been suggested that the likely cause of injection pain is the acidity of the local anesthetic.12 As mentioned, studies have shown that the pH of lidocaine with epinephrine used in dentistry is as low as 2.86, which is essentially the pH of lemon juice. Most people have at some point had the painful experience of getting lemon juice or its equivalent in a cut. It stands to reason that creating a wound with a needle and bathing that wound with local anesthetic having the pH of lemon juice would cause the same kind of pain. However, at least one study73 suggests that injection pain does not strongly correlate to the acidity of the local anesthetic used.
Increasing the base form of anesthetic may block the injection pain signal. As previously mentioned, only the de-ionized, free-base, or “active” form of the local anesthetic42,43 readily penetrates the lipid-rich nerve sheath. Alkalinizing the anesthetic solution before injection immediately increases the active form of the drug. Talu and colleagues studied alkalinized anesthetic in facial surgery and believed that the improved speed of the block with alkalinized anesthetic resulted in less injection pain: “Neutralizing anesthetic solutions would increase their uncharged basic form; thus, the diffusion of the anesthetic solution through interstitial tissues is increased. This would result in a higher concentration of the drug in the nerve axoplasm and a more rapid block of the sensory fibers. As a more rapid block develops, the pain on skin infiltration is believed to be blocked before it has even been sensed.”30
Similarly, in a study of alkalinized lidocaine used for wound repair in an emergency department, Brogan and colleagues attributed reduced injection pain to faster onset speed: “The increased ratio of uncharged to charged molecules of any given anesthetic is responsible for the decrease in pain, perhaps because it increases the speed of onset of anesthetic.”74
Free CO2 produced during alkalinization may block the injection pain signal. The process of combining sodium bicarbonate with lidocaine HCl creates water, salt, and free carbon dioxide. Catchlove demonstrated that free CO2 in lidocaine solution has an independent anesthetic effect and that CO2 and local anesthetics have similar effects on peripheral nerves. He suggested that where a solution contains both lidocaine and free CO2, it is the CO2 that may cause the more immediate form of analgesia, writing: “Since CO2 diffuses rapidly through the sheath and has an independent anesthetic effect, it probably reaches the axon before the local anesthetic, causing the earliest phase of the block.”75
Could it be that CO2 represents a faster-acting anesthetic that provides analgesia before the body can register pain from the injection’s acidity, temperature, tissue distention, or any other source?
Similarly, Bokesch and colleagues demonstrated a significantly faster, more profound conduction block with lidocaine when there was free CO2 in the solution.76 Raymond and colleagues reported that free CO2 made lidocaine twice as potent,77 while Condouris and Sakalis reported that free CO2 created a 10-fold increase in procaine action.78
Curatolo attributes the performance improvement to ion trapping,79 which is also sometimes referred to as “diffusion trapping,”75 or the “cation-trap effect.”80 The ion-trapping theory postulates that the CO2 rapidly penetrates the nerve membrane and enters the axoplasm, where it reacts with water to create carbonic acid. This process acidifies the axoplasm,81 which makes hydrogen ions (H+) available to convert into ionized anesthetic the de-ionized anesthetic molecules (made more abundant via alkalinization) that have crossed the membrane and entered the axoplasm. The converted ionized anesthetic molecules are not lipid-soluble; hence, they cannot easily leave the axon once converted82 and are effectively “trapped” in the axon of the nerve. The trapped ionized form of lidocaine blocks sodium channels from within the axon, eventually resulting in analgesia.6,83
By acidifying the axoplasm, the free CO2 created in the alkalinization process may also establish a pH gradient across the nerve membrane, promoting diffusion of the more basic de-ionized anesthetic molecules across the membrane and into the axon.83,84 Alternatively, Catchlove suggested that the acidification process sets up a concentration gradient, causing the more abundant de-ionized anesthetic outside the nerve sheath to diffuse into the axon.75
All these studies suggest that the creation and retention of free CO2 in the bolus of the injection may play a role in the performance improvement observed using alkalinized local anesthetics. Attention to preserving or optimizing dissolved CO2 in the bolus of an alkalinized anesthetic injection may, therefore, be worth considering in determining the mixing and delivery methods for alkalinized local anesthetic.
The Mixing and Delivery System Should Prevent Loss of Free CO2 Created in the Alkalinization Process
Presuming that free CO2 plays a role in creating faster, more comfortable, and more profound analgesia, it would be important to understand the volatility of the free CO2 that can be a part of the bolus of the anesthetic injection, and methods for ensuring that the CO2 is not lost prior to injection.
If left exposed to the ambient air, alkalinized anesthetic will gradually lose CO2 at the air–liquid interface. Ackerman and colleagues suggested that practitioners alkalinize local anesthetic solutions in mixing vessels that have no air–liquid interface at all.85 Unfortunately, several of the alkalinization papers that have recommended compounding processes and ratios over the years have failed to follow this advice and mixed the sodium bicarbonate solutions and the local anesthetic solutions in open beakers or graduated cylinders.45,86,87
A similar problem controlling CO2 loss is presented where a disposable tuberculin syringe is used to draw up and mix the anesthetic and the bicarbonate solutions from multi-dose vials. This appears to be a commonly employed compounding process for alkalinizing local anesthetics.88 Typically after mixing, the same syringe is used to deliver the injection.89,90 However, DeLuca and Kowalsky91 noted that CO2 easily permeates through most plastic containers, including polyethylene, polypropylene, polystyrene, and polyvinylchloride syringes. The rate of loss is a function of the density of the plastic, the surface area ratio of the container, time, and temperature. It is, therefore, quite difficult to assess the level of CO2 loss that might occur when alkalinizing local anesthetics using even a properly sealed plastic mixing vessel. Optimally, both the mixing vessel and the fluid path would be composed of materials not permeable to CO2 in order to maximize the CO2 retained in the bolus of the injection.
In this study, the mixing vessel was the anesthetic cartridge itself, which is made of glass (impermeable to CO2), as was the stainless steel needle that served as the fluid path for delivering the injection.
A final observation during preparation for this study should be noted. The investigators noticed that within approximately 90 seconds of adding sodium bicarbonate to a cartridge of lidocaine/epinephrine, visible bubbles began to form and adhere to the sides of the cartridge. It is presumed that these were microscopic bubbles of free CO2 that had begun to aggregate to form macroscopic bubbles. These bubbles continued to grow over time. It was also noted that, when the plunger was driven forward to empty the cartridge, the plunger swept these bubbles together in a squeegee effect and that these bubbles remained in the neck of the anesthetic cartridge when it was emptied; hence, they would not be part of the injection. Based on this observation, the study protocol required that the alkalinized cartridges not be compounded in advance, but rather they would be prepared and delivered to the operatory only when the dentist was ready to deliver the injection.
Adsorption of the Active Form of the Anesthetic
The literature also suggests that the choice of mixing vessel used for alkalinization may impact the amount of de-ionized anesthetic that makes it into the bolus of the injection. A series of Japanese studies demonstrated that the de-ionized form of lidocaine tends to adsorb (adhere) to silicon and other plastics.92-94 To measure the effects of adsorption when lidocaine was held in containers made of plastic, Takakura and colleagues tested the concentration of samples of pH 7.4 lidocaine and pH 5.5 lidocaine placed in silicon plastic infusion balloons for 30 minutes.92 The authors hypothesized that the pH 7.4 sample, having a much greater de-ionized fraction (24.1% vs. 0.4%), would show a higher loss of anesthetic concentration after identical exposure to the balloons. The starting anesthetic concentration in each tested solution was 1%. After 30 minutes, the concentration of the pH 7.4 sample had dropped by one fourth, to 0.8%. The measurements suggest that nearly all the de-ionized anesthetic was lost to adsorption. In the pH 5.5 sample, which contained virtually no de-ionized anesthetic to begin with, the testing showed no measurable change in concentration. The authors concluded that only the de-ionized fraction adsorbed to the container, and that if higher pH anesthetic solutions were exposed to plastics, they would always suffer more adsorption than more acidic solutions.
Mizogami and colleagues wrote that alkalinization should not be used where the alkalinized anesthetic will interact with synthetic resins and plastics, such as in IV fluid containers, delivery sets, and syringes, because the alkalinized local anesthetics may adsorb to the surfaces of these medical devices, and the dosage of anesthetic delivered will be less than intended.93
Satake and colleagues determined that adsorption occurred with silicon plastics, but that adsorption was absent when Teflon was used.94 In the present study, the fluid path of the mixing and delivery armamentarium contained stainless steel and glass components without any plastics. None of these materials are known to be permeable to CO2 or to adsorb de-ionized anesthetic. Delivery of the alkalinized lidocaine epinephrine solution occurred almost immediately after alkalinization.
Conclusions
This study demonstrated a statistically significant improvement in onset of surgical analgesia (P = 0.001) by precisely alkalinizing lidocaine with epinephrine solution and delivering the alkalinized solution using an optimized method and armamentarium. In this study, 70% of the participants receiving alkalinized lidocaine with epinephrine achieved pulpal analgesia in 2 minutes or less. This normally takes 15 minutes, suggesting that the traditional wait times and routines that have been built around slower anesthetic onset might be reconsidered. That analysis would require further study.
The participants in this study also rated injections with alkalinized lidocaine/epinephrine as more comfortable than injections with non-alkalinized lidocaine/epinephrine
(P = 0.013). This difference approached statistical significance. Participants were seven times more likely to report a completely pain-free injection with the alkalinized anesthetic than with the non-alkalinized anesthetic. This difference was statistically significant (P = 0.004).
Practitioners can expect that alkalinizing cartridges of lidocaine/epinephrine immediately before delivering the anesthetic will hasten onset of analgesia and make it more likely that they will deliver a pain-free injection.
References
1. Haas DA. An update on local anesthetics in dentistry. J Can Dent Assoc. 2002;68(9):546-551.
2. Bowles WH, Frysh H, Emmons R. Clinical evaluation of buffered local anesthetic. Gen Dent. 1995;43(2):182-186.
3. Burns CA, Ferris G, Feng C, et al. Decreasing the pain of local anesthesia: A prospective double-blind comparison of buffered, premixed 1% lidocaine with epinephrine versus 1% lidocaine freshly mixed with epinephrine. J Am Acad Dermatol. 2006;54(1):128-131.
4. Brogan GX, Giarrusso E, Hollander JE, et al. Comparison of plain, warmed, and buffered lidocaine for anesthesia of traumatic wounds. Ann Emerg Med. 1995;26(2):121-125.
5. Carvalho B, Fuller A, Brummel C, Cohen SE. Local infiltration of epinephrine-containing lidocaine with bicarbonate reduces superficial bleeding and pain during labor epidural catheter insertion: A randomized trial. Int J Obstet Anesth. 2007;16(2):116-121.
6. Christoph RA, Buchanan L, Begalla K, Schwartz S. Pain reduction in local anesthetic administration through pH buffering. Ann Emerg Med. 1988;17(2):117-120.
7. Colaric KB, Overton DT, Moore K. Pain reduction in lidocaine administration through buffering and warming. Am J Emerg Med. 1998;16(4):353-356.
8. Fitton AR, Ragbir M, Milling MA. The use of pH adjusted lignocaine in controlling operative pain in the day surgery unit: a prospective, randomised trial. Br J Plast Surg. 1996;49(6):404-408.
9. Friedman HE, Jules KT, Springer K, Jennings M. Buffered lidocaine decreases the pain of digital anesthesia in the foot. J Am Podiatr Med Assoc. 1997;87(5):219-223.
10. Krause RS, Moscatti R, Filice M, et al. The effect of injection speed on the pain of lidocaine infiltration. Acad Emerg Med. 1997;4(11):1032-1035.
11. Lugo-Janer G, Padial M, Sanchez JL. Less painful alternatives for local anesthesia. J Dermatol Surg Oncol. 1993;19(3):237-240.
12. Martin AJ. pH-adjustment and discomfort caused by the intradermal injection of lignocaine. Anaesthesia. 1990;45(11):975-978.
13. Masters JE. Randomised control trial of pH buffered lignocaine with adrenaline in outpatient operations. Br J Plast Surg. 1998;51(5):385-387.
14. Matsumoto AH, Reifsnyder AC, Hartwell GD, et al. Reducing the discomfort of lidocaine administration through pH buffering. J Vasc Intervent Radiol. 1994;5(1):171-175.
15. McGlone R, Bodenham A. Reducing the pain of intradermal lignocaine injection by pH buffering. Emerg Med J. 1990;7(2):65-78.
16. McKay W, Morris R, Mushlin P. Sodium bicarbonate attenuates pain on skin infiltration with lidocaine, with or without epinephrine. Anesth Analg. 1987;66(6):572-574.
17. Metzinger SE, Bailey DJ, Boyce RG, Lyons GD. Local anesthesia in rhinoplasty: A new twist? Ear Nose Throat J. 1992;71(9):405-406.
18. Metzinger SE, Rigby PL, Bailey DJ, Brousee RG. Local anesthesia in blepharoplasty: A new look? South Med J. 1994;87(2):225-227.
19. Nakyama M, Munemura Y, Kanaya N, et al. Efficacy of alkalinized lidocaine for reducing pain on intravenous and epidural catheterization. J Anesth. 2001;15(4):201-203.
20. Nelson AL. Neutralizing pH of lidocaine reduces pain during Norplant system insertion procedure. Contraception. 1995;51(5):299-301.
21. Nuttal GA, Barnett MR, Smith RL, et al. Establishing intravenous access: a study of local anesthetic efficacy. Anesth Analg. 1993;77(5):950-953.
22. Ong EL, Lim NL, Koay CK. Towards a pain-free venipuncture. Anaesthesia. 2000;55(3):260-262.
23. Palmon SC, Lloyd AT, Kirsch JR. The effect of needle gauge and lidocaine pH on pain during intradermal injection. Anesth Analg. 1998;86(2):379-381.
24. Ricktsmeier AJ, Hatcher JW. Buffered lidocaine for skin infiltration prior to hemodialysis. J Pain Symptom Manage. 1995;10(3):198-203.
25. Ruegg TA, Curran CR, Lamb T. Use of buffered lidocaine in bone marrow biopsies: a randomized controlled trial. Oncol Nurs Forum. 2009;36(1):52-60.
26. Samdal F, Arctander K, Skolleborg KC, Amland PF. Alkalinization of lignocaine-adrenaline reduces the amount of pain during subcutaneous injection of local anesthetic. Scand J Plast Reconstr Hand Surg. 1994;28(1):33-37.
27. Sapin P, Petrozzi R, Dehmer GJ. Reduction in injection pain using buffered lidocaine as a local anesthetic before cardiac catheterization. Cathet Cardiovasc Diagn. 1991:23(2):100-102.
28. Scarfone RJ, Jasani M, Gracely EJ. Pain of local anesthetics: rate of administration and buffering. Ann Emerg Med. 1998;31(1)36-40.
29. Stewart JH, Cole GW, Klein JA. Neutralized lidocaine with epinephrine for local anesthesia. J Dermatol Surg Oncol. 1989;15(10):1081-1083.
30. Talu H, Elibol O, Yanyali A, et al. Effect of warming and buffering lidocaine on pain during facial anesthesia. Annals of Ophthalmology. 2001;33(1):43-47.
31. Vossinakis IC, Stavroulaki P, Paleochorlidis I, Badras LS. Reducing the pain associated with local anaesthetic infiltration for open carpal tunnel decompression. J Hand Surg Br. 2004;29(4):399-401.
32. Watts AC, Gaston P, Hooper G. Randomized trial of buffered versus plain lidocaine for local anaesthesia in open carpal tunnel decompression. J Hand Surg Br. 2004;29(1):30-31.
33. Xia Y, Chen E, Tibbits DL, et al. Comparison of effects of lidocaine hydrochloride, buffered lidocaine, diphenhydramine, and normal saline after intradermal injection. J Clin Anesth. 2002;14(5):339-343.
34. Yang CH, Hsu HC, Shen SC, et al. Warm and neutral tumescent anesthetic solutions are essential factors for a less painful injection. Dermatol Surg. 2006;32(9):1119-1123.
35. Yiannakapolous CK. Carpal ligament decompression under local anaesthesia: the effect of lidocaine warming and alkalinisation on infiltration pain. J Hand Surg Br. 2004;29(1)32-34.
36. Younis I, Bhutani RP. Taking the ‘ouch’ out – effect of buffering commercial xylocaine on infiltration and procedure pain – a prospective, randomised, double-blind, controlled trial. Ann R Coll Surg Engl. 2004;86(3):213-217.
37. Cepeda MS, Tzortzopulou A, Thackrey M, et al. Adjusting the pH of lidocaine for reducing pain on injection. Cochrane Database Syst Rev. 2010;12:CD006581.
38. Meit SS, Yasek V, Shannon CK, et al. Techniques for reducing anesthetic injection pain. An interdisciplinary survey of knowledge and application. J Am Dent Assoc. 2004;135(9):1243-1251.
39. Cornelius P, Kendall J, Meek S, Rajan R. Alkalinisation of lignocaine to reduce the pain of digital nerve blockade. J Accid Emerg Med. 1996;13(5):339-340.
40. Al-Sultan FA, Fathie WKh, Hamid RS. A clinical evaluation on the alkalization of local anesthetic in periapical surgery. Al-Rafidian Dent J. 2006;6(1):71-77.
41. Kim TH, Kim KW, Kim CH. A clinical study of anesthetic efficacy of alkalinizing lidocaine in inferior alveolar nerve blocks. J Korean Assoc Maxillofac Plast Reconstr Surg. 2005;27(3):276-282.
42. Narahashi T, Frazier T, Yamada M. The site of action and active form of local anesthetics. I. Theory and pH experiments with tertiary compounds. J Pharmacol Exp Ther. 1970;171(1):32-44.
43. Strichartz GR. Current concepts of the mechanism of action of local anesthetics. J Dent Res. 1981;60(8):1460-1470.
44. Hondrum SO, Ezell JH. The relationship between pH and concentrations of antioxidants and vasoconstrictors in local anesthetic solutions. Anesth Prog. 1996;43(3):85-91.
45. Crews JC, Clark RB. Effect of alkalinization on the pH of local anesthetic solutions. Anesth Analg. 1987;66(11):1203.
46. De Jong RH, Cullen SC. Buffer-demand and pH of local anesthetic solutions containing epinephrine. Anesth. 1963;24(6):801-807.
47. Fernandez C. A prospective, randomized, double-blind comparison of bupivacaine and lidocaine for inferior alveolar nerve blocks. J Endod. 2005;31(7):499-503.
48. Childers M, Reader A, Nist R, et al. Anesthetic efficacy of the periodontal ligament injection after an inferior alveolar nerve block. J Endod. 1996;22(6):317-320.
49. Clark K, Reader A, Beck M, Meyers WJ. Anesthetic efficacy of an infiltration in mandibular anterior teeth following an inferior alveolar nerve block. Anesth Prog. 2002;49(2):49-55.
50. Dunbar D, Reader A, Nist R, et al. Anesthetic efficacy of the intraosseous injection after an inferior alveolar nerve block. J Endod. 1996;22(9):481-486.
51. Foster W, Drum M, Reader A, Beck M. Efficacy of buccal and lingual infiltrations following an inferior alveolar nerve block in mandibular posterior teeth. Anesth Prog. 2007;54;(4):163-169.
52. Gallatin E, Stabile P, Reader A, et al. Anesthetic efficacy and heart rate effects of the intraosseous injection of 3% mepivacaine after an inferior alveolar nerve block. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;89;(1):83-87.
53. Goldberg S, Reader A, Drum M, et al. Comparison of the anesthetic efficacy of the conventional inferior alveolar, Gow-Gates, and Vazirani-Akinosi techniques. J Endod. 2008;34(11):1306-1311.
54. Hannan L, Reader A, Nist R, et al. The use of ultrasound for guiding needle placement for inferior alveolar nerve blocks. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;87(6):658-665.
55. Hung PC, Chang HH, Yang PJ, et al. Comparison of the Gow-Gates mandibular block and inferior alveolar nerve block using a standardized protocol. J Formos Med Assoc. 2006;105(2):139-146.
56. Kanaa MD, Meechan JG, Corbell IP, Whitworth JN. Speed of injection influences efficacy of inferior alveolar nerve blocks, a double-blind randomized controlled trial in volunteers. J Endod. 2006;32(10):919-923.
57. Lai TN, Lin CP, Kok SH, et al. An evaluation of mandibular block using a standardized method. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102(4):462-468.
58. Mikesell P, Nusstein J, et al. A comparison of articaine and lidocaine for inferior alveolar nerve blocks. J Endod. 2005;31(4):265-270.
59. Nist RA, Reader A, Beck M, Meyers WJ. An evaluation of the incisive nerve block and combination inferior alveolar and incisive nerve blocks in mandibular anesthesia. J Endod. 1992;18(9):455-459.
60. Steinkruger G, Nusstein J, Reader A, et al. The significance of needle bevel orientation in achieving a successful inferior alveolar nerve block. J Am Dent Assoc. 2006;137(12):1685-1691.
61. Willett J, Reader A, Drum M, et al. The anesthetic efficacy of diphenhydramine and the combination diphenhydramine/lidocaine for the inferior alveolar nerve block. J Endod. 2008;34(12):1446-1450.
62. Chaney MA, Kerby R, Reader A, et al. An evaluation of lidocaine hydrocarbonate compared with lidocaine hydrochloride for inferior alveolar nerve block. Anesth Prog. 1991;38(6):212-216.
63. Dagher FB, Yared GM, Machtou P. An evaluation of 2% lidocaine with different concentrations of epinephrine for inferior alveolar nerve block. J Endod. 1997;23(3):178-180.
64. Hinkley SA, Reader A, Beck M, Meyers WJ. An evaluation of 4% prilocaine with 1:200,000 epinephrine and 2% mepivacaine with 1:20,000 levonnordefrine compared with 2% lidocaine with 1:100,000 epinephrine for inferior alveolar nerve blocks. Anesth Prog. 1991;38(3):84-89.
65. McLean C, Reader A, Beck M, Meyers WJ. An evaluation of 4% prilocaine and 3% mepivaciane compared with 2% lidocaine (1:100,000 epinephrine) for inferior alveolar nerve block. J Endod. 1993;19(3):146-150.
66. Vreeland D, Reader A, Beck M, et al. An evaluation of volumes and concentrations of lidocaine in human inferior alveolar nerve blocks. J Endod. 1989;15(1):6-12.
67. Wali M, Reader A, Beck M, Meyers WJ. Anesthetic efficacy of lidocaine and epinephrine in human IAN block. J Endod. 1988;14(4):193.
68. Yared GM, Dagher FB. Evaluation of lidocaine in human inferior alveolar nerve block. J Endod. 1997;23(9):575-578.
69. Haase A, Reader A, Nusstein J, et al. Comparing anesthetic efficacy of articaine versus lidocaine as a supplemental buccal infiltration of the mandibular first molar after an inferior alveolar nerve block. J Am Dent Assoc. 2008;139(9):1228-1235.
70. Alonso PE, Perula LA, Rioja LF. Pain-temperature relation in the application of local anesthesia. Br J Plast Surg. 1993;46(1):76-78.
71. Hanna MN, Elhassan A, Veloso PM, et al. Efficacy of bicarbonate in decreasing pain on intradermal injection of local anesthetics: a meta-analysis. Reg Anesth Pain Med. 2009;34(2):122-125. [citing Perl ER. Cutaneous polymodal receptors: characteristics and plasticity. Prog Brain Res. 1996;113:21-37.]
72. Brazeau GA, Cooper B, Svetic KA, et al. Current perspectives on pain upon injection of drugs. J Pharma Sci 1998;87(6):667-677.
73. Morris R, McKay W, Mushlin P. Comparison of pain associated with intradermal and subcutaneous infiltration with various local anesthetic solutions. Anesth Analg. 1987;66(11):1180-1182.
74. Brogan GX, Gearrusso E, Hollander JE, et al. Comparison of plain, warmed, and buffered lidocaine for anesthesia of traumatic wounds. Ann Emerg Med. 1995;26(2):121-125.
75. Catchlove RFH. The influence of CO2 and pH on local anesthetic action. J Pharmacol Exp Ther. 1972;181(2):298-309.
76. Bokesch PM, Raymond SA, Strichartz GR. Dependence of lidocaine potency on pH and PCO2. Anesth Analg. 1987;66(1):9-17.
77. Raymond S, Wong K, Strichartz G. Mechanisms for potentiation of local anesthetic action by CO2: bicarbonate solutions. Anesth. 1989;71(suppl):A711.
78. Condouris GA, Shakalis A. Potentiation of the nerve-depressant effect of local anaesthetics by carbon dioxide. Nature. 1964;204:57-59.
79. Curatolo M, Petersen-Felix S, Arendt-Nielsen L, et al. Adding sodium bicarbonate to lidocaine enhances the depth of epidural blockade. Anesth Analg. 1988;86(2):341-347.
80. Bromage PR. Improved conduction blockade in surgery and obstetrics: carbonated local anesthetics. Can Med Assoc J. 1967;97(23):1377-1384.
81. Wong K, Strichartz GR, Raymond SA. On the mechanisms of potentiation of local anesthetics by bicarbonate buffer: drug structure-activity studies on isolated peripheral nerve. Anesth Analg. 1993;76(1):131-143.
82. Liepert DJ, Douglas MJ, McMorland GH, et al. Comparison of lidocaine CO2, two per cent lidocaine hydrochloride and pH adjusted lidocaine hydrochloride for caesarean section anaesthesia. Can J Anaesth. 1990;37(3):333-336.
83. Ibusuki S, Katsuki H, Takasaki M. The effects of extracellular pH with and without bicarbonate on intracellular procaine concentrations and anesthetic effects in crayfish giant axons. Anesthesiology. 1998;88(6):1549-1577.
84. Hille B. Local anesthetics: hydrophilic and hydrophobic pathways for the drug-receptor reaction. J Gen Physiol. 1977;69(4):497-515.
85. Ackerman WE III, Ware TR, Juneja M. The air-liquid interface and the pH and PCO2 of alkalinized local anaesthetic solutions. Can J Anaesth. 1992;39(4):387-389.
86. Peterefreund RA, Datta S, Ostheimer GW. pH adjustment of local anesthetic solutions with sodium bicarbonate: laboratory evaluation of alkalinization and precipitation. Reg Anesth. 1989;14(6):265-270.
87. Schwab RA, Watson WA. Bicarbonate buffering of local anesthetics. Am J Emerg Med. 1996;14(3):339.
88. Ikuta PT, Raza SM, Durrani Z, et al. pH adjustment schedule for the amide local anesthetics. Reg Anesth. 1989;14(5):229-235.
89. Bedder MD, Kozody R, Craig DB. Comparison of bupivacaine and alkalinized bupivacaine in brachial plexus anesthesia. Anesth Analg. 1988;67(1):48-52.
90. McMorland GH, Douglas MJ, Jeffrey WK, et al. Effect of pH-adjustment of bupivacaine on onset and duration of epidural analgesia in parturients. Can Anaesth Soc J. 1986;33(5):537-541.
91. DeLuca PP, Kowalsky RJ. Problems arising from the transfer of sodium bicarbonate injection from ampules to plastic disposable syringes. Am J Hosp Pharma. 1972;29(3):217-222.
92. Takakura K, Miramatsu I, Miyamoto E, Fukuda S. Adsorption of lidocaine into plastic infusion balloon. Anesth Analg. 2000;91(1):192-194.
93. Mizogami M, Tsuchiya H, Takakura K. Local anesthetics adsorbed into infusion balloon. Anesth Analg. 2004;99(3):764-768.
94. Satake H, Kageyama T, Kaneshina S, et al. Adsorption of lidocaine into plastics resin and its effect on the conduction block of action potential in the squid giant axon membrane. Hiroshima J Anest. 1998;34(4):251-255.
About the Authors
Stanley F. Malamed, dds
Professor of Anesthesia & Medicine
Herman Ostrow School of Dentistry
University of Southern California
Los Angeles, California
Susan Tavana, dds
Resident
Division of Pediatric Dentistry
Department of Orofacial Sciences
University of California San Francisco
San Francisco, California
Mic Falkel, dds
Co-founder
Chief Medical Officer
Onpharma Inc.
Los Gatos, California
Private Practice
Monterey, California



![Figure 5 Figure 5. Shown in red (lidocaine with epinephrine) and gold (articaine with epinephrine) are composite curves for the percentage of patients achieving pulpal analgesia at any time from 0 to 20 minutes, as set out in 21 published studies conducted between 1991–2010, all of which evaluated IANBs. The authors of this study prepared the above composite curves based on previous studies for lidocaine/epinephrine[48-61] and for articaine/epinephrine.[62-69] More than three fourths of these studies were conducted by endodontic students at the Ohio State University under the guidance of Al Reader, Mike Beck, Robert Nist, Melissa Drum, John Nusstein, Joel Weaver, and/or William J. Meyers. These studies consistently implemented a useful, practical, and repeatable measure of analgesia that makes comparisons of success rates and the time course of analgesia across published studies and in the practice of dentistry possible, for which the current authors are sincerely appreciative.](/media/thumbnail/26551/150x150)

