What’s Hindering Dentistry From the Widespread Adoption of CT-Guided Surgery?
Gary Orentlicher, DMD; Andrew Horowitz, DMD, MD; and Marcus Abboud, DMD
Abstract
Although computed tomography (CT)-guided technology has been shown to increase implant placement accuracy, decrease surgical complications, and improve the predictability of implant case outcome, many in the dental implant community continue to resist using it for three main reasons: time, money, and fear. While it is true that there are additional preparatory steps necessary in patient case planning and the practitioner must invest in unfamiliar technologies and overcome a learning curve, increased efficiency, improved restorative outcomes, and clear benefits to both patients and practitioners make a strong case for adopting this approach.
Treatment with dental implants has become commonplace in dentistry today. The goals of treatment for doctors and their patients can at times be different. Doctors want predictable implant placement while considering a patient’s medical history, bone biology, anatomy, and the planned dental restoration position. They want to optimize osseointegration and case success by minimizing tissue trauma and involvement of surrounding anatomy and maximizing bone and soft-tissue support, with the planned restoration in mind. Patients want teeth that look and function like natural teeth that, ideally, will last many years. They desire quick, predictable, successful treatment with minimal pain, swelling, stress, complications, and interruption in their lives.
CT-Guided Workflow and Surgery Overview
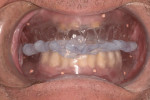
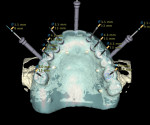
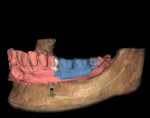
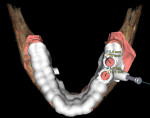
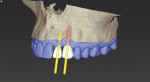
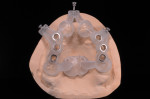
In the past decade, many major implant manufacturers have introduced techniques and instrumentation for computed tomography (CT)-guided surgery planning and placement. Workflows for treating both fully and partially edentulous patients have been developed. Some workflows require scans while patients are wearing scanning appliances that reproduce the planned implant restoration (Figure 1 and Figure 2), others incorporate optical intraoral patient scans and/or optical scans of models and wax-ups (Figure 3 and Figure 4), and some allow planning using “virtual teeth” (Figure 5). All workflows require treatment planning with software that allows for the review of the patient’s 3-dimensional (3-D) bony architecture and associated anatomy and their relationship to the planned prosthesis. Implants and abutments can then be “virtually” planned, driven by knowledge of the position of the planned restoration. Surgical drilling guides are then fabricated using 3-D printing technologies (eg, stereolithography, fused deposition molding, and laser sintering) (Figure 6 through Figure 8).1
CT-guided surgery can facilitate flapless implant placement.2-4 Accurate one-to-one measurements of the width and height of bone in planned implant sites, as well as distances and angulations both between implants and from one side of an arch to another, can be predetermined. It also allows predetermination of prosthesis path of insertion, placement of screw chambers, componentry space, and presurgical abutment choices. CT-guided technology makes it possible to foresee soft-tissue thicknesses, possible bony fenestrations, and the volume of bone graft material necessary, as well as to evaluate for sinus lift bone grafting, ridge splitting, or ridge augmentation. Additionally, it enables preoperative knowledge of ideal implant lengths and widths and the proximity of vital anatomic structures.5-8 As a result, there are minimal intraoperative surprises.
Once the doctor is comfortable and familiar with the drilling instrumentation, placing implants through a surgical guide reduces chairside surgical time. Moreover, because implant positioning is predetermined by the “virtual” treatment plan and incorporated into the surgical guide, implants can be placed with minimal stress to the clinician.
No dental implant placement technique or technology is free of error, and CT-guided surgery is no exception. Potential error, which is additive and cumulative, is found in each step of the workflow. Minimization of error requires the dental team to pay close attention to detail throughout the workflow process.
Most importantly, this technology allows the clinician to plan and place implants according to a prosthetically driven treatment plan. That is, the positions of the planned restorations, as related to the underlying bone, are determined before surgery. The treatment-planning process starts with visualizing the final restoration and working backward from there with precision.
Challenges to Widespread Adoption
If this technology has been shown to increase implant placement accuracy, decrease surgical complications, and improve the predictability of implant case outcome,9-13 then why has it not been more widely adopted by the dental implant community? The answer can be summed up in three essential factors: time, money, and fear.
Time—It takes additional time for the doctor to make impressions, work up the planned positions of the restorations with a diagnostic wax-up of the planned implant prosthetics, fabricate a radiographic template for the patient to wear while having a CT scan, order or make the CT scan, import the CT data into the desired software, plan the case “virtually” before actually treating the patient, order the surgical guide, and learn the techniques and equipment necessary to place implants through a surgical guide.9 Training courses may be necessary to help clinicians master these technologies, taking time away from their offices.
Money—Both doctors and patients will need to incur additional costs for the laboratory work to fabricate a radiographic scan prosthesis, perform the CT scan, and fabricate the surgical guide. Doctors must also bear additional costs involved in learning the new technologies, possibly upgrading computer hardware, purchasing new software programs, and learning and purchasing new drilling and instrumentation kits.
Fear—Dentists tend to be slow to adopt new technologies into practice, preferring to take a “wait and see” approach and reasoning that because they have successfully planned and placed implants freehand for many years there is no reason to do it another way. Often, there is fear of: change, new technology in clinical practice, failure, frustration, losing respect if unsuccessful, harming patients, losing patients, and litigious patients.9 Further, dentists may be afraid that they will “lose the case” if more costs are presented to the patient and added onto what may be seen as an already expensive treatment plan. Additionally, some implant and software manufacturers are marketing 3-D implant planning products with names that imply fast restorative results, which may lead some doctors to believe that these technologies are only useful in specific, limited, fully edentulous cases. Doctors might also feel that the additional radiation involved in a CT scan may not be warranted.
CT-Guided Surgery Realities
Compared with conventional treatment approaches, “virtually” planning and placing implants does in fact require more preparatory steps before surgery for both the doctor and the patient (including impressions, models, diagnostic wax-ups, CT and cone-beam CT scans, and virtual case planning). Additionally, doctors must commit both time and money to learning these technologies, planning the cases on a computer, and purchasing computer hardware, software, and new surgical instrumentation. However, once they become familiar with the instrumentation, time savings can be realized chairside. (Estimated time savings is subjective and related to the individual clinician.) Most of the time spent treatment planning is not done during office hours.
There is also a practice-building aspect to adoption of these technologies. During an implant consultation, in the author’s experience, patients’ biggest concerns usually involve the amount of pain they will have, what the recuperation period will be, and how long they will be incapacitated or out of work. Patients respond well to the concept of minimally invasive dental implant placement, which can sometimes be done with flapless insertion that minimizes swelling and pain and maximizes their comfort and ability to return to their everyday lives. The concept that their doctor is using state-of-the-art technology is reassuring to patients and helps them build confidence in the doctor. There is typically little need to “sell” this technology to patients, who generally have little trouble seeing its benefits.
Regarding money, the patient cost for the laboratory work to fabricate the radiographic scan prosthesis, perform the CT scan, and fabricate the surgical guide adds to the expense of the treatment plan. Newer digital workflows are more time- and cost-effective, allowing for the importation of optical scans of diagnostic wax-ups and planning using “virtual teeth.” Many patients have been presented with surgical and restorative implant treatment plans that will cost in the multiple thousands of dollars. Commonly, when patients are told that using this technology will require a CT scan with added expense and radiation but that, in the end, the implants will likely be placed in ideal positions, with proper spacing for gingival and bone health, with proper implant angulations to distribute the bite forces well, and with minimal unforeseen issues encountered at the time of surgery, there are few questions asked and they are likely to consent to the treatment.9 The technology can sell itself, and the benefits typically outweigh any financial considerations.9
The Future of Implant Dentistry
Any patient case can be treatment-planned “virtually.” This technology can be used to place single, multiple, or full arches of implants. Implants can be buried (two-stage approach), placed in a single stage (using healing abutments), or immediately loaded. Implants can be placed after extractions or with concurrent bone grafting or reduction procedures. In essence, implants can be placed exactly as in the past but with more precision, greater avoidance of vital anatomic structures, and less pain and swelling for patients.10-14 Many consider these technologies and workflows to be the future of implant dentistry.
About the Authors
Gary Orentlicher, DMD
Section Chief
Division of Oral and Maxillofacial Surgery
White Plains Hospital
White Plains, New York
Private Practice
New York Oral
Maxillofacial, and Implant Surgery
Scarsdale, New York
Andrew Horowitz, DMD, MD
Associate Attending
White Plains Hospital
White Plains, New York
Private Practice, New York
Oral, Maxillofacial, and Implant Surgery
Scarsdale, New York
Marcus Abboud, DMD
Associate Professor and Chair
Department of Prosthodontics and Digital Technologies
School Of Dental Medicine
Stony Brook University
Stony Brook, New York
References
1. Abboud M, Orentlicher G. Computer-aided manufacturing in medicine. Atlas Oral Maxillofac Surg Clin North Am. 2012;20(1):19-36.
2. Campelo LD, Camara JR. Flapless implant surgery: a 10-year clinical retrospective analysis. Int J Oral Maxillofac Implants. 2002;17(2): 271-276.
3. Becker W, Goldstein M, Becker BE, Sennerby L. Minimally invasive flapless implant surgery: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S21-S27.
4. Becker W, Wikesjö UM, Sennerby L, et al. Histologic evaluation of implants following flapless and flapped surgery: a study in canines. J Periodontol. 2006;77(10):1717-1722.
5. van Steenberghe D, Ericsson I, Van Cleynenbreugel J, et al. High precision planning for oral implants based on 3-D CT scanning. A new surgical technique for immediate and delayed loading. Appl Osseointegration Res. 2004;4:27-31.
6. Wendelhag I, van Steenberghe D, Blombäck U, Glauser R. Immediate function in edentulous maxillae with flapless surgery including a 3-D CT-scan based treatment planning procedure [abstract]. Clin Oral Implants Res. 2004. Abstract poster 144.
7. Sanna AM, Molly L, van Steenberghe D. Immediately loaded CAD-CAM manufactured fixed complete dentures using flapless implant placement procedures: a cohort study of consecutive patients. J Prosthet Dent. 2007;97(6):331-339.
8. van Steenberghe D, Glauser R, Blombäck U, et al. A computed tomographic scan-derived customized surgical template and fixed prosthesis for flapless surgery and immediate loading of implants in fully edentulous maxillae: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S111-S120.
9. Orentlicher G, Goldsmith D, Horowitz A. The power of 3-D computer-generated implant planning and surgery. Selected Readings Oral Maxillofac Surg. 2009;17(6):1-32.
10. Cassetta M, Giansanti M, Di Mambro A, Stefanelli LV. Accuracy of positioning of implants inserted using a mucosa-supported stereolithographic surgical guide in the edentulous maxilla and mandible. Int J Oral Maxillofac Implants. 2014;29(5):1071-1078.
11. Schneider D, Marquardt P, Zwahlen M, Jung RE. A systematic review on the accuracy and the clinical outcome of computer-guided template-based implant dentistry. Clin Oral Implants Res. 2009;20(suppl 4):73-86.
12. Farley NE, Kennedy K, McGlumphy EA, Clelland NL. Split-mouth comparison of the accuracy of computer-generated and conventional surgical guides. Int J Oral Maxillofac Implants. 2013;28(2): 563-572.
13. Cassetta M, Stefanelli LV, Giansanti M, Calasso S. Accuracy of implant placement with a stereolithographic surgical template. Int J Oral Maxillofac Implants. 2012;27(3):655-663.
14. Sarment DP, Sukovic P, Clinthorne N. Accuracy of implant placement with a stereolithographic surgical guide. Int J Oral Maxillofac Implants. 2003;18(4):571-577.