Intraoral Repair of a Glass-Ceramic Restoration With Nanohybrid Ormocers: A Case Report
Juergen Manhart, Prof. Dr. med. dent.; and Iris Frasheri, DDS
Abstract:
The interest in repairing failed restorations has been growing; such a procedure may prolong the life span of defective direct and indirect dental restorations. The intraoral repair using resin-based composite materials has major benefits, as it preserves the main body of the restoration, avoids unnecessary removal of sound tooth structure, is an easy and fast treatment, avoids additional appointments, and is inexpensive. This case report demonstrates the use of nanohybrid Ormocer®, or organically modified ceramic, materials.
The use of all-ceramic dental restorations has become more popular during the last 30 years for several reasons, such as their favorable optical properties, excellent esthetics, wear resistance, color stability, chemical durability, biocompatibility, and strengthening of the remaining tooth structure when the restorations are adhesively bonded.1,2 This trend has been fueled, in large part, by the increasing number of patients requesting esthetic restorations and metal-free alternatives to traditional prosthodontics.
Dental ceramics can be classified as predominantly glassy (feldspathic glass, eg, Vitablocs Mark II, Vita, vita-zahnfabrik.com); particle-filled glassy (eg, lithium disilicate-reinforced, e.max® Press, Ivoclar Vivadent, ivoclarvivadent.us); and polycrystalline (alumina, eg, Procera®, Nobel Biocare, nobelbiocare.com; zirconia, eg, Lava™, 3M Oral Care, 3m.com).3-6
The most common clinical indications for all-ceramic restorations consist of inlays, onlays, partial crowns, full crowns, bridges, veneers, and posterior occlusal veneers (table tops).7-9 These restorations present a scientifically proven, high-quality permanent treatment option for the esthetically challenging anterior and load-bearing posterior regions when the indications and limitations of the respective ceramic systems are respected and an appropriate luting procedure is employed; their reliability has been documented in the literature.8,10,11
However, every ceramic restoration is vulnerable to failure during clinical service due to biologic (eg, secondary caries, pulp status, periodontal disease) and mechanical/technical (eg, chipping of veneer ceramic, ceramic bulk fracture, cusp fracture of retaining tooth, loss of retention) reasons.12 Brittleness, crack initiation and crack propagation, and low tensile strength are the weaknesses of ceramics8,13 and jeopardize the materials’ integrity during clinical service when errors in restoration fabrication or inappropriate use of the material occurs.14 Flaws in the microstructure of the ceramic or related to fabrication have been identified as causes for catastrophic failure of ceramic restorations.15,16 Also, the cusps of the restored teeth are vulnerable.15,17 In recent years, fracture-related failures have been observed to be an increasing problem as a result of the fast-growing popularity of ceramic restorations.18
Intraoral repair of failed ceramic restorations offers both the dentist and patient a time-saving and cost-effective alternative to replacement1,19,20 and contributes to the preservation of tooth structure, as every complete removal and replacement of a defective restoration leads to an extension of the original preparation.21 The repair involves the bonding of resin-based products to the remaining ceramic and, if applicable, other adjacent structures such as dentin, enamel, and metal. Each involved structure requires a specific pretreatment-protocol sequence to prepare these different substrates for the application of the repairing composite.22
The intraoral pretreatment of defective glass-ceramic restorations prior to the application of the repairing composite can be done sufficiently using hydrofluoric acid etching or tribochemical silica coating (CoJet™, 3M Oral Care).23-25 Etching with hydrofluoric-acid gel followed by silanization is a well-established conditioning method to promote adhesion of resin-based materials to feldspathic, leucite, and lithium-disilicate glass-ceramic restorations.26-29 However, the hazardous effects of hydrofluoric acid in vivo, which include the irritation of soft tissues, are well recognized.28,30,31 Hydrofluoric acid is one of the most corrosive inorganic acids, and its use can cause a chemical burn with progressive and serious tissue necrosis accompanied by severe pain.31,32 Due to this serious potential hazard, intraoral application of hydrofluoric acid seems to be too dangerous and should be avoided, especially as an effective and nonhazardous alternative exists in tribochemically assisted bonding (intraoral silicatization and subsequent silanization) of repair composite to the failed ceramic structure.20,23,25,29 The use of acidulated phosphate fluoride gel has been proposed as a safe intraoral alternative to the employment of hydrofluoric acid for ceramic surface etching with little risk for injuring the soft tissues; however, this pretreatment has never became a clinical standard probably due to its deficient efficiency.20,30,33 Airborne-particle abrasion with aluminum oxide (Al2O3) is used for preconditioning ceramic surfaces prior to a resin application.20,33,34 Alternatively, micromechanical roughening of the glass-ceramic surface prior to the application of the repair components using diamond burs or rotary carborundum stones is employed.20,33,35 However, completely and reliably pretreating all respective surfaces, especially difficult-to-reach areas, with these rotary instruments without endangering the integrity of neighboring teeth is difficult. Sandblasting or air-abrasion pretreatments provide, along with enhanced effectiveness, access to difficult-to-reach repair sites.25
After physical or etching pretreatment of the glass-ceramic surface, the subsequent application of a silane is critical for promoting covalent chemical bonding of composite to ceramic with sufficient bond strengths.33-36 The ceramic-resin bond is formed by both etching the surface to create micromechanical attachment features and by the application of silane coupling agents to provide chemical interaction between the silicon-based ceramic and carbon-based composite resins.1 The fracture resistance of the ceramic-composite adhesion zone is controlled primarily by the ceramic microstructure and ceramic surface treatment.37
Recently, multimode universal adhesives were introduced that enable clinicians to use self-etch bonding procedures to dental substrates as well as etch-and-rinse regimens (selective enamel etch or total etch of enamel and dentin) with prior application of phosphoric acid.38,39 Some of these universal adhesives also incorporate a silane primer for chemical bonding to silica-based ceramics or methacryloyloxydecyl dihydrogen phosphate for chemical bonding to zirconia-based ceramics without the need for an extra primer step.22,38,40 This is advantageous for the intraoral repair of defective ceramic restorations, especially in complex clinical scenarios, when different dental tissues (enamel, dentin) and prosthetic substrates (glass-ceramic, veneering porcelain, zirconia coping, metal framework) both need adhesive pretreatment.
Most composite materials contain organic monomer matrices based on conventional methacrylate chemistry (eg, bisphenol A-glycidyl methacrylate [bisGMA], urethane dimethacrylate [UDMA], triethyleneglycol dimethacrylate [TEGDMA]). Alternative chemical formulations use silorane resins41 and Ormocers® (The Fraunhofer Insitute for Silicate Research ISC, www.isc.fraunhofer.de),42 which is the abbreviation for organically modified ceramics. Patented technology, these nonmetallic inorganic compound materials are composed of an inorganic Si-O-Si-glass network (backbone molecule) and an organic polymer phase. Ormocer technology was developed by the Fraunhofer Institute for Silicate Research ISC in Würzburg, Germany and introduced in 1998 as a dental restorative material.42 Since then, further development of Ormocer-based composites for this range of applications has been significant. In dental Ormocer products of the past, conventional methacrylates were supplemented in the pure Ormocer chemistry in the monomer matrix (in addition to initiators, stabilizers, pigments, and inorganic filler particles) to improve workability. Newer products feature a pure Ormocer matrix chemistry without conventional dimethacrylates and nanohybrid inorganic filler particles. A low polymerization shrinkage and a low shrinkage stress have been measured for these materials.
This clinical report illustrates the intraoral repair of a defective ceramic inlay using air-abrasive tribochemical pretreatment and subsequent application of a multimode adhesive and an Ormocer restorative combination.
Case Presentation
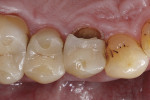
A 56-year-old male presented to the dental office with a fractured palatal cusp adjacent to a glass-ceramic inlay in a first maxillary bicuspid (Figure 1). The main reasons for failure of ceramic inlays, onlays and partial crowns are bulk fracture of the restoration, tooth fracture, postoperative symptoms, and secondary caries.43,44 In vivo, tooth fractures are usually the result of the accumulation of repeated stress during oral function, producing cracks, which extend from internal preparation line angles at the floor of a restoration cervically toward the cementoenamel junction.15,45 Although fractures can occur in healthy teeth, they are more common in decayed or restored teeth.46 Fractured cusps usually result from insufficient cusp support when an intracoronal restoration weakens the marginal ridge.45 The crack often extends in mesiodistal and buccolingual directions, commonly involving one or both marginal ridges and a buccal or lingual groove. The crack terminates in the cervical region either parallel to the gingival margin or slightly subgingival.45
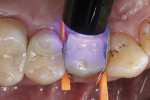
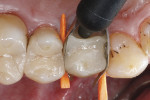
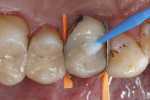
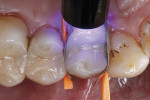
A flowable nanohybrid Ormocer was applied to the internal line and point angles of the fracture site (Figure 11). Due to its excellent wetting properties, the low-viscosity Ormocer ensures a good and void-free adaptation of the first repair increment to the surrounding tissues and materials. Positive effects of low-viscosity materials on internal adaptation and interfacial integrity have been shown in literature.51 The flowable restorative was polymerized with a high-performance curing light for 20 seconds (light intensity > 800 mW/cm²) (Figure 12). Subsequently, the remaining volume of the defect was filled using a regular-viscosity nanohybrid Ormocer to rebuild the lost palatal cusp of the premolar (Figure 13). A clean new microbrush was engaged as a perfect modeling instrument to carefully adapt the restorative material to the edge of the ceramic inlay (Figure 14). The Ormocer was again light cured for 20 seconds (light intensity > 800 mW/cm²) (Figure 15). After removal of the metal matrix bands, additional 10-second curing cycles from mesial-palatal and distal-palatal directions were executed to guarantee sufficient material polymerization in these areas, thus ensuring excellent physicomechanical properties.
In this situation, the adhesive bond between the ceramic inlay and palatal cusp was no longer strong enough after many years of clinical service (bond degradation) to withstand intraoral stresses and occlusal/masticatory forces (Figure 2). Opposing tooth contacts in dynamic occlusion generating high loads on the oral cusp also may have contributed to the fracture. The complete palatal cusp separated from the ceramic inlay, because of harmful stress distribution resulting in crack formation and propagation along the adhesive interface, and disintegrated parallel to the gingival margin due to peak stresses exceeding its remaining load-bearing capacity. A mixture of fatigue effects as a result of normal function (subcritical stresses) for many years, detrimental excessive loads during mastication or unphysiologic tooth contacts (bruxism, clenching), and hydrolytic degradation of the adhesive and luting resin were probably responsible for that catastrophic event. In most clinical cases of teeth showing fracture-related failures, the weakest bond in restorative technique is the interface between the restorative material and tooth structures.47 The steep cusp-fossa relation of maxillary premolars is a predisposing factor for crack initiation, especially in teeth with cavity preparations.48 However, the treatment of a fractured cusp has a good prognosis, especially when the crack does not extend below the gingival attachment.49 But clinicians face challenges when a missing functional cusp of a maxillary premolar has to be replaced.50
During the clinical inspection, the damaged tooth demonstrated sensitivity in the cold test and showed no negative reaction to the percussion test. After discussing with the patient the possible restorative alternatives and treatment fees, the patient decided on a tooth structure-saving direct repair restoration using nanohybrid Ormocer materials.
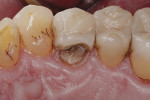
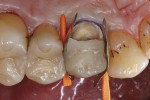
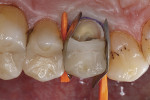
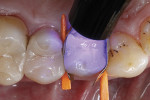
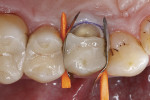
Treatment started with thoroughly cleaning the affected tooth of external deposits using a fluoride-free prophylaxis paste and a rubber cup. Subsequently, the fracture site was cleaned, loose fragments were removed, and the fracture margins were smoothened (Figure 3). A retraction cord was placed in the gingival sulcus besides the fractured cusp to control the soft tissues (Figure 4). After placing metal matrix bands in the mesial and distal proximal areas to protect the neighboring teeth from subsequent pretreatment steps of the repair procedure (Figure 5), the intaglio surface of the ceramic inlay at the fracture site was roughened and tribochemically silica coated (CoJet) using an intraoral air-abrasion device (Figure 6). Subsequently, the operation site was cleaned, using oil-free compressed air.
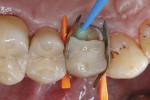
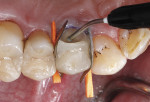
A multimode adhesive was selected for bonding. These modern one-bottle adhesives are compatible with all conditioning techniques of hard dental tissues, the self-etch technique, and the phosphoric acid-based conditioning techniques (selective enamel etching or complete etch-and-rinse pretreatment of enamel and dentin). The selected adhesive was also qualified for the repair of all-ceramic restorations without the need to use an additional ceramic primer. In this repair case, the adhesive was used, employing the self-etch technique. Ample amounts of the multimode adhesive were applied and distributed generously using a microbrush in the area of the fracture site on the respective dentin, enamel, and glass-ceramic surfaces (Figure 7). All bonding areas were sufficiently covered by the adhesive.
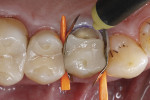
After at least 20 seconds of carefully scrubbing the adhesive into the hard dental tissues and on the intaglio ceramic surface, the solvent was carefully evaporated from the bonding agent with oil-free compressed air (Figure 8). The adhesive was subsequently light cured for 10 seconds (Figure 9). The result was a shiny surface, evenly covered with adhesive (Figure 10). This should be carefully checked, as any bonding areas that appear dull are an indication that insufficient amount of adhesive has been applied to those sites. In the worst case, this could result in reduced bonding of the subsequent restoration in these areas and, at the same time, in reduced dentin sealing, which could lead to postoperative sensitivity. If such areas are found in the visual inspection, an additional bonding agent is selectively applied to them.
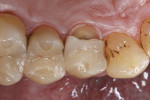
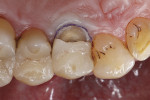
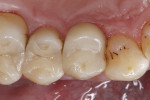
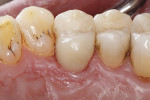
The repair restoration was finished first with fine-grit diamond-bur instrumentation and then with abrasive disks; occlusal interferences in static and dynamic occlusion were eliminated. The use of diamond-impregnated composite polishers achieved a high-gloss finish on the restoration’s surface. Figure 16 and Figure 17 show the completed direct Ormocer repair restoration with cusp replacement, reconstructing the original tooth shape.
Discussion
A laboratory investigation showed preparing maxillary premolars with large and deep mesial-occlusal-distal (MOD) preparations can severely weaken teeth by approximately 59% of their original fracture strength, and composite and ceramic inlays both were not able to completely restore the original load capacity of the teeth.15 However, other reports in the literature demonstrate that direct and indirect adhesively bonded restorations can partly restore the fracture resistance of teeth weakened by cavity preparations up to values not significantly different to unprepared healthy teeth.52-54
The palatal cusps of maxillary premolars present a special fracture risk.15,17,53 Maxillary bicuspids have an unfavorable anatomic shape, crown volume, and crown/root proportion, making them more susceptible to cusp fractures compared with other posterior teeth when submitted to occlusal load application.55,56 Each preparation weakens the tooth, and once the continuity of the enamel is broken, the tooth is at higher risk for fracture.15,47,57,58 The depth of the cavity preparation seems to be the most critical factor with regard to fracture risk for the prepared tooth.59,60 The fractured cusp usually results from a lack of cusp support due to a weakened marginal ridge because of Class II restorations or extensive caries.47,49,56,61 Optimal restorations for teeth with extensive Class II MOD preparations are onlays that include cuspal coverage to reduce cuspal flexion under masticatory load, but it is debatable at which degree of tooth destruction or cavity size onlays should be preferred instead of bonded inlays.15,54,62,63
Findings from an in vitro investigation showed ceramic coverage of compromised cusps did not demonstrate an increased risk for fracture resistance after fatigue loading when compared with less invasive partial-coverage restorations.54 However, the same study reported enhanced exposure of restoration margins to occlusal wear could result in more extensive marginal discrepancies. Occlusal contact points of antagonist teeth sliding over restoration margins of intracoronal ceramic inlay restorations could compromise the integrity of the marginal seal under clinical conditions.54 Marginal fit decreases faster when ceramic margins are directly loaded and marginal chipping is able to occur.64 After adhesive cementation, the quality of marginal adaptation of ceramic restorations is likely to deteriorate because of hydrolytic and mechanical stresses.44,65 When marginal quality decreases, the risk for partial debonding, gap formation, and secondary-caries formation increases. This can promote crack initiation and propagation in the layer of the adhesive cement, and, in a worst-case scenario, eventually result in cuspal fracture adjacent to ceramic inlays. This is because the mechanisms (adhesive bond) that transfer stresses during occlusal loading from the ceramic restoration to the tooth structure and vice versa no longer function reliably.66
Meanwhile, many dental schools in Europe and North America have begun to include teaching repair of restorations in their academic curricula.67,68 The intraoral repair of defective ceramic restorations using resin composite compared with complete restoration replacement has major benefits, as it preserves the main body of the restoration, avoids unnecessary removal of sound tooth structure, is an easy and fast treatment procedure, avoids additional appointments, and is an inexpensive solution for such problems.19,21 The main disadvantage of repair restorations is the anticipation of the long-term survival of this kind of dental restorations.30 However, when weighing the potential benefits against the risk for early failure in suitable and carefully selected clinical cases, this treatment alternative deserves at least one attempt. The results of a prospective cohort study showed 1 year after repair of defective restorations a low failure rate.69 A practice-based study reported a better prognosis for repairs on restorations that failed due to caries compared with fracture-related failures.70
Nonetheless, a high-quality direct resin-based repair restoration continues to be dependent on several prerequisites, such as protection of patients’ soft tissues, careful and adequate pretreatment of the different involved substrates, effective and correct application of the adhesive system, appropriate handling of the restorative material, and sufficient curing of the composite.22 The repair restorations are typically performed using direct resin-based materials. Supplementary to composites based on traditional methacrylate chemistry, the materials options in the sector of light-activated direct-placement restoratives were expanded by a pure Ormocer version that does not contain added conventional dimethacrylates in its chemical formulation.
Conclusion
Interest in the repair of insufficient restorations has garnered increasing popularity and is a valuable method for considerably prolonging the clinical life span of defective direct and indirect dental restorations.14,70 It is reported that porcelain repair systems form durable bonds to fractured porcelain and exposed metal surfaces.1
Disclosure
Dr. Manhart has received materials support from VOCO America.
About the Authors
Juergen Manhart, Prof. Dr. med. dent.
Department of Restorative Dentistry
Dental School of the Ludwig-Maximilians-University
Munich, Germany
Iris Frasheri, DDS
Assistant Professor
Department of Restorative Dentistry
Dental School of the Ludwig-Maximilians-University
Munich, Germany
References
1. Kelly JR, Benetti P. Ceramic materials in dentistry: historical evolution and current practice. Aust Dent J. 2011;56 Suppl 1:84-96.
2. Li RW, Chow TW, Matinlinna JP. Ceramic dental biomaterials and CAD/CAM technology: state of the art. J Prosthodont Res. 2014;58(4):208-216.
3. Kelly JR. Dental ceramics: what is this stuff anyway? J Am Dent Assoc. 2008;139 Suppl:4S-7S.
4. Kelly JR. Dental ceramics: current thinking and trends. Dent Clin North Am. 2004;48(2):513-530.
5. Denry IL. Recent advances in ceramics for dentistry. Crit Rev Oral Biol Med. 1996;7(2):134-143.
6. Miyazaki T, Nakamura T, Matsumura H, Ban S, Kobayashi T. Current status of zirconia restoration. J Prosthodont Res. 2013;57(4):236-261.
7. Magne P, Schlichting LH, Maia HP, Baratieri LN. In vitro fatigue resistance of CAD/CAM composite resin and ceramic posterior occlusal veneers. J Prosthet Dent. 2010;104(3):149-157.
8. Beier US, Dumfahrt H. Longevity of silicate ceramic restorations. Quintessence Int. 2014;45(8):637-644.
9. Kern M, Beuer F, Frankenberger R, et al. All-Ceramics at a glance. Guidelines for indications, material selection, preparation and inclusion of all-ceramic restorations [In German]. 6th ed. Ettlingen. 2015.
10. Layton DM, Clarke M, Walton TR. A systematic review and meta-analysis of the survival of feldspathic porcelain veneers over 5 and 10 years. Int J Prosthodont. 2012;25(6):590-603.
11. Kramer N, Frankenberger R. Clinical performance of bonded leucite-reinforced glass ceramic inlays and onlays after eight years. Dent Mater. 2005;21(3):262-271.
12. Banerjee A, Watson TF. Pickard’s Guide to Minimally Invasive Operative Dentistry. 10th ed. Oxford, GB: Oxford University Press; 2015.
13. Conrad HJ, Seong WJ, Pesun GJ. Current ceramic materials and systems with clinical recommendations: A systematic review. J Prosthet Dent. 2007;98(5):389-404.
14. Blum IR, Jagger DC, Wilson NH. Defective dental restorations: to repair or not to repair? Part 2: All-ceramics and porcelain fused to metal systems. Dent Update. 2011;38(3):150-158.
15. St-Georges AJ, Sturdevant JR, Swift EJ Jr, Thompson JY. Fracture resistance of prepared teeth restored with bonded inlay restorations. J Prosthet Dent. 2003;89(6):551-557.
16. Kelly JR, Giordano R, Pober R, Cima MJ. Fracture surface analysis of dental ceramics: clinically failed restorations. Int J Prosthodont. 1990;3(5):430-440.
17. Eakle WS, Maxwell EH, Braly BV. Fractures of posterior teeth in adults. J Am Dent Assoc. 1986;112(2):215-218.
18. Kimmich M, Stappert CF. Intraoral treatment of veneering porcelain chipping of fixed dental restorations: a review and clinical application. J Am Dent Assoc. 2013;144(1):31-44.
19. Reston EG, Filho SC, Arossi G, Cogo RB, Rocha Cdos S, Closs LQ. Repairing ceramic restorations: final solution or alternative procedure? Oper Dent. 2008;33(4):461-466.
20. Ozcan M. Evaluation of alternative intra-oral repair techniques for fractured ceramic-fused-to-metal restorations. J Oral Rehabil. 2003;30(2): 194-203.
21. Frankenberger R. Korrektur zahnärztlicher Restaurationen. Zahnärztliche Mitteilungen. 2012;102(22):32-41.
22. Ozcan M. Surface conditioning protocol for multiple substrates in repair of cervical recessions adjacent to ceramic. J Adhes Dent. 2014;16(4):394.
23. Frankenberger R, Kramer N, Sindel J. Repair strength of etched vs silica-coated metal-ceramic and all-ceramic restorations. Oper Dent.2000;25(3):209-215.
24. Guggenberger R. Rocatec system--adhesion by tribochemical coating. Dtsch Zahnärztl Z. 1989;44(11):874-876.
25. Frankenberger R, Heidemann D, Staehle HJ, Hellwig E, Blunck U, Hickel R. Neue Begriffe in der restaurativen Zahnerhaltung. Dtsch Zahnärztl Zeitschr. 2014;69(12):722-734.
26. Ozcan M, Valandro LF, Pereira SM, Amaral R, Bottino MA, Pekkan G. Effect of surface conditioning modalities on the repair bond strength of resin composite to the zirconia core / veneering ceramic complex. J Adhes Dent. 2013;15(3):207-210.
27. Brentel AS, Ozcan M, Valandro LF, Alarca LG, Amaral R, Bottino MA. Microtensile bond strength of a resin cement to feldpathic ceramic after different etching and silanization regimens in dry and aged conditions. Dent Mater. 2007;23(11):1323-1331.
28. Ozcan M, Allahbeickaraghi A, Dundar M. Possible hazardous effects of hydrofluoric acid and recommendations for treatment approach: a review. Clin Oral Investig. 2012;16(1):15-23.
29. Matinlinna JP. Processing and bonding of dental ceramics. In: Vallittu P, ed. Non-Metallic Biomaterials for Tooth Repair and Replacement. Oxford, GB: Woodhead Publishing Ltd.; 2013:129-160.
30. Tylka DF, Stewart GP. Comparison of acidulated phosphate fluoride gel and hydrofluoric acid etchants for porcelain-composite repair. J Prosthet Dent. 1994;72(2):121-127.
31. Moore PA, Manor RC. Hydrofluoric acid burns. J Prosthet Dent. 1982; 47(3):338-339.
32. Ohata U, Hara H, Suzuki H. 7 cases of hydrofluoric acid burn in which calcium gluconate was effective for relief of severe pain. Contact Dermatitis. 2005;52(3):133-137.
33. Lacy AM, LaLuz J, Watanabe LG, Dellinges M. Effect of porcelain surface treatment on the bond to composite. J Prosthet Dent.1988;60(3):288-291.
34. Blatz MB, Sadan A, Kern M. Resin-ceramic bonding: a review of the literature. J Prosthet Dent. 2003;89(3):268-274.
35. Kumbuloglu O, User A, Toksavul S, Vallittu PK. Intra-oral adhesive systems for ceramic repairs: a comparison. Acta Odontol Scand. 2003;61(5):268-272.
36. Kupiec KA, Wuertz KM, Barkmeier WW, Wilwerding TM. Evaluation of porcelain surface treatments and agents for composite-to-porcelain repair. J Prosthet Dent. 1996;76(2):119-124.
37. Della Bona A, Anusavice KJ, Shen C. Microtensile strength of composite bonded to hot-pressed ceramics. J Adhes Dent. 2000;2(4):305-313.
38. Tay F. An update on adhesive dentistry. Compend Contin Educ Dent. 2014;35(4):286.
39. Perdigao J, Loguercio AD. Universal or Multi-mode Adhesives: Why and How? J Adhes Dent. 2014;16(2):193-194.
40. Giannini M, Makishi P, Ayres AP, et al. Self-etch adhesive systems: a literature review. Braz Dent J. 2015;26(1):3-10.
41. Guggenberger R, Weinmann W. Exploring beyond methacrylates. Am J Dent. 2000;13(Special Issue):82-84.
42. Wolter H, Storch W, Ott H. New inorganic/organic copolymers (ORMOCERs) for dental applications. MRS Proceedings. 1994;346(Mat. Res. Soc. Symp. Proc.):143-149.
43. van Dijken JWV, Hasselrot L, Ormin A, Olofsson AL. Restorations with extensive dentin/enamel-bonded ceramic coverage: A 5-year follow-up. Eur J Oral Sci. 2001;109:222-229.
44. Felden A, Schmalz G, Federlin M, Hiller KA. Retrospective clinical investigation and survival analysis on ceramic inlays and partial ceramic crowns: results up to 7 years. Clin Oral Investig. 1998;2(4):161-167.
45. Kahler W. The cracked tooth conundrum: terminology, classification, diagnosis, and management. Am J Dent. 2008;21(5):275-282.
46. Schuurs A. Pathology of the Hard Dental Tissues. West Sussex, UK: Wiley-Blackwell; 2012.
47. Reeh ES, Douglas WH, Messer HH. Stiffness of endodontically-treated teeth related to restoration technique. J Dent Res. 1989;68(11):1540-1544.
48. Lubisich EB, Hilton TJ, Ferracane J, Northwest P. Cracked teeth: a review of the literature. J Esthet Restor Dent. 2010;22(3):158-167.
49. Rivera E, Walton RE. Cracking the Cracked Tooth Code: Detection and Treatment of Various Longitudinal Tooth Fractures. Chicago, IL: American Association of Endodontists; 2008.
50. Kuo WC, Chang YH. Tooth-colored onlay restorations of cusp-fractured maxillary premolars - case report. J Dent Sci. 2008;3(1):49-56.
51. Frankenberger R, Krämer N, Pelka M, Petschelt A. Internal adaptation and overhang formation of direct class II resin composite restorations. Clin Oral Investig. 1999;3(4):208-215.
52. Cotert HS, Sen BH, Balkan M. In vitro comparison of cuspal fracture resistances of posterior teeth restored with various adhesive restorations. Int J Prosthodont. 2001;14(4):374-378.
53. Ragauska A, Apse P, Kasjanovs V, Berzina-Cimdina L. Influence of ceramic inlays and composite fillings on fracture resistance of premolars in vitro. Stomatologija. 2008;10(4):121-126.
54. Stappert CF, Abe P, Kurths V, Gerds T, Strub JR. Masticatory fatigue, fracture resistance, and marginal discrepancy of ceramic partial crowns with and without coverage of compromised cusps. J Adhes Dent. 2008;10(1):41-48.
55. Soares PV, Santos-Filho PC, Martins LR, Soares CJ. Influence of restorative technique on the biomechanical behavior of endodontically treated maxillary premolars. Part I: fracture resistance and fracture mode. J Prosthet Dent. 2008;99(1):30-37.
56. Burke FJ. Tooth fracture in vivo and in vitro. J Dent. 1992;20(3):131-139.
57. Mondelli J, Steagall L, Ishikiriama A, de-Lima-Navarro MF, Soares FB. Fracture strength of human teeth with cavity preparations. J Prosthet Dent. 1980;43(4):419-422.
58. Reeh ES, Messer HH, Douglas WH. Reduction in tooth stiffness as a result of endodontic and restorative procedures. J Endod. 1989;15(11):512-516.
59. Khera SC, Goel VK, Chen RC, Gurusami SA. Parameters of MOD cavity preparations: a 3-D FEM study, Part II. Oper Dent. 1991;16(2):42-54.
60. Lin CL, Chang CH, Wang CH, Ko CC, Lee HE. Numerical investigation of the factors affecting interfacial stresses in an MOD restored tooth by auto-meshed finite element method. J Oral Rehabil. 2001;28(6):517-525.
61. Larson TD, Douglas WH, Geistfeld RE. Effect of prepared cavities on the strength of teeth. Oper Dent. 1981;6(1):2-5.
62. Bitter K, Meyer-Lueckel H, Fotiadis N, et al. Influence of endodontic treatment, post insertion, and ceramic restoration on the fracture resistance of maxillary premolars. Int Endod J. 2010;43(6):469-477.
63. Frankenberger R, Zeilinger I, Krech M, et al. Stability of endodontically treated teeth with differently invasive restorations: Adhesive vs. non-adhesive cusp stabilization. Dent Mater. 2015;31(11):1312-1320.
64. Stappert CF, Chitmongkolsuk S, Silva NR, Att W, Strub JR. Effect of mouth-motion fatigue and thermal cycling on the marginal accuracy of partial coverage restorations made of various dental materials. Dent Mater. 2008;24(9):1248-1257.
65. Kramer N, Reinelt C, Richter G, Frankenberger R. Four-year clinical performance and marginal analysis of pressed glass ceramic inlays luted with ormocer restorative vs. conventional luting composite. J Dent. 2009;37(11):813-819.
66. Ona M, Watanabe C, Igarashi Y, Wakabayashi N. Influence of preparation design on failure risks of ceramic inlays: a finite element analysis. J Adhes Dent. 2011;13(4):367-373.
67. Lynch CD, Blum IR, Frazier KB, Haisch LD, Wilson NH. Repair or replacement of defective direct resin-based composite restorations: contemporary teaching in U.S. and Canadian dental schools. J Am Dent Assoc. 2012;143(2):157-163.
68. Blum IR, Lynch CD, Wilson NH. Teaching of the repair of defective composite restorations in Scandinavian dental schools. J Oral Rehabil. 2012;39(3):210-216.
69. Gordan VV, Riley JL, 3rd, Rindal DB, et al. Repair or replacement of restorations: a prospective cohort study by dentists in The National Dental Practice-Based Research Network. J Am Dent Assoc. 2015;146(12):895-903.
70. Opdam NJ, Bronkhorst EM, Loomans BA, Huysmans MC. Longevity of repaired restorations: a practice based study. J Dent. 2012;40(10):829-835.