Prebiotics, Probiotics, and Oral Microbial Wellness
John G. Thomas, MS, PhD
A balanced oral microbial environment is essential for the promotion of health and prevention of disease. Bacteria are a major component of the oral environment where more than 800 different microbial species co-exist, making the oral cavity the most diverse microbial population of the body.
Complex ecosystems also exist in the gastrointestinal tract, the urogenital tract, and the skin. Complex and dynamic relationships between microbiota exist not only within but between these different ecosystems.
Under normal conditions, the diversity of the oral flora is well balanced and the microorganisms work together as a natural defense system. If this protective oral balance is disrupted, harmful bacteria proliferate and a disease-induced state may ensue. The ability to maintain the delicate balance of the microbial flora within the biofilm is important to the maintenance of an environment conducive to good health.
The diversity of the normal flora is extensive. There are 2 million genes in the bacterial genome versus 35,000 in the human. Within the human microflora there are 100 trillion living bacteria and over 800 species in the oral cavity.
There are four predominant anatomic reservoirs of bacteria within the human body: gastrointestinal, urogenital, mouth/sinus, and skin. Table 1 illustrates the community and diversity of these reservoirs by noting their location and the amount of microbes that are present. The essential ratio of the organisms within the four anatomic sites provides the bacterial bioburden and defines the diversity. Although the gastrointestinal region has the highest diversity ratio when compared to the mouth/sinus region (1000:1), the mouth/sinus region has the most diverse bacterial reservoir level (1000).
The oral cavity and maxillary sinus are now beginning to be recognized as the most biodiverse area within human microbiology (Figure 1). Because the sinus is continuous with the oral mucosa, it is simultaneously colonized by multiple microorganisms. A sustained reinnoculation from the four predominant anatomic sites creates a microbial community of continual diversity. An extensive number of niches, or sites, harbor many pools of disparate organisms, in varying degrees of concentration. It is a continually evolving environment where niches are in a constant state of change to increase the density of their populations.
Biofilm is the preeminent form by which colonies of microbes grow, proliferate and mature. It is a primitive type of developmental biology that is more established and coordinated and allows microorganisms to support growth to one another, enabling optimal utilization of available nutritional sources. Intraoral sites harbor organisms that are not only free floating, but survive and thrive in this biofilm structure. The term biofilm was first described in the 1980s as engineers observed that water did not flow freely through filters contaminated with a film. Biofilms have now been recognized in many diverse systems from oil pipelines, endotracheal tubes, and dental unit waterlines. The organization of the microbial population in a biofilm structure is very well defined. Biofilm organisms will attach, adhere, and grow as a community, resembling a viable organ system.
Dental plaque is organized both structurally and functionally as biofilm. Plaque develops in a prescribed way with a varied microbial composition. Species of organisms from diseased sites differ phenotypically from those encountered in healthy sites. A microorganism in a biofilm is much harder to kill than when freely floating in a planktonic state.
The development of dental plaque is very organized, a cascade of gram-positive cocci (initial or pioneering colonizers) to gram negative rods (secondary colonizers) (Figure 2). As the microbial community grows in diversity and complexity, the recalcitrant biofilm reflects a simple, co-biofilm, and complex 3-D architecture.
Stewardship of this microbial world and ongoing maintenance are major factors in dealing with disease processes. Stewardship calls for a shift in thinking from simply killing the pathogenic microbes to creating an ecosystem where healthy microbes predominate and as such inhibit excessive growth of pathogenic microbes. The daily use of probiotics helps maintain the balance of microbial flora and thus helps to promote overall health.
Pathophysiology and Changing Paradigms
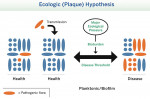
There are changing paradigms associated with the pathophysiology of biofilms and disease. The ecological or plaque hypothesis was originally described by P.D. Marsh, and involved the “We” Theory. His studies focused on dental plaque as a biofilm and a microbial community, as well as the active relationship between the environment and the structure of the oral microflora. A holistic concept to manage and control disease involved directly inhibiting pathogens and interfering with environmental factors driving the selection of these bacteria.1
The plaque hypothesis postulates that these biofilm communities are more established and more coordinated. Each of these coordinated communities has its own means of communication, permitting the organisms to gather information from one another and to enable supportive growth. It defines a steady state that is stressed and the availability of nutrients defines the structure, so that again structure equals function. Critical colonization is a microbiologic term that defines the ratio of organisms that are present. It is a situation where diverse organisms live in multiply defined communities. This idea differs from the earlier “one organism” concept of disease causation advanced by Koch’s postulates. While Pasteur and Koch together are regarded as the fathers of germ theory,2 Koch was the first scientist to devise a series of tests used to assess the germ theory of disease: one organism—one disease (or the “I” theory).3 The accepted concept today is that there are multiple organisms with the ability to interact in multiple ways. The means of bringing these biofilm communities back into balance is best achieved not through use of antimicrobials, but by reestablishing a normal flora, aided by probiotic agents.
Figure 3 illustrates the ecologic plaque hypothesis. In a healthy state the microbial pool contains a microbiota that is generally balanced and non pathogenic, with minimal pathogenic flora. Given the opportunity and further pressure from the environment, pathogenic flora may become predominant. Achieving a state of equilibrium among these multiple-interacting organisms is best achieved not by the use of antimicrobials, but by reestablishing an ecologically or well-balanced microbiota.
Prebiotics and Probiotics—Replacement Therapy
While probiotics are the beneficial bacteria themselves, prebiotics are functional food ingredients that are the carriers and provide a complementary delivery system to the probiotics. By acting as a food source, prebiotics give the probiotic bacteria a chance to exert their influence by aiding digestion and boosting immunity. Stress, a poor diet, certain medical conditions, medications, and other factors may inhibit these beneficial bacteria. It is estimated by the World Health Organization (WHO) that $85 billion will be spent on probiotics by 2020. This expected increase in popularity signals a growing interest in probiotics as a means of maintaining a balance in oral and overall health.
Probiotics are living microorganisms that are non-toxic and provide a benefit to a host organism. The WHO defines probiotics as, “Live microorganisms which when administered in adequate amounts confer a health benefit on the host.” 4 Researchers have also observed that certain organisms, primarily the most common species of lactobacillus or bifidobacterium, have health promoting benefits and may be used as probiotics. The mechanisms of action for probiotics are based on the concept of elimination or reduction of plaque biofilm.
The concept of probiotics was first introduced in the 1870s by Russian scientist and Nobel Laureate Éli Metchnikoff, who observed that lactic acid bacilli had a way of balancing gut flora. He suggested that it would be possible to modify the gut flora by replacing harmful microbes with useful microbes. He explained that “Probiotics are viable bacteria that beneficially affect the host by improving its intestinal microbial balance.” 5
Lactobacillus is the genera which has the greatest capacity to form a biofilm that permits the reestablishment of a normal flora and undermines the cascade of events that cause tissue damage.
Prebiotics are food ingredients that stimulate the growth and/or activity of bacteria in the digestive system, in ways claimed to be beneficial to health. Marcel Roberfroid offered a refined definition in the Journal of Nutrition6 stating, “A prebiotic is a selectively fermented ingredient that allows specific changes, both in the composition and/or activity in the gastrointestinal microflora that confers benefits upon host well-being and health.” Prebiotics effectively stimulate the colonization of the probiotic microorganisms, providing an initial advantage to their adherence.
Probiotic supplements have been specifically designed to achieve and maintain optimal oral health (eg, G•U•M® Perio-Balance®, Sunstar Americas, Inc., www.sunstaramericas.com.) These special blends of microorganisms may be used as an adjunct to existing oral health care regimens. Probiotics can be delivered via a wide range of vehicles including lozenges, straws, tablets, capsules and liquids, rinse solutions, cheese, yogurt, and yogurt drinks. Recent advances and breakthroughs have been made in probiotics that help clinicians manage the overall health of their patients. One example is the use of a lozenge as a probiotic delivery system. Lozenges have clinical and manufacturing advantages. They are easy to use, do not adhere to oral surfaces, and are stable when held in storage. G•U•M PerioBalance is a probiotic product offered in lozenge form.
The diversity of microorganisms included as “Probiotics” is astounding and reflective of the ongoing research. The two most common genera are Lactobacillus and Bifidobacterium, each having multiple species evaluated (Table 2). The benefits of two organisms, each with a specific target, highlight the greater clarification of mechanism of action of individual species, recognizing an optimum two-pronged approach: 1) reduce the existing pathogens; and 2) decrease the detrimental consequences of an immunologic cascade. To accomplish this task, a variety of carrier vehicles have been utilized, as shown in Table 3. Historically, most required refrigeration, but new options are stable at room temperature and reflect a global benefit with minimal refrigeration. Further, prebiotics and probiotics show proven benefits for hospitalized patients that have experienced an overuse or misuse of antibiotics.
A comprehensive oral-care approach to re-establishing normal flora includes the traditional mechanical methods (eg, brushing and flossing, scaling and root planing), in combination with chemical means (eg, antiseptic mouthrinses) and probiotics to create a beneficial and balanced normal flora environment. Restorative microbiology is a process that includes all these oral care approaches with the need to tailor according to specific patient requirements.
Conclusion
Probiotics are not just adjunctive prescriptions but should be considered as a key component of initial therapy directed at the establishment and maintenance of a normal flora. This method is the optimum defense mechanism against overgrowth and changes within the body associated not only with oral flora but also with systemic flora. The advent of probiotics for use in the oral cavity presents dental professionals and their patients with yet another line of therapy to support existing and well-proven mechanical removal of plaque biofilm through toothbrushing and interproximal cleaning, anti-plaque (antimicrobial or anti-adhesive) rinses, and scaling and root planing. Whereas the latter approaches are all about removal, the probiotic approach is complementary by encouraging the growth of mouth-friendly micoflora.
Disclosure
The author has received an honorarium from Sunstar Americas, Inc.
References
1. Marsh PD. Are dental diseases examples of ecological catastrophes? Microbiology. 2003;149(2):279-294.
2. Ullmann A. Pasteur-Koch: distinctive ways of thinking about infectious diseases. Microbe. 2007;2(8):383-387.
3. Madigan MT, Martinko JM. Brock Biology of Microorganisms. 11th ed. Upper Saddle River, NJ: Pearson Prentice Hall; 2006.
4. Food and Agriculture Organization of the United Nations, World Health Organization. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria. https://www.who.int/entity/foodsafety/publications/fs_management/en/probiotics.pdf. Published October 4, 2001. Accessed: November 4, 2009.
5. Metchnikoff E. The Prolongation of Life. Optimistic Studies. Mitchell PC, trans-ed. London: Heinemann, 1907.
6. Roberfroid M. Prebiotics: the concept revisited. J Nutr. 2007;137(3)(suppl 2):830S-837S.
About the Author
John G. Thomas, MS, PhD
Professor
West Virginia University
Director
Biofilm Research Laboratory for Translational Studies in Medicine, Dentistry, and Industry
West Virginia University
Morgantown, West Virginia