Cutting-Edge Implant Rehabilitation Design and Management: A Tapered Abutment Approach
Xavier Rodríguez, MD, PhD; Xavier Vela, MD, DDS; and Maribel Segalà, MD, DDS
Abstract
Soft-tissue recession around an implant rehabilitation over time has been considered a physiologic phenomenon. The divergent profile of the abutment and the abutment’s dis/reconnections are the most critical predisposing and precipitating factors regarding such gingival recession. Recent publications have discussed how tapered and marginless abutments that allow no disconnections and increase soft-tissue thickness could prevent implant rehabilitations from experiencing gingival recession. The objective of this article is to demonstrate the biologic rationale of tissue behavior surrounding tapered abutments and their clinical application.
Peri-implant soft-tissue recession must be prevented to attain healthy, esthetic results when implants are placed. Paradoxically, physiological soft-tissue recession over time has been described around implant rehabilitations with a divergent emergence profile at the soft-tissue level.1,2 This type of restoration design has been the most commonly used since the beginning of implantology. This hypothetically ideal restoration design assumes that by imitating both the tooth neck shape and function, soft-tissue stabilization would be achieved.3,4
Recent articles in the literature have shown that some gingival coronal migration could be attained with the use of tapered emergence profile abutments.5,6 In fact, by using these tapered abutments or an implant design with a tapered shape at the neck implant level (transmucosal level), not only soft-tissue migration but also hard-tissue migration could be achieved over time.7,8 In this article, the authors will show the biologic rationale of the behavior of tissue surrounding tapered abutments and their clinical application.
Soft- and Hard-Tissue Recession as a Physiological Tendency
To reiterate, both soft- and hard-tissue recession has been related mainly to the anatomic or divergent abutment.2 Several treatments have been proposed for solving this physiological tendency.9 From a prosthetic point of view, for example, the search for more biocompatible and adhesive surfaces has been proposed.10 From a surgical viewpoint, connective tissue grafting to increase soft-tissue thickness has been the most successful surgical treatment.11
Conceptually, it is presumed that the more tissue thickness there is, the more tissue stability will be achieved. The question is whether there is an implant rehabilitation design that exerts on the peri-implant tissues the same effect that the treatments proposed for solving tissue recession do. In general, the implant anatomic abutment replaces the tooth neck area, and its function is primarily to create the emergence profile, whereas the function of the tooth neck is to hold up the connective tissue fibers on the tooth.
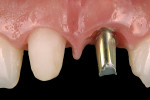
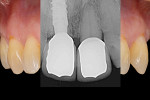
Recently, an implant prosthetic design and management approach has been proposed that is intended to not only preserve the implant rehabilitation from recession, but also improve the tissue level (hard and soft tissue) in both the early and late stages of healing (Figure 1).6-8 The principle of this abutment design is based on interplay with the surrounding tissues. The healing of the tissues would be enhanced by the abutment design (from which the acronym “HEAD” is derived). Basically, this type of restoration takes advantage of the features of second healing intention wounds (namely, growth and contraction) to make the tissue thicker and prone to migrate in the coronal direction over time. Understanding the histomorphogenesis surrounding the HEAD abutment approach and comparing this with conventional abutment design will help clinicians grasp how this rehabilitation works.
Clinical Approach and Histomorphogenesis: Biological Events Addressed by the HEAD Prosthetic Design and Protocol
Each stage of any implant rehabilitation protocol exerts a specific tissue response that modifies the tissue volume and esthetics. To achieve healthy and good esthetic results, all stages must be well-managed. After implant placement, whether in one or two stages, the soft and hard tissues heal. Wound healing is a coordinated and sequentially organized mechanism that has four phases: hemostasis, inflammatory phase, proliferative phase, and remodeling phase.5 To clarify the influence of the abutment shape on tissue behavior, the relationship between the clinical approach and biological events when following the HEAD protocol will be discussed.
Implant Placement
The implant should be considered the apical extension of the restoration, and the preferred design of the restoration should guide the surgical implant placement.12 Following the HEAD protocol, a platform-switching rehabilitation design with a tapered abutment shape will be chosen to preserve not only the hard-tissue level but also the soft-tissue level.13 The implant platform level should be placed 1 mm subcrestally.14 This implant platform position allows the periosteum and connective tissue to stabilize on the implant platform surface from the time tissue healing begins.11,14,15 After implant placement, if the tissue volume and implant stability are good, placement of the definitive abutment is recommended. If a submerge technique is preferred, a 2-mm high abutment is recommended for ease in exposing the implant.
Implant Exposure
After osseointegration, the implant must be exposed. The abutment dis/reconnections are a main source of bone resorption. To prevent or minimize early peri-implant bone resorption, placement of the definitive abutment is advised. To place the definitive abutment at the time of implant exposure, the implant impressions should have been taken at the time of implant placement. Also, because of the variability of the gingival margin level, the abutment should be marginless. Once the implant has been exposed, soft-tissue healing begins. Clinical approach and tissue biology must reinforce each other not only during the early stages of healing, but always.
Hemostasis
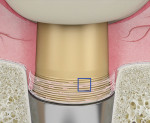
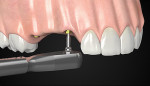
Hemostasis is the stage in which hemorrhaging stops and the scaffold is prepared for tissue regeneration. The abutment and crown design should leave space for the clot during this phase. The standard anatomic abutment (divergent shape) does not leave sufficient room for the clot and, therefore, would be prone to promote gingival recession.16 To give the clot an opportunity to occupy the implant platform discrepancy between the abutment and implant, the implant platform would need to have been placed subcrestally.5,11,14,15 During healing, the space provided for the blood clot will transform into connective tissue, creating a 3-dimensional network around the abutment.17
The second important part of this phase of healing is promoting the stability of the blood clot (Figure 2 and Figure 3). Use of a standard anatomic abutment and crown makes it impossible to cultivate a “chamber” where the clot can mature. Conversely, the tapered shape of the abutment and the emergence profile of the provisional crown when using HEAD abutment design enable the clot to be undisturbed in this chamber.18 The apical part of the crown maintains contact with the gingival margin, sealing the space. In this way, the clot remains undisturbed and protected by the sealing space formed by the soft tissue, bone, implant platform, abutment, and bottom of the crown.
In addition, recession has long been considered a result of predisposing and precipitating factors. Regarding implants, the most important precipitating factor is the abutment dis/reconnections. Avoiding abutment dis/reconnections diminishes soft-tissue collapse and bone resorption.11,19,20 Therefore, a non-dis/reconnection abutment protocol is recommended.
Inflammatory Stage
Ten minutes after implant exposure, the second healing stage—the inflammatory stage—begins and continues for the next 4 to 7 days. The blood clot is infiltrated by several polymorphonuclear leukocytes among a dense fiber network. The use of antibiotic and anti-inflammatory therapy is advisable during this stage. Fortunately, after a few days, the proliferative phase takes place.
Proliferative Phase
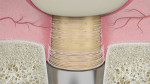
During the proliferative phase the tissues that provide the structural cells and blood supply are repaired. The tissue healing surrounding the implant restoration heals by second intention. The main characteristic of the second healing intention wound is the capability of contracting, which is mediated by the myofibroblasts.21 After 15 days, approximately 70% of the fibroblasts in granulation tissue express alfa-smooth muscle actin.21 Fibroblasts and myofibroblasts grow and fill the space left by the temporary restoration, and new blood vessels accompany them in their growth, encircling the abutment.23 The contraction of the myofibroblasts produces a soft-tissue migration from the wider diameter area of the abutment to the narrower diameter area of the abutment.5 Depending on the shape of the abutment, this contraction will move the tissues apically or coronally.
The use of HEAD abutment design promotes soft-tissue migration toward the coronal area of the restoration instead of the apical area.5 In other words, the abutment directs the contraction energy of the tissues to a coronal tissue movement.5 The stability of the connective tissue essentially depends on the circular fibers.5,14,23 The connective and granulation tissue can prevent the apical migration of the epithelium.24 Furthermore, the presence of a microthreaded surface at the base of the HEAD abutment enhances the contact guidance capability, ie, early fibroblast adhesion and activation (Figure 4 and Figure 5).25 By using tapered abutments, the apical migration of the epithelium is stopped by the connective tissue at the crown surface level instead of at the lateral side of the implant platform.5 Eventually, if the peri-implant tissue remains undisturbed, the remodeling stage will take place after 6 weeks of healing and will continue indefinitely (Figure 6).
Remodeling Phase
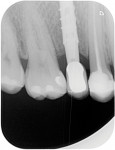
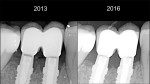
The remodeling phase is when tissue maturation, collagen realignment, and tissue growth occur as a result of homeostasis. Homeostasis comes from the Greek and means “similar” and “stability.” It is the property of a living organism to maintain its internal stable condition by adapting itself to the changes of the environment. The ideal implant rehabilitation is one that improves the tissue response over time. Several studies show bone resorption and gingival recession on standard restorations over time.2,16 On the other hand, some implant systems show not only crestal bone level preservation but also crestal bone gain over time.26 All of the aforementioned studies showed the transmucosal part of the restoration having a tapered shape.7 The homeostasis of the tissues is mainly related to the forces exerted over them.27
Soft-tissue remodeling—Fibroblasts sense changes in the physical parameters of their extracellular matrix (ECM) environment, transduce mechanical information into chemical information, and integrate these signals with growth factor-derived stimuli to achieve specific changes in gene expression.27 The ECM of connective tissue bears most of the physical loads that act on the mouth. Chewing and speaking generate forces (tension, compression, or shear) on cells and the ECM. This tiny mechanical stimulus can be transduced into chemical signals, eliciting a multitude of cellular responses. Mechanical stress influences cell proliferation and survival of oral fibroblasts (homeostasis). It has been described that the prosthesis design could originate a negative pressure on the tissues and stimulate tissue growth.28 In fact, it has been suggested that the more overcontoured the implant crown is, the more gingival thickness occurs.29 The authors use the term gullwing to describe this. The gingival growth might relate to the tension effect produced by the negative pressure (mechanotransduction) at the area delimited by the gingival margin, the buccal face of the implant crown, and the internal mucosa of the lip.
Hard-tissue remodeling—The first bone–implant contact has been related to implant treatment success.30 A great deal of literature discusses the causes of early and late bone resorption.2,16,31 However, changes on the macrodesign of the restoration at the transmucosal level have been shown to preserve the peri-implant bone level closer than 1.5 mm from the implant–abutment interface.11,13,32,33 The common feature in these studies is the tapered shape of the implant restoration at the transmucosal part of the restoration, regardless of whether this part belongs to the implant (the first thread or implant neck) or the abutment.5,7
The part of the organism that would drive the bone growth and bone repair is the periosteum,34 which has an outer fibrous layer and an inner osteoblastic potential layer. Fibers that emerge from the outer layer of the periosteum join the connective tissue of the gingiva, surround the implant abutment, or merge into the tooth neck cementum. The circular connective tissue fibers migrate from the wider diameter area of the implant abutment to the narrower diameter area.5 The existence of the tapered shape at the level of the transmucosal interface, with a narrower diameter and negative pressure, promotes soft-tissue migration to the coronal implant restoration area. Physical load is very important for connective tissue development, maintenance, and remodeling.27 Furthermore, mechanical stress controls the production and release, by fibroblasts, of inflammatory mediators in bone remodeling. This may suggest that fibers that grow around the implant abutment pull out the periosteum and osteoblastic cells similar to how strings pull puppets (Figure 7).
Discussion
The stability of hard and soft tissues around implant restorations has been recognized as a key factor for long-term implant success. It has been suggested that mimicking the tooth shape could help achieve peri-implant tissue stability.3 However, peri-implant bone resorption and gingival recession around matched implant restorations with anatomical abutments usually have been found over time.2,10
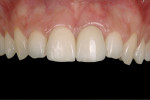
To solve such tissue apical migration, the present authors propose a restoration design and implant protocol based primarily on imitating the tooth function instead of tooth shape. Several studies have found that changing the shape of the transmucosal part of the restoration (abutment) can improve the peri-implant bone level.5-8,13 It has also been shown that the use of tapered abutments might be a key factor in enhancing the stability of the soft tissues more coronally than previously thought.5,6 In addition, another precipitating factor of apical tissue migration has been found: the abutment dis/reconnections.11 For this reason, the authors propose a non-disconnection abutment protocol to attain tissue stability beginning at the start of the healing period. The use of tapered abutments following a disconnection-less clinical protocol (Figure 8 through Figure 13) is described.
Clinical Protocol to Enhance Tissue Stabilization
The HEAD abutment protocol is aimed at promoting peri-implant tissue stabilization; therefore, the proper abutment position and timing of placement is crucial to clinical success.
Implant placement and abutment selection—Before implant placement, the clinician should decide whether the restoration will be cement- or screw-retained. In screwed restorations, the implant axis should follow the axis of the cingulum of the tooth. In cemented restorations, the implant axis should follow the axis from the incisal edge of the tooth. Regarding abutment position, the microthreaded area should be placed subcrestally; consequently, the implant-abutment platform should be placed 1 mm subcrestally. In cases involving bone regeneration, the implant platform should be placed 4 mm to 5 mm apically from the gingival margin. To achieve optimal crown stability, the longest possible abutment should be chosen. Finally, the abutment should be placed at the time of implant exposure, in either the first or second stage of implant surgery.
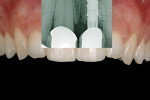
Abutment type and customization (reshaping)—If a screwed abutment is used, at least 5 mm of distance should be left from the top of the abutment to the antagonist crown to allow enough space for the crown and prosthetic screw (Figure 14). For cemented restorations, the shape of the cemented abutment should be modified to leave enough distance from the antagonist crown to achieve adequate metal and porcelain thickness. These abutment changes will be transferred to the technician via a cutting template (Figure 15). Once the definitive abutment has been placed, the appropriate screw torque is applied. The abutment should not be disconnected again. After the prosthetic screw placement, with cemented restorations the access hole for the abutment is filled with Teflon™ tape and covered with composite to protect the gap from bacteria contamination.
Once the abutment is fitted, the crown provisionalization can be considered.
Temporary crown—The technician should have previously prepared a hollowed acrylic resin crown. The biologically oriented preparation technique (BOPT) protocol suggested by Loi29 should be used for the design and development of the provisional crown on the abutment.
Definitive crown—After 2 to 6 months of healing and integration, the definitive restoration should be considered. For a screwed abutment, after the impression copings are fixed onto the abutments, the impression is taken using an open tray (Figure 16). For a cemented abutment, a direct impression onto the abutment must be taken. To provide all the necessary information to the technician, a cutting template should be used (Figure 17 and Figure 18).
Conclusion
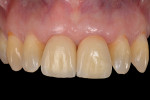
When the HEAD implant restoration design and disconnection-less clinical protocol is used, the biologic width becomes more coronally placed and thicker compared with the biologic width that surrounds standard restorations. As a result, the soft tissues are more resistant to oral insults, and the bone level is preserved and improves over time.
Disclosure
The authors received research support from Sweden & Martina SpA. Images in this article are used with permission from Sweden & Martina SpA.
About the Authors
Xavier Rodríguez, MD, PhD
Visiting Professor
International University of Catalonia (UIC)
Barcelona, Spain
Private Practice
Cambra Clínic & Associates
Barcelona, Spain
Member, Barcelona Osseointegration Research Group (BORG)
Xavier Vela, MD, DDS
Visiting Professor
International University of Catalonia (UIC)
Barcelona, Spain
Private Practice
BORG Center of Viladecans
Barcelona, Spain
Member, BORG
Maribel Segalà, MD, DDS
Private Practice
BORG Center of Viladecans
Barcelona, Spain
Member, BORG
References
1. Small PN, Tarnow DP. Gingival recession around implants : a 1-year longitudinal prospective study. Int J Oral Maxillofac Implants. 2000;15 (4):527-532.
2. Apse P, Zarb GA, Schmitt A, Lewis DW. The longitudinal effectiveness of osseointegrated dental implants. The Toronto study: peri-implant mucosal response. Int J Periodontics Restorative Dent. 1991;11(2):94-111.
3. Marchack CB, Yamashita T. Fabrication of a digitally scanned, custom-shaped abutment: a clinical report. J Prosthet Dent. 2001;85(2):113-115.
4. De Rouck T, Collys K, Cosyn J. Immediate single-tooth implants in the anterior maxilla: a 1-year case cohort study on hard and soft tissue response. J Clin Periodontol. 2008;35(7):649-657.
5. Rodríguez X, Navajas A, Vela X, et al. Arrangement of peri-implant connective tissue fibers around platform-switching implants with conical abutments and its relationship to the underlying bone : a human histologic study. Int J Periodontics Restorative Dent. 2016;36(4): 533-540.
6. Cocchetto R, Canullo L. The “hybrid abutment”: a new design for implant cemented restorations in the esthetic zones. Int J Esthet Dent. 2015; 10(2):186-208.
7. Braun E, Iacono VJ. Tapered tips to think about. Int J Periodontics Restorative Dent. 2006;26(1):7.
8. Urdaneta RA, Leary J, Lubelski W, et al. The effect of implant size 5 × 8 mm on crestal bone levels around single-tooth implants. J Periodontol. 2012; 83(10):1235-1244.
9. Bengazi F, Wennström JL, Lekholm U. Recession of the soft tissue margin at oral implants. A 2-year longitudinal prospective study. Clin Oral Implants Res. 1996;7(4):303-310.
10. Welander M, Abrahamsson I, Berglundh T. The mucosal barrier at implant abutments of different materials. Clin Oral Implants Res. 2008;19(7):635-641.
11. Rodríguez X, Vela X, Méndez V. The effect of abutment dis/reconnections on peri-implant bone resorption: a radiologic study of platform-switched and non-platform-switched implants placed in animals. Clin Oral Implants Res. 2013;24(3):305-311.
12. Al-Sabbagh M. Implants in the esthetic zone. Dent Clin North Am. 2006; 50(3):391-407.
13. Atieh MA, Ibrahim HM, Atieh AH. Platform switching for marginal bone preservation around dental implants: a systematic review and meta-analysis. J Periodontol. 2010;81(10):1350-1366.
14. Rodríguez X, Vela X, Calvo-Guirado JL, et al. Effect of platform switching on collagen fiber orientation and bone resorption around dental implants: a preliminary histologic animal study. Int J Oral Maxillofac Implants. 2012;27(5):1116-1122.
15. Weng D, Nagata MJ, Leite CM, et al. Influence of microgap location and configuration on radiographic bone loss in nonsubmerged implants: an experimental study in dogs. Int J Prosthodont. 2011;24(5):445-452.
16. Cosyn J, Eghbali A, Hermans A, et al. A 5-year prospective study on single immediate implants in the aesthetic zone. J Clin Periodontol. 2016;43(8):702-709.
17. Wikesjö UM, Nilvéus R. Periodontal repair in dogs: effect of wound stabilization on healing. J Periodontol. 1990;61(12):719-724.
18. Degidi M, Daprile G, Nardi D, Piattelli A. Immediate provisionalization of implants placed in fresh extraction sockets using a definitive abutment: the chamber concept. Int J Periodontics Restorative Dent. 2013;33(5):559-565.
19. Abrahamsson I, Berglundh T, Sekino S, Lindhe J. Tissue reactions to abutment shift: an experimental study in dogs. Clin Implant Dent Relat Res. 2003;5(2):82-88.
20. Abrahamsson I, Berglundh T, Lindhe J. The mucosal barrier following abutment dis/reconnection. An experimental study in dogs. J Clin Periodontol. 1997;24(8):568-572.
21. Häkkinen L, Uitto VJ, Larjava H. Cell biology of gingival wound healing. Periodontol 2000. 2000;24:127-152.
22. Makigusa K. Histologic comparison of biologic width around teeth versus implant: The effect on bone preservation. J Implant Reconstr Dent. 2009;1(1):20-24.
23. Schierano G, Ramieri G, Cortese M, et al. Organization of the connective tissue barrier around long-term loaded implant abutments in man. Clin Oral Implants Res. 2002;13(5):460-464.
24. Rompen E, Domken O, Degidi M, et al. The effect of material characteristics, of surface topography and of implant components and connections on soft tissue integration: a literature review. Clin Oral Implants Res. 2006;17(suppl 2):55-67.
25. Guillem-Marti J, Delgado L, Godoy-Gallardo M, et al. Fibroblast adhesion and activation onto micro-machined titanium surfaces. Clin Oral Implants Res. 2013;24(7):770-780.
26. Urdaneta RA, Seemann R, Dragan IF, et al. A retrospective radiographic study on the effect of natural tooth-implant proximity and an introduction to the concept of a bone-loading platform switch. Int J Oral Maxillofac Implants. 2014;29(6):1412-1424.
27. Chiquet M, Gelman L, Lutz R, Maier S. From mechanotransduction to extracellular matrix gene expression in fibroblasts. Biochim Biophys Acta. 2009;1793(5):911-920.
28. Payne AG, Solomons YF, Tawse-Smith A, Lownie JF. Inter-abutment and peri-abutment mucosal enlargement with mandibular implant overdentures. Clin Oral Impl Res. 2001;12(2):179-187.
29. Loi I, Di Felice A. Biologically oriented preparation technique (BOPT): a new approach for prosthetic restoration of periodontically healthy teeth. Eur J Esthet Dent. 2013;8(1):10-23.
30. Misch CE, Perel ML, Wang HL, et al. Implant success, survival, and failure: the International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant Dent. 2008;17(1):5-15.
31. Oh TJ, Yoon J, Misch CE, Wang HL. The causes of early implant bone loss: myth or science? J Periodontol. 2002;73(3):322-333.
32. Warren P, Chaffee N, Felton DA, Cooper LF. A retrospective radiographic analysis of bone loss following placement of TiO2 grit-blasted implants in the posterior maxilla and mandible. Int J Oral Maxillofac Implants. 2002;17(3):399-404.
33. Vela-Nebot X, Rodríguez-Ciurana X, Rodado-Alonso C, Segalà-Torres M. Benefits of an implant platform modification technique to reduce crestal bone resorption. Implant Dent. 2006;15(3):313-320.
34. Saulacic N, Schaller B, Iizuka T, et al. Analysis of new bone formation induced by periosteal distraction in a rat calvarium model. Clin Implant Dent Relat Res. 2013;15(2):283-291.