Dental Space Deficiency Syndrome: An Anthropological Perspective
Colin S. Richman, DMD, BDS, LDS, RCS, H. Dip Dent.
Abstract: A new syndrome in dentistry, the dental space deficiency syndrome is proposed in this article. Signs and symptoms of this entity may include one or more of the following clinical dental features: tooth crowding, gingival recession, tooth impactions, rapid resorption of facial alveolar bony plates following premature tooth loss, dentally oriented sleep disorders, extended orthodontic treatment time, and malocclusion relapse following orthodontic therapy. These oral conditions, individually or collectively, seem to be associated with both genetic and functional factors. From an anthropological-functional perspective, the human jaws (basal bone and/or alveolar bone) have been shrinking. This results in a three-dimensional discrepancy between jawbone and tooth volumes, which are genetically determined. Consequently, the reduced volume of alveolar bone is not adequately able to accommodate the associated genetically determined dentition in functional and esthetic harmony. This paper describes the common etiology for the conditions listed above, namely the discrepancy between alveolar bone volume (essentially determined by functionality), and associated tooth volume (essentially determined by genetics), when considered in a three-dimensional perspective.
Both genetic and functional factors1,2 seem to be associated with discrepant tooth size and available alveolar bone phenomenon. From an evolutionary perspective, current anthropologic evidence suggests the volume of human jawbones is decreasing, when evaluated in three dimensions.3,4-9
Conversely, studies evaluating tooth size in monozygotic and dizygotic twins have suggested actual tooth morphology and, thus, tooth size appears to be predominantly determined by genetics.10,11 Thus, the evolving discrepancy between tooth size and available alveolar jawbone size, when evaluated in three dimensions, results in a net deficiency of available alveolar bone unable to naturally accommodate the human dentition. Using cone-beam computed tomography (CBCT) imaging, Richman has demonstrated the phenomenon of tooth size versus alveolar bone discrepancies, when viewing alveolar bone anatomy in three dimensions. This phenomenon is particularly evident in sagittal plane sections.12,13
Texts describing human dental, head, and neck anthropologic changes suggest the volume of the human jaws are decreasing.3,14-16 This is associated with the concept that as we changed from prehistoric hunters to gatherers, farmers, and subsequently utilizers of industrialized food production, the human jaws began shrinking due to functional disuse.3
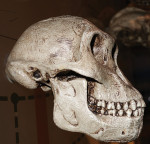
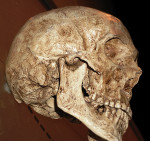
Our foods have become highly processed. Therefore, large human powerful jaws are no longer necessary for human survival from a masticatory or defensive perspective. However, the cranium and frontal lobe of the forebrain is increasing at the expense of the human faces4-8 (Figure 1 and Figure 2). This particularly relates to our prefrontal cortices, the gray matter of the anterior part of the frontal lobe, which is highly developed in humans and plays a role in the regulation of complex cognitive, emotional, and behavioral functions. Relative to the maxilla and mandible, less functionality over time is resulting in gradual atrophy of these bones, with less capacity to accommodate our genetically determined teeth.
A new syndrome for dentistry, dental space deficiency syndrome (DSDS) is proposed in this article. Clinical signs and symptoms of this entity may include one or more of the following conditions: orthodontic tooth crowding; gingival recession; tooth impactions, especially third molars; rapid resorption of facial alveolar plate, following premature tooth extractions; dentally oriented sleep disorders; extended orthodontic treatment times; and relapse following orthodontic therapy.
Dental Maladies Possibly Attributed to DSDS
Studies of human inhabitants of metropolitan cities suggest a high incidence of individuals demonstrating orthodontic tooth crowding.17-25 A US Public Health Service Report suggests 75% of American children have some degree of malocclusion.23,26,27 Based on the author’s observations and studies of mammalian and primitive skulls, he believes these problems are not observed in native human communities or animals living in their natural environments and eating their natural diets.
Relative to the United States and other metropolitan cities, gingival recession occurs in more than 58% of the total population. Surveyed population reports identify significant evidence of non-periodontitis–induced gingival recession.25-31 Tooth impactions, especially third molars, occurred in more than 58% of surveyed Australian populations, 73% of surveyed European populations, and in excess of 50% of surveyed American populations.32 Post-orthodontic treatment relapse, in surveyed populations, may be present in more than 80% of patients, possibly many years following treatment completion.23,33-36 Rapid loss of alveolar facial bony plates following tooth extractions is a routine finding, especially in the facial bony plate.37-41
Dentally related sleep disturbances are substantial and significant in surveyed communities.42
These phenomena, individually or collectively, seem to be associated with either a discrepancy between available alveolar bone and basal bone volume and tooth volume, (when considered in three dimensions), or are iatrogenically induced due to protracted orthodontic treatment associated with adapting larger teeth into a smaller jawbone capacity. Three case studies are presented to illustrate the signs and symptoms of DSDS.
Patient 1
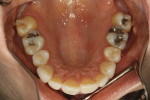
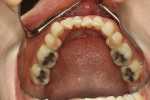
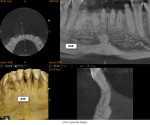
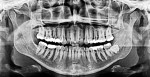
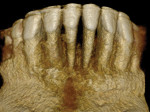
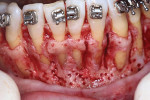
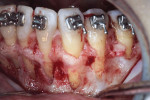
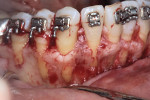
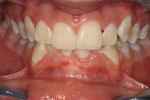
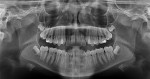
A healthy 38-year-old female was referred to the author’s specialty periodontal office for corrective mucogingival therapy. She was also referred to a local orthodontist, skilled in the principles of periodontally accelerated osteogenic orthodontics (PAOO),43,44 for correction of her malocclusion. Treatment was indicated both for esthetic and functional reasons. She reported having undergone previous orthodontic therapy as a teenager. At that time, treatment included removal of 4 bicuspids and 4 third-molar teeth (Figure 3). Clinical images demonstrated multiple sites of gingival recession, including lingual-gingival recession (Figure 4 through Figure 6). Images from CBCT scanning revealed deficient facial and lingual alveolar bone (Figure 7) with a radiographic-supporting bone index (RSBI) of a class C risk factor.13 RSBI utilizes CBCT technology to evaluate both tooth and bone volume in the sagittal plane. Alveolar bone thickness at the facial and lingual alveolar crest are measured on the CBCT scan, in the sagittal plan. For example, a patient with RSBI class A presents with at least 1.5 mm of alveolar bone thickness, which will adequately support gingival soft tissue. A person with RSBI class B presents radiographically with <1.5 mm but >0.05 mm of bone volume, which may be a risk factor for future gingival recession, especially following orthodontic expansion of tooth arches. An individual with RSBI class C presents with <0.5 mm of alveolar bone, supporting gingival soft tissue, and indicates a high risk for gingival recession and subsequent orthodontic relapse.
The patient presented with the following signs and symptoms relative to DSDS: orthodontic tooth crowding, radiographic apical root resorption, gingival recession, orthodontic relapse, and bicuspid and third-molar tooth extractions.
Patient 2
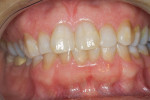
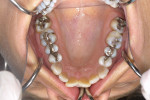
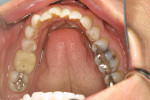
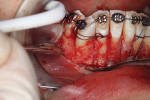
A 46-year-old healthy female was referred to the author’s office by her orthodontist. She requested PAOO43,44 treatment to expedite her proposed orthodontic re-treatment plan. Her orthodontist suspected the presence of facial bony dehiscence lesions associated with many of her teeth (Figure 9). She presented with significant post-orthodontic treatment relapse, malocclusion, and esthetic dissatisfaction (Figure 10 through Figure 12), as well as a dental history of comprehensive orthodontic treatment with the removal of her third molars (Figure 8) during her adolescence. At the time of periodontal augmentation surgery, both hard and soft tissues together with corticotomies, numerous bony dehiscences, and fenestrations were identified (Figure 13 through Figure 15). This suggested a discrepancy of inadequate alveolar bone width in the sagittal plane, relative to the same dimension of each associated tooth. The patient manifested the following signs or symptoms of DSDS: crowding in both arches, previous extraction of impacted teeth, post-orthodontic treatment relapse, and localized gingival recession.
Patient 3
A healthy 11-year-old female was referred to the author’s office for localized PAOO treatment. She presented with a severe anterior overbite, early gingival recession on mandibular canines, a thin phenotype, and prominent root surfaces visible through her alveolar mucosa (Figure 16). Radiographically, potentially impacted third-molar teeth were identified, with over-eruption of her mandibular anterior teeth (Figure 17).
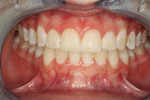
Two phases of treatment were proposed, including: (1) mandibular anterior frenectomy plus simultaneous connective tissue gingival onlay graft to minimize the risk for frenum regrowth (Figure 19); and (2) both hard- and soft-tissue gingival and alveolar bone augmentation, for her 6 mandibular anterior teeth. It was anticipated that the proposed treatment plan for her thin mandibular anterior alveolar bone, including alveolar bony dehiscences should minimize the risk for future gingival recession following orthodontic arch expansion.45 Literature also suggests greater post-orthodontic treatment stability will occur.45 At the time of surgical augmentation, significant alveolar bony dehiscence were noted (Figure 18). Figure 19 demonstrates the same patient 2 years following PAOO treatment. A robust mucogingival complex was noted, with ideal occlusion and orthodontic stability. Features of DSDS for this 11-year-old patient included tooth impactions, gingival recession, orthodontic tooth crowding and mandibular incisor over-eruption, and bony dehiscences.
Summary and Conclusion
DSDS is a new syndrome proposed for dentistry. The three representative, yet different, case reports presented demonstrate the discrepancies between alveolar bone volume and tooth volume, in three dimensions, resulting in an inability to effectively accommodate the patient’s teeth, as seen in three dimensions. In this author’s opinion and clinical experience, viable treatment for this syndrome includes both hard- and soft-tissue augmentation, prior to initiation of orthodontic treatment utilizing the principles of PAOO.43,44,46 Further research is indicated to support these concepts.
About the Author
Colin S. Richman, DMD, BDS, LDS, RCS, H. Dip Dent.
Assistant Clinical Professor
Department of Periodontics
Georgia Regents University
Augusta, Georgia
Private Practice
Periodontics
Roswell, Georgia
References
1. Mossey PA. The heritability of malocclusion: part 2. The influence of genetics in malocclusion. Br J Orthod. 1999;26(3):195-203.
2. Mossey PA. The heritability of malocclusion: part 1—genetics, principles and terminology. Br J Orthod. 1999;26(2):103-113.
3. von Cramon-Taubadel N. Global human mandibular variation reflects differences in agricultural and hunter-gatherer subsistence strategies. Proc Natl Acad Sci U S A. 2011;108(49):19546-19551.
4. Rose JC, Roblee RD. Origins of dental crowding and malocclusions: an anthropological perspective. Compend Contin Educ Dent. 2009;30(5):292-300.
5. Moss ML. The functional matrix hypothesis revisited. 4. The epigenetic antithesis and the resolving synthesis. Am J Orthod Dentofacial Orthop. 1997;112(4):410-417.
6. Moss ML. The functional matrix hypothesis revisited. 3. The genomic thesis. Am J Orthod Dentofacial Orthop. 1997;112(3):338-342.
7. Moss ML. The functional matrix hypothesis revisited. 2. The role of an osseous connected cellular network. Am J Orthod Dentofacial Orthop. 1997;112(2):221-226.
8. Moss ML. The functional matrix hypothesis revisited. 1. The role of mechanotransduction. Am J Orthod Dentofacial Orthop. 1997;112(1):8-11.
9. Moss ML, Rankow RM. The role of the functional matrix in mandibular growth. Angle Orthod. 1968;38(2):95-103.
10. Dempsey PJ, Townsend GC. Genetic and environmental contributions to variation in human tooth size. Heredity (Edinb). 2001;86(Pt 6):685-693.
11. Dempsey PJ, Townsend GC, Martin NG, Neale MC. Genetic covariance structure of incisor crown size in twins. J Dent Res. 1995;74(7):1389-1398.
12. Bolton WA. The clinical application of a tooth-size analysis. Am J Orthod. 1962;48(23):504-529.
13. Richman C. Is gingival recession a consequence of an orthodontic tooth size and/or tooth position discrepancy? A paradigm shift. Compend Contin Educ Dent. 2011;32(1):62-69.
14. Hirshfeld I. A study of skulls in the American Museum of Natural History in relation to periodontal disease. J Dent Res. 1923;5:241-265.
15. Corruciccini R. How Anthropology Informs the Orthodontic Diagnosis of Malocclusion’s Causes. Lewiston, NY: The Edwin Mellen Press;1999.
16. Lieberman DE. The Evolution of the Human Head. Boston, MA: Harvard University Press; 2011.
17. Hixon EH, Oldfather RE. Estimation of the sizes of unerupted cuspid and bicuspid teeth. Angle Orthod. 1958;28(24):236-240.
18. Howe R, McNamara JA, O’Connor, KA. An examination of dental crowding and its relationship to tooth size and arch dimension. Am J Orthod. 1983;83(26):363-373.
19. Hunter W. Application of analysis of crowding and spacing of the teeth. Dent Clin North Am. 1978;22(19):563-577.
20. Moyers RE. Handbook of Orthodontics. 3rd ed. Chicago, IL: Year Book Medical Publishers, Inc;1973.
21. Stanley RN, Kerber RE. A revision of the Hixon and Oldfather mixed-dentition prediction method. Am J Orthod. 1980;78(22):296-302.
22. Shigenobu N, Hisano, M, Shima, S, et al. Patterns of dental crowding in the lower arch and contributing factors. A statistical study. Angle Orthod. 2007;77(25):303-310.
23. Little RM. The irregularity index: a quantitative score of mandibular anterior alignment. Am J Orthod. 1975;68(5):554-563.
24. Proffit WR, Fields HW Jr, Moray LJ. Prevalence of malocclusion and orthodontic treatment needs in the United States: estimates from the NHANES III survey. Int J Adult Orthodon Orthognath Surg. 1998;13(2):97-106.
25. Renkema AM, Fudalej PS, Renkema AA, et al. Gingival labial recessions in orthodontically treated and untreated individuals: a case-control study. J Clin Periodontol. 2013;40(6):631-637.
26. Health, United States, 2004. Hyattsville, MD: National Center for Health Statistics; 2007.
27. Daprile G, Gatto MR, Checchi L. The evolution of buccal gingival recession in a student population: a 5-year follow-up. J Periodontol. 2007;78(P)(16):611-614.
28. Kassab MM, Cohen RE. The etiology and prevalence of gingival recession. J Am Dent Assoc. 2003;1342:220-225.
29. Rothe LE, Bollen AM, Little RM, et al. Trabecular and cortical bone as risk factors for orthodontic relapse. Am J Orthod Dentofacial Orthop. 2006;130(4):476-484.
30. Serino G, Wennstrom JL, Lindhe J, Eneroth L. The prevalence and distribution of gingival recession in subjects with a high standard of oral hygiene. J Clin Periodontol. 1994;21(1):57-63.
31. Vlaskalic V, Boyd RL, Baumrind S. Etiology and sequelae of root resorption. Semin Orthod. 1998;4(2):124-131.
32. Nardozzo TM. Third molar impactions: need for removal and care. Pa Dent J (Harrisb). 1966;33(8):303-306 passim.
33. Riedel RA. A post-retention assessment of relapse, recidivism, adjustment, change and stability. In: Moorrees, CFA, van der Linden, FPGM, eds. Orthodontics: Evaluation and Future. Proceedings of the International Conference on the Occasion of the 25th Anniversary of the Orthodontic Department of the University of Nymegen, The Netherlands. 1988;22-24(34):281-306.
34. Kuijpers-Jagtman AM, Al Yami EA, van ‘t Hof MA. Long-term stability of orthodontic treatment. Ned Tijdschr Tandheelkd. 2000;107(4):178-181.
35. Lakhani MJ, Kadri W, Mehdi H, et al. Anterior arch crowding—a possible predictor for mandibular third molar impaction. J Ayub Med Coll Abbottabad. 2011;23(1):63-65.
36. Little RM, Riedel RA, Artun J. An evaluation of changes in mandibular anterior alignment from 10 to 20 years postretention. Am J Orthod Dentofacial Orthop. 1988;93(5):423-428.
37. Katranji A, Misch, K, Wang, HL. Cortical bone thickness in dentate and edentulous human cadavers. J Periodontol. 2007;78(5):874-878.
38. Larato D. Alveolar plate fenestrations and dehiscences of the human skull. Oral Surg Oral Med Oral Pathol. 1970;29(32):816.
39. Araujo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
40. Araujo MG, Sukekava F, Wennstrom JL, Lindhe J. Ridge alterations following implant placement in fresh extraction sockets: an experimental study in the dog. J Clin Periodontol. 2005;32(6):645-652.
41. Mandelaris GA, Vence BS, Rosenfeld AL, Forbes DP. A classification system for crestal and radicular dentoalveolar bone phenotypes. Int J Periodontics Restorative Dent. 2013;33(3):289-296.
42. Elliott J, Bowers, GM. Alveolar dehiscence and fenestrations. Periodontics. 1963;1(31):245.
43. Wilcko WM, Wilcko MT, Bouquot JE, et al. Rapid orthodontics with alveolar reshaping: two case reports of decrowding. Int. J Periodontics Restorative Dent. 2001;21(33):9-19.
44. Wilcko MT, Wilcko WM, Pulver JJ, et al. Accelerated osteogenic orthodontics technique: a 1-stage surgically facilitated rapid orthodontic technique with alveolar augmentation. J Oral Maxillofac Surg. 2009;67(10):2149-2159.
45. Makki L, Ferguson DJ, Wilcko MT, et al. Mandibular irregularity index stability following alveolar corticotomy and grafting: a 10-year preliminary study. Angle Orthod. 2015;85(5):743-749.
46. Wilcko WM, Wilcko T, Bouquot JE, Ferguson DJ. Rapid orthodontics with alveolar reshaping: two case reports of decrowding. Int J Periodontics Restorative Dent. 2001;21(1):9-19.