Preparing Porcelain Surfaces for Optimal Bonding
Gary Alex, DMD
Abstract
When placing an indirect restoration such as a porcelain veneer, inlay, or onlay, the intaglio surface is typically treated in some fashion to ensure optimal bonding to the resin-based adhesives and luting cements required to affix them to the tooth tissues. A variety of surface preparation techniques have been advocated, including the use of acids, particle abrasion, various adhesives, and chemical couplers such as silane. There is often equivocation in the literature regarding the effectiveness of these materials and surface preparation techniques. This article will attempt to make some sense of the various methodologies currently advocated for the treatment of porcelain surfaces prior to placement.
Optimal surface preparation techniques for chemical and/or mechanical bonding to porcelain substrates are crucial in order to ensure clinical success when placing indirect porcelain restorations and, when required, repairing them intraorally. Clinicians are often confused regarding the most effective way to treat the intaglio surfaces of indirect porcelain restorations prior to placement with various adhesives and luting resins. They are often equally perplexed about the ideal surface treatment for the intraoral repair of preexisting porcelain restorations. This is not surprising, as there appears to be no clear consensus in the dental literature, among opinion leaders, or from dental manufacturers on exactly what the optimal surface treatment should, in fact, be. Dental laboratory technicians also appear to lack standardized protocols on how they should treat the surface of finished porcelain. For example, in 2007, this author spoke with the owners and/or ceramists from 11 different dental laboratories regarding their specific surface treatment protocol prior to sending out finished feldspathic porcelain veneers. All treated the inner surface of the veneers with hydrofluoric acid (HF). Only two knew the actual concentration of the HF they were using. Application times varied from as short as 30 seconds to as long as 10 minutes. Several laboratory technicians stated they simply etched the veneers until the surface looked frosty. Some laboratories sandblasted the veneers prior to HF treatment and some did not. After etching, some laboratories placed the veneers in an ultrasonic cleaner, some steam cleaned the veneers, some placed the veneers back in the oven at a low temperature to burn off any white residue left over from etching, and others merely rinsed them with tap water. Three laboratories stated they sometimes used a basic solution to neutralize the acid after HF treatment, although they were not sure if this was a necessary step. This author also questioned approximately 24 opinion leaders at a recent symposium1 regarding their recommendations for the intraoral repair of both composite and porcelain restorations. While there were many similarities, there were also significant differences, and no treatment protocols were exactly the same.
The lack of clear and consistent guidelines regarding the treatment of composite and porcelain surfaces raises several significant questions. Is there, in fact, an optimal surface treatment protocol for porcelain restorations prior to placement and for intraoral repair? Is it possible that several viable surface treatment options exist? Is it even practical to recommend a specific universal treatment protocol because of material differences (eg, not all porcelains are the same)?
This article will attempt to address these questions, provide some general guidelines, and no doubt raise additional questions regarding the management of porcelain surfaces. A good place to start is by examining what we think we know.
Bonding to Porcelain Use of HF
HF is an inorganic acid capable of etching glass surfaces. It has been used for hundreds of years to create decorative designs and patterns on glass and various ceramic materials.2 It is also used as a precursor in the manufacturing of numerous pharmaceuticals, various polymers (eg, Teflon), and many other synthetic materials that contain fluorine.3 Contrary to what most dentists believe, because of its low tendency to dissociate into H+ and F- ions, HF is considered to be a relatively weak acid from a chemical standpoint. This doesnt mean HF is not dangerous. Quite the contrary; due to its low dissociation potential, HF has the ability to readily penetrate skin tissues (often without causing an external burn), where it can cause extensive internal tissue damage, as well as alter blood calcium levels (due to the formation of CaF2), which can lead to dangerous heart arrythmias.4 There have been industrial accidents where death has occurred due to accidental skin exposure to HF.5 The duration of exposure, the concentration of HF, and the total surface area affected will obviously have an effect on clinical outcomes relating to accidental dermal exposure.6 In typical dental applications (eg, the etching of porcelain veneers and the intraoral repair of fractured porcelain restorations), concentrations of 4% to 10% HF are typically utilized. In this authors opinion, HF in this concentration range can be used safely for dental procedures, including intraoral repair, provided caution and common sense are employed. Whenever HF acid is employed for intraoral repair, the use of appropriate barrier techniques, viscous HF gel formulations (ie, that stay where you put them), and continual visual observation during the application period are mandatory. For those dentists not comfortable with the intraoral use of HF, acidulated phosphate fluoride gel (APF) has been shown to have the ability to etch porcelain, although the application time must be considerably longer than for HF etching. One recent study showed that a seven- to 10-minute application of 1.23% APF gel on a leucite containing porcelaina produced a shear bond strength to compositeb similar to a four-minute etch with 9.6% HF.7 It should be pointed out that this same study showed that etching with 9.6% HF for four minutes resulted in a very definitive microscopic etching pattern, while etching for seven to 10 minutes with 1.23% APF resulted in a pitted, but much smoother-appearing surface. This difference in surface morphology could be significant over the long term, and it would have been interesting to see what effect thermocyling of the samples, which was not done, would have had on bond strength. Also, it is unclear why the investigators used 9.6% HF for four minutes when the manufacturer of the porcelain tested recommends that 5% HF acid be used for only 60 seconds. It is possible that the bond strength to the HF-treated porcelain samples would have been even higher had the manufacturers recommended protocol been followed. Other studies have also shown that etching with APF, even with prolonged application times, results in very shallow etching patterns when compared to HF etching for much shorter time periods.8 Silica coating, followed by silane application, has also been explored as an alternative to HF etching of feldspathic-based porcelains. The authors of one study found that both the high and low leucite containing feldspathic porcelains they tested had significantly higher tensile bond strength to composite when samples were first etched with HF followed by silane treatment, vs. just silica coating followed by the application of silane.9 Still another study looking at alternatives to HF etching found that simply roughening porcelain surfaces, followed by treatment with 35% phosphoric acid gel and silane priming, was as effective as HF-treated samples to bonded composite.10 Once again, thermocyling of the samples, which was not done, might have had a significant effect on the results. Other products also exist and are marketed as alternatives to HF etching of porcelain.
In this authors opinion, while other alternatives do exist, they are less predictable, especially over the long term, when compared to HF-etched porcelain. Numerous in vitro studies clearly demonstrate that etching feldspathic-based porcelain with HF has the potential to significantly increase its bond strength to composite resin.10-16 Dental porcelain generally consists of an amorphous glassy phase or matrix and a crystalline phase. Treating porcelain with HF tends to selectively dissolve the glassy matrix, resulting in a microscopically porous, high-energy, and microretentive surface12,17,18 (Figure 1). In principle, this is similar to what happens to enamel surfaces after etching with phosphoric acid.
Several important questions arise when using HF to etch porcelain. Is there an optimal etching time and concentration of HF that should be used? Can porcelain be overetched? What is the white residue often seen on the surface of the porcelain after etching with HF, and how should it be handled? The first question is not easily answered. This is because the chemical and physical makeup of dental porcelain is variable, depending on the type of ceramic being utilized. Years ago, Calamia recommended adjusting the etching times and concentration of HF depending on the specific porcelain being treated.19 While all feldspathic porcelains contain silica dioxide (SiO2), aluminum oxide (Al2O3), and potassium oxide (K2O), the relative amount of each can vary considerably depending on the specific porcelain being utilized. Alumina content, crystalline microstructure, size, and concentration can all influence HF etching times and concentrations.11, 20, 21 As an example, leucite crystals, which affect optical, thermal, and physical properties, are incorporated into the matrix of many dental porcelains.22,23 A study by Barghi showed a 50% leucite content porcelainc required a longer etching time (ie, 150 to 180 seconds) with 10% HF when compared to a 27% leucite content ceramicd that only required a 60-second etch with 10% HF.24 Some manufacturers recommend very specific HF concentrations and etching times for their ceramics. The manufacturer of one popular pressable ceramic,a which has a reported leucite concentration of 35% to 40%, recommends etching for 60 seconds with 5% HF.25,26 Longer etching times or higher concentrations of HF with this particular ceramic may actually result in over-etching and weakening of the porcelain substrate by excessively degrading the surface.26 At least one study supports this contention and found that extending the etching time of this particular ceramics ingots from one to two minutes with 5% HF significantly decreased the biaxial flexure strength of the ceramic.27 The manufacturer of another popular pressable leucite reinforced ceramice recommends lightly sandblasting the internal surface with 50 µm Al2O3 at a maximum of 20 PSI and then placing 5% to 12% HF for 90 seconds to five minutes.28 One logical interpretation of this is that dentists could use 12% HF for 90 seconds or 5% HF for five minutes. So, one manufacturera recommends etching with 5% HF for one minute when treating the inner surface of its leucite containing pressable ceramic, and another manufacturere appears to recommend etching with 5% HF for five minutes when treating its leucite containing pressable ceramic. Why there is such a difference in recommended 5% HF application times between these two ceramics is difficult to understand. It is certainly possible that a difference in leucite concentration, size/orientation of crystals, or microstructure might affect etching times. According to one source,29 the leucite concentration for the aforementioned popular pressable ceramice is approximately 8%, depending on the shade. This author was unable to corroborate this information despite an extensive Internet search and several phone and e-mail contacts with representatives of the manufacturer. At least one company representative suggested etching the ceramic with 7% HF for one to two minutes, although it was unclear on what this recommendation was based.30
Two lithium disilicate-based ceramicsf,g replaced a now defunct producth.26 The manufacturer of these products recommends a very specific etching time of only 20 seconds with 5% HF,25,26 and studies exist supporting this protocol.17,31 Powder/liquid or stackable porcelainsi,j,k,l,m,n,o,p are used extensively in the fabrication of ceramic restorations. Some opinion leaders/studies recommend 90 seconds of etching with 10% HF for stackable feldspathic porcelains,32 while others recommend a 120- to 150-second etch with 9.5% HF.33 The manufacturers of two popular 9.5% HF etching gelsq,r recommend etching times of 90 seconds (ie, the former manufacturer) and 60 seconds (ie, the latter manufacturer).34
In an unpublished study, Byoung Suh tested 10 different feldspathic porcelainss,t,u,v,w,x,y by HF etching (ie, 9.5% and 4.0%), followed by contact angle analysis and microscopic evaluation. He first particle-abraded the samples (ie, sandblasting) and found they all showed an acceptable microscopic etching pattern after a four- to five-minute etch with 4% HF. These same porcelains showed a similar etching pattern when 9.5% HF was used for 90 seconds.35 The authors of another study found statistically higher composite shear bond strength to several stackable porcelain disks when they were etched with 10% HF for two minutes as opposed to a longer etch for four minutes at the same HF concentration. They attributed the lower bond strength with the longer etching time of four minutes to over-etching and actual weakening of the porcelain surface.36 The man-ufacturer of a popular laboratory ceramic etching gel sells a 12% to 13% HF etching gel for stackable porcelainsz and recommends what, to this author, appears to be a rather long etching time of 10 to 15 minutes. According to the manufacturer, this application time was based on observation of the contact angle of water droplets placed on ceramic surfaces and the microscopic appearance of the porcelain surface after etching for different time periods (no shear or tensile bond testing was performed).37 Interestingly, this same manufacturer sells another product marketed for pressed ceramicsaa that uses 5% HF with a recommended etching time of only 30 to 60 seconds.
Most dentists and ceramists have noticed that a white residue sometimes remains on the surface of HF-etched porcelain restorations. At times this residue can be quite extensive and cover virtually the entire etched porcelain surface. The extent of this precipitate is no doubt related to the type of porcelain, concentration of HF, and the time of application. One study, through energy dispersive spectrometer analysis, determined this residue to consist of the reaction products of porcelain and HF.8 Basic chemistry has shown that when an acid and base react, various salts are produced as byproducts of the reaction. In the case of HF etching, the porcelain acts as a base, and when it reacts with HF, various insoluble metallic salts are formed.31 In this authors opinion, the white residue consists not merely of porcelain salts, but also of numerous microscopic crystalline fragments exposed, possibly weakened by HF exposure, and displaced after the glassy matrix supporting them has been dissolved by HF and the porcelain surface washed and dried. This water insoluble crystalline residue/salt mix rather tenaciously adheres to the porcelain surface and is often difficult to remove. While many believe this white frosting to be indicative of a good etch of the porcelain, this is probably an inaccurate assessment. In fact, in this authors opinion, if the white precipitate is excessive, this may be indicative of over-etched porcelain (Figure 2). Several authors have discussed the potential for over-etching porcelain and how this could adversely affect porcelain physical properties and/or bond strength to composite15, 27, 36, 39-42 In any case, this surface residue is a potential contaminant, and it makes sense to remove it before proceeding to the bonding phase of treatment.
In this authors experience, removal of this material with water alone is usually not effective, even after a vigorous air/water spray. Acetone or alcohol rubbing is only slightly more effective. This author has found that placing the etched restorations in an ethanol solution followed by ultrasonication for five minutes is usually, but not always, effective in removing any residue (Figures 3A, Figure 3B and Figure 3C ). Sometimes light brushing with a toothbrush and/or longer ultrasonication times are also required. Other authors have recommended a similar protocol using distilled water.32 This author prefers an alcohol solution because it evaporates more easily than water and is probably a better solvent when it comes to removing potential contaminates (eg, grease and oils from handling, surfactants from acid gels, saliva, try-in paste residue, etc.). Other authors have recommended the use of steam cleaning to remove any residue/salts left after HF etching of porcelain.43 In any case, the idea is to have an etched, clean, reactive, and high-energy surface prior to the next phase of porcelain surface preparation (ie, treatment with a silane coupling agent).
Bonding to Porcelain Use of Silane
Silanes are a class of organic molecules that contain one or more silicon atoms. Dozens of different silane compounds exist, and they are used extensively in industry and manufacturing. The specific silane typically used in dentistry for both intraoral repair and treatment of ceramic restorations prior to placement is 3-methacryloxypropyltrimethoxysilane. Note: all other use of the term silane in this article will refer specifically to this monomer.
The silane most often used in dentistry is the 3-methacryloxypropyltrimethoxysilane, which is a difunctional molecule. The left side of this molecule is nothing more than a methacrylate group capable of co-polymerization with methacrylate-based adhesives and resins routinely used for dental procedures. The right side, after hydrolysis, has the potential to form chemical bonds to the porcelain surface.
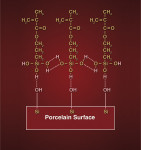
In dentistry, silane is potentially useful as a chemical coupler linking organics (ie, resin-based materials) to inorganics (eg, porcelain, some oxidized metals, and glass fillers in resin-based composites). A number of theories ranging from its ability to simply act as a good wetting agent to the actual formation of covalent chemical bonds at the involved interfaces exist regarding how silane actually functions.44-46 Note: this author highly recommends an excellent paper on silane chemistry and interactions written by Meyer Rosen in 1978 while working for Union Carbide.44 Infrared spectroscopy has shown that silane has the potential to react with hydroxyl (-OH) groups present on the surface of porcelain and metal substrates.46-48 To be able to function as a coupling agent and interact chemically with porcelain surfaces, silane must first be hydrolyzed.45 Acetic acid is commonly used to activate or hydrolyze silane by reacting with the three methoxy (-OCH3) groups located at one end of the silane molecule (Figure 4). Ideally, all three of the terminal methoxy groups will convert to hydroxyl (-OH) groups, although only one or two may actually react, resulting in incomplete hydrolysis and a potentially less effective silane.46,47
Single-bottle silanes are pre-hydrolyzed by the manufacturer and, while variations in chemistry exist, typically consist of 1% to 5% silane in a water/ethanol solution with an acetic acid adjusted pH of 4 to 5.44,45 Once hydrolyzed, silane molecules have a tendency to react with one another, forming high-molecular weight oligomers44,47 (ie, polysiloxanes) that can actually function as a lubricant49-51 and potentially decrease bond strength to porcelain. It is important to remember that one-bottle pre-hydrolyzed silane solutions have a limited shelf life, and this author recommends refrigerated storage and replacement after one year, as well as bringing refrigerated silane to room temperature prior to use. If the silane solution appears at all cloudy or milky in appearance, or any type of precipitate is noticed, it should be discarded.
Two-bottle silane systems typically consist of an unhydrolyzed silane/ethanol solution in one container and an acetic acid/water solution in the other.35 These are mixed together by the clinician to hydrolyze the silane prior to application. It is unclear to this author precisely how much time is required for an acceptable degree of hydrolysis to occur once the solutions of two-part silane systems are mixed. The authors of one paper state 0.5 to 2 hours should be sufficient,45 while another author cites a study showing hydrolysis was complete 10 minutes after mixing with 0.1% acetic acid.44 The authors of another study determined the hydrolysis rate of silane by the use of Raman spectroscopy and determined that 30 to 50 minutes was required.52 Still another study found that the two-part systembb they tested did not have the same degree of hydrolysis 20 minutes after activation when compared to the two pre-hydrolyzed systems they tested.47 One reputable manufacturer and respected researcher stated to this author that, based on contact angle analysis and in-house testing, its two-part silane was effective immediately after mixing, but performed optimally if left alone on the porcelain surface for five minutes after it was applied.35 This is probably an indication of further hydrolysis of the silane and/or increased chemical interaction with the porcelain during the waiting period. Two-bottle systems should have a longer shelf life than pre-mixed systems and might be a better choice for dentists whose use of silane is infrequent.
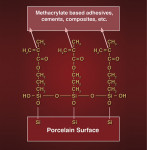
The important point to remember is that the goal of silane hydrolysis is to create terminal hydroxyl groups on each silane molecule. These hydroxyl groups are then capable of reacting directly with corresponding hydroxyl groups present on the surface of feldspathic porcelain (through oxidation of SiO2). The opposing hydroxyl groups interact with one another (via hydrogen bonding) and then, through a condensation polymerization (ie, loss of water) reaction, covalent bonds are formed between the silane and porcelain. Not only does the silane bond directly to the porcelain in this fashion, but the individual silane molecules bond to one another, forming a polymer network on the porcelain surface44 (Figure 5 and Figure 6). In theory, the ideal situation would be one in which a monolayer of silane molecules lines the surface of the porcelain.45 Clinically, this is not possible, but it still makes sense to use the minimum amount of silane required, and this author recommends that no more than one to two coats be placed. Excessive application of silane could, in principle, create too thick a coating by consecutive silane layers bonding to each other, one on top of another, and creating an unnecessarily thick and intrinsically weak layer, which could be prone to cohesive failure. Clinically, the surface of the porcelain should NOT look shiny after silane application and drying. In this authors opinion, a shiny surface on the porcelain after silane application and drying could be an indication of excessive silane deposition and, if seen, the surface should be sandblasted under low pressure, re-etched with HF, cleaned with ethanol in an ultrasonic, and the silane re-applied. A properly silane-treated porcelain veneer visually appears essentially the same as it did prior to placing the silane (ie, matt/dull finish). Numerous studies have shown that heat treatment of silane-treated porcelain has the potential to significantly improve bond strength to composite.41,51,53-56 This author recommends that warm-air drying be used for 60 seconds to dry the surface of the porcelain after silane application. This is a simple and clinically feasible chairside procedure that has been shown in some studies to significantly improve bond strength of silane-treated porcelain to composite.51,54,55 Complete removal of water is critical as it is the basis behind the condensation polymerization reactions that allow silane to actually bond to porcelain. Warm/dry air is simply very effective at removing alcohol and water from the surface, and by heating up the substrate, one can speculate that the reaction rate will be accelerated, molecular interactions become more frequent, and the formation of chemical bonds occurs in greater numbers. Once the inorganic end of the silane molecule is bonded to the porcelain, the methacrylate group on the other end is free to bond (via free radical addition polymerization) with methacrylate groups in resin-based dental materials and adhesives (Figure 6). While there is equivocation in the literature,11 it appears that most studies find proper use of silane significantly improves the bond strength of resin-based composites to porcelain.10,15,57-60
Bonding to Porcelain: High-content Alumina and Zirconia Core-based Ceramics
High-content alumina and zirconia core-based ceramicscc,dd,ee,ff,gg,hh,ii are highly resistant to chemical attack from HF,11,61-63 and a different approach is required if the clinician elects to bond these restorations into place using resin-based adhesives and luting cements (as opposed to conventional cementation). One method that has been shown to be quite effective in increasing bond strength to these materials is the technique of silica coating followed by silane application.61-63 One simple and effective technique utilizes silica coated 30 µm aluminum oxide particlesjj followed by the application of silane. According to the manufacturer, sandblasting with this material utilizes impact energy to apply a silica coating to the target surface.64 Whether this transfer of silica is due to particles actually becoming embedded in the target surface, actual mechanical/chemical transference (ie, tribochemistry), or both, is unclear to this author. In this authors opinion, it is unlikely that, in the case of high alumina or zirconia cores, there is actual imbedding of silica-coated particles due to the intrinsic hardness of the target material. It may be more feasible that the silica-coated particles actually bounce off these ceramics, but prior to doing so there is an actual transference of silica from the particles to the target substrate (ie, tribochemistry). In any case, this technique has proven to be effective (more so than conventional sandblasting) not just with high-strength alumina and zirconia-based ceramics, but also when bonding to composite65,66 and metal surfaces.67-69 One can speculate that by silica-coating metal or composite substrates, not only is the surface mechanically roughened, but the number of available hydroxyl groups for surface silane coupling is significantly increased. Silica coating is not effective, or required, with conventional feldspathic porcelains simply because significant amounts of SiO2 and free hydroxyl groups are already present (much more so than metals and composites). If the dentist opts to conventionally cement zirconia or high alumina core-based crowns/bridges, silane is not required. However, this author still recommends briefly sandblasting the inner surface of the restoration under low pressure (ie, approximately 20 PSI), followed by placement with whatever conventional cement is chosen by the dentist. Note: this author prefers resin-modified glass ionomer cements.kk,ll Care should be taken when sandblasting these restorations, making sure to use low pressure for short time periods, as at least one study has found that sandblasting with 50 µm AL2O3 (at 40 PSI) had the potential to induce flaws in dense alumina and zirconia ceramic plates, resulting in decreased physical properties.70
Understanding All of This
After a fairly extensive literature review, numerous discussions with manufacturers, laboratory technicians, and researchers, it is apparent to this author that no single specific HF concentration and application time exists that is optimal for etching all porcelains. As a consequence, it is not surprising that currently recommended HF concentrations and application times vary significantly. It also appears that there is potential to both under-etch and over-etch porcelain. As Calamia suggested years ago,19 HF concentrations and application times should, ideally, be adjusted depending on the specific nature of the porcelain being treated. In this regard, dentists are somewhat at the mercy of manufacturers, and it should be incumbent on them to provide accurate and scientifically supported recommendations on precisely how to optimally treat and place their specific product(s). It is then incumbent on dentists and ceramists to actually follow these protocols. As it is, even when specific protocols do exist, this author suspects they all too often are ignored, not understood, or dentists and ceramists are simply not aware of them. In regards to etching porcelain, one goal should be to determine the time frame required, at a very specific HF concentration, that will adequately etch various porcelains without excessively degrading and/or weakening the substrate.
In any case, while recognizing that exceptions are likely to exist, it is probably safe to make some generalizations regarding porcelain surface treatment.
- It appears that low-pressure sandblasting, followed by 60 to 120 seconds of etching with 9% to 10% HF, has scientific support and validity when treating conventional powder/liquid stackable porcelains. A variation that also appears effective is sandblasting followed by 4% to 5% HF applied for four to five minutes.
- In the case of a popular pressable ceramic,a the recommended treatment protocol of 5% HF applied for 60 seconds has scientific support and should be followed. The manufacturer of this particular product is one of the few that this author found to have very specific recommendations, as far as HF concentrations and application times, for a number of its bondable ceramics. Specific manufacturer recommendations for many of the other pressable and/or high leucite containing ceramics are vague, do not exist, or could not be found and/or verified by this author. Some ceramic manufacturers shift responsibility by simply recommending following the directions of the manufacturer of whatever HF-etching gel or liquid is being utilized. As a result, specific treatment protocols appear to be largely speculative. Perhaps these are the restorations many ceramists stated (to this author) that they simply etch until the surface looks frosty. While this is certainly not very scientific, and likely not optimal, in the absence of specific guidelines, it may be the best ceramists and dentists have in some situations.
- High-strength alumina and zirconia core-based crowns cannot be etched with HF. As a result, it is not possible to bond these restorations with conventional HF/silane treatment. Silica coating, followed by silane application, has been shown to be a viable alternative.
- Proper use of hydrolyzed silane, in conjunction with warm-air drying, has scientific support and is advisable after HF etching of feldspathic porcelains or silica coating of high alumina, zirconia, metal, and composite surfaces
Treatment Protocols
Two general treatment protocols successfully used by the author for surface treatment of stacked (powder/liquid) porcelain veneers prior to placement are presented below. These protocols may not be valid for all stacked porcelains and when available, manufacturer recommendations regarding etching times and HF concentrations should be followed. The first protocol assumes that the veneers are not etched at the dental laboratory. The advantage of this protocol is that the dentist has complete control over the surface preparation. In the second protocol, the veneers have been etched at the dental laboratory. This requires a certain degree of faith in the lab technician and assumes he/she has properly etched the porcelain. The author recognizes other protocols exist, are valid, and are clinically successful.
A. Protocol #1: Veneers NOT Etched by the Dental Laboratory
- Try in case for fit and esthetics. Once the decision to place the veneers is made, proceed to Step #2.
- Briefly sandblast veneers under low pressure (ie, 20 PSI) with 50 µm Al2O3 or 30 µm aluminum oxide particles.
- Etch with 9% to 10% HF gel for 90 seconds, wash, and dry.
- Ultrasonicate for five minutes in ethanol; dry well.
- Apply one to two coats of pre-hydrolyzed silane and warm-air dry for 60 seconds.
- Place a thin layer of HEMA (hydroxyethylmethacrylate)-free unfilled resin.mm Do not cure. HEMA-free resins are less hydrophilic than HEMA-containing resins and may be less prone to water sorption.
- Fill veneers with a light-cure only resin veneer cementnn,oo and bond in place. Veneer cements that have a dual-cure capacity contain tertiary aromatic amines that may discolor with time, potentially altering the shade of the veneers.71-73
B. Protocol #2: Veneers Etched by the Dental Laboratory
- After receiving the veneers from the dental laboratory, inspect the inner surface. Properly etched veneers should have a dull/matt finish and no shiny areas.
If the veneers have shiny areas, briefly sandblast and re-etch with 9% to 10% HF for 90 seconds. If the veneers appear well etched, proceed to Step #2. - Ultrasonicate for five minutes in ethanol; dry well. Note: an alternative to using an ultrasonic is to clean the veneers with phosphoric acid, wash, and dry. The basic idea is to ensure a clean, fresh, high-energy surface before applying silane, as it may be possible the veneers were contaminated from normal handling and exposure at some point between the laboratory and the time of placement.
- Before the veneers are tried in, apply one to two coats of pre-hydrolyzed silane and warm-air dry for 60 seconds.
- Try in veneers to access fit and esthetics. Note: this author rarely uses try-in pastes. Once approved, place the veneers back in the ultrasonic to remove any surface contaminates. Silane does not need to be re-applied.
- The cleaned veneers should be dried thoroughly and brushed with a thin coat of HEMA-free bonding resin. Note: see Step #6 in Protocol #1.
- Perform the same as Step #7 in Protocol #1.
Conclusion
Optimizing porcelain surfaces prior to treatment with various adhesives and luting resins requires an understanding of the involved substrates and materials, as well as a logical and systematic methodology in their manipulation. It is clear that while much is known, even more remains to be learned. It is hoped that this article has answered some questions, raised others, and provided greater insight into the nature of bonding to porcelain.
Acknowledgment
The author would like to thank Byoung Suh of Bisco Dental for allowing the author unfettered use of his scientific facilities for personal testing of materials, as well as many years of mentorship, encouragement, insight, advice, and friendship.
References
1. 3M ESPE Symposium. July 2007; Wonewok, Minnesota.
2. Matcham J, Dreiser P. The Techniques of Glass Engraving. 1982; London, England: Larousse Art Craft Series, B.T. Bratsford LTD.
3. Wikipedia. Available at: https://www.en.wikipedia.org/wiki/Hydrofluoric_acid. Accessed December 21, 2007.
4. Rich WM. By what mechanism does hydrofluoric acid cause death? Posted May 19, 1998. Accessed December 21, 2007.
5. Muriale L, Lee E, Genovese J, et al. Fatality due to acute fluoride poisoning following dermal contact with hydrofluoric acid in a palynology laboratory. Ann Occup Hyg. 1996;40: 705-710.
6. Sullivan JB, Krieger GR. Hydrofluoric acid. In: Hazardous Materials Toxicology: Clinical Principles of Environmental Health. Baltimore, MD: Williams & Wilkins; 1992.
7. Kukiattrakoon B, Thammasitboon K. The effect of different etching times of acidulated phosphate fluoride gel on the shear bond strength of high-leucite ceramics bonded to composite resin. J Prosthet Dent. 2007;98: 17-23.
8. Canay S, Hersek N, Ertan A. Effect of different acid treatments on a porcelain surface. J Oral Rehabil. 2001;28: 95-101.
9. Boscato N, Della Bona A, Del Bel Cury AA. Influence of ceramic pre-treatments on tensile bond strength and mode of failure of resin bonded to ceramics. Am J Dent. 2007;20: 103-108.
10. Kussano CM, Bonfante G, Batista JG, et al. Evaluation of shear bond strength of composite to porcelain according to surface treatment. Braz Dent J. 2003;14:132-135.
11. Sorensen JA, Engelman MJ, Torres TJ, et al. Shear bond strength of composite resin to porcelain. Int J Prosthodont. 1991;4:17-23.
12. Hussain MA, Bradford EW, Charlton G. Effect of etching on the strength of aluminous porcelain jacket crowns. Br Dent J. 1979;147:89-90.
13. Suliman AH, Swift EJ Jr., Perdiago J. Effects of surface treatment and bonding agents on bond strength of composite resin to porcelain. J Prosthet Dent. 1993;70:118-120.
14. Della Bona A. Characterizing ceramics and the interfacial adhesion to resin: The relationship of surface treatment, bond strength, interfacial toughness and fractography. J Appl Oral Sci. 2005;13(2): 101-109.
15. Chen JH, Matsumura H, Atsuta M. Effect of etchant, etching period, and silane priming on bond strength to porcelain of composite resin. Oper Dent. 1998;23:250-257.
16. Kondo M, Ikeda M, Takeuchi N, et al. [Study on the porcelain veneer restoration. 3. Effect of various treatments of porcelain surface on the bonding strength at porcelain-resin interface]. Aichi Gakuin Daigaku Shigakkai Shi. 1990;28: 631-638.
17. Pisani-Proenca J, Erhardt MC, Valandro LF, et al. Influence of ceramic surface conditioning and resin cements on microtensile bond strength to a glass ceramic. J Prosthet Dent. 2006;96:412-417.
18. Olorunfemi BO. Hydrofluoric acid etching of Flexoceram inlay porcelain: A microscopic study. Saudi Dental J. 1995;7:82-86.
19. Calamia JR, Simonsen RJ. Effects of coupling agents on bond strength of etched porcelain. J Dent Res. 1985;64:296.
20. Della Bona A, Anusavice KJ, Mecholsky JJ Jr. Apparent interfacial fracture toughness or resin/ceramic systems. J Dent Res. 2006;85: 1037-1041.
21. Ozcan M. Evaluation of alternative intra-oral repair techniques for fractured ceramic-fused-to-metal restorations. J Oral Rehabil. 2003;30:194-203.
22. Rosenblum MA, Schulman A. A review of all-ceramic restorations. J Am Dent Assoc. 1997;128:297-307.
23. Westbrook PH. Bonded esthetic onlay. Contemp Esthet Restor Pract. 1999; July/Aug:52-54.
24. Barghi N, Fischer DE, Vatani L. Effects of porcelain leucite content, types of etchants, and etching time on porcelain-composite bond. J Esthet Restor Dent. 2006;18:47-52.
25. Ivoclar Vivadent. https://www.ivoclarvivadent.us/en-us/
26. Tysowsky G. Ivoclar Vivadent. Personal communication. August 28, 2007.
27. Mona H, Abou-Daya M. Effect of ceramic surface treatments on the biaxial flexural strength of heat-pressed glass ceramic. J Egyptian Dent Assoc. 2000;46:1477.
28. Jensen Dental. www.jensendental.com
29. Jensen C. Jensen Industries. Indirect personal communication via West B, Frontier Dental Lab.
30. Chevalier S. Jensen Industries. Personal communication.
31. Kim JS, Hwang HS, Jeong CM, et al. A study on the bond strength of resin cements to Empress 2 ceramic. J Korean Acad Prost. 2001;39:184-196.
32. Magne P, Magne M. Treatment of extended anterior crown fractures using Type IIIA bonded porcelain restorations. J Calif Dent Assoc. 2005;33:387-396.
33. Soares CJ, Soares PV, Pereira JC, et al. Surface treatment protocols in the cementation process of ceramic and laboratory-processed composite restorations: a literature review. J Esthet Restor Dent. 2005;17: 224-235.
34. Bisco and Ultradent Products, Inc. www.bisco.com and www.ultradent.com
35. Suh B. Bisco Dental. Personal communication.
36. Nagayassu PM, Shintome LK, Uemura ES, et al. Effect of surface treatment on the shear bond strength of a resin-based cement to porcelain. Braz Dent J. 2006;17:290-295.
37. Hochhauser L. American Dental Supply, Allentown Pa. Information supplied.
38. Bailey LF, Bennett RJ. DICOR surface treatments for enhanced bonding. J Dent Res. 1988;67:925-931.
39. Addison O, Marquis PM, Fleming GJ. The impact of hydrofluoric acid surface treatments on the performance of a porcelain laminate restorative material. Dent Mater. 2007;23:461-468.
40. Shimada Y, Yamaguchi S, Tagami J. Micro-shear bond strength of dual-cured resin cement to glass ceramics. Dent Mater. 2002;18: 380-388.
41. Roulet JF, Sšderholm KJ, Longmate J. Effects of treatment and storage conditions on ceramic/composite bond strength. J Dent Res. 1995;74:381-387.
42. Suh B. Bisco Dental. Unpublished data from contact angle analysis. 2007.
43. Barghi N. To silanate or not to silanate: making a clinical decision. Compend Contin Educ Dent. 2000;21:659-662, 664.
44. Rosen RR. From treating solution to filler surface and beyond Ð the life history of a silane coupling agent. J Coatings Technology. 1978;50:70-82.
45. Matinlinna JP, Lassila LV, Ozcan M, et al. An introduction to silanes and their clinical applications in dentistry. Int J Prosthodont. 2004;17:155-164.
46. Culler S. ScotchPrime Ceramic Primer Product Profile. 3M Dental Products. 1985. Also personal communication.
47. Anagnostopoulos T, Eliades G, Palaghias G. Composition, reactivity and surface interactions of three dental silane primers. Dent Mater. 1993;9:182-190.
48. Söderholm KJ, Shang SW. Molecular orientation of silane at the surface of colloidal silica. J Dent Res. 1993;72: 1050-1054.
49. Daniels MW, Francis LF. Silane Adsorption Behavior, Microstructure, and Properties of Glycidoxypropyltrimethoxysilane-Modified Colloidal Silica Coatings. J Colloid Interface Sci. 1998;205:191-200.
50. Liu Q, Ding J, Chambers DE, et al. Filler-coupling agent-matrix interactions in silica/polymethylmethacrylate composites. J Biomed Mater Res. 2001;57:384-393.
51. Monticelli F, Toledano M, Osorio R, et al. Effect of temperature on the silane coupling agents when bonding core resin to quartz fiber posts. Dent Mater. 2006;22: 1024-1028.
52. Shih PTK, Koenig JL. Mater Sci Eng. 1975;20:137.
53. Wood WE, Johnson DL, Duncanson MG. Variables affecting silicone-polymethyl methacrylate interfacial bond strengths. J Prosthodont. 1993;2:13-18.
54. Shen C, Oh WS, Williams JR. Effect of post-silanization drying on the bond strength of composite to ceramic. J Prosthet Dent. 2004;5: 453-458.
55. Papacchini F, Monticelli F, Hasa I, et al. Effect of air-drying temperature on the effectiveness of silane primers and coupling blends in the repair of a microhybrid resin composite. J Adhes Dent. 2007;9:391-397.
56. Barghi N, Berry T, Chung K. Effects of timing and heat treatment of silanated porcelain on the bond strength. J Oral Rehabil. 2000;27: 407-412.
57. Kupiec KA, Wuertz KM, Barkmeier WW, et al. Evaluation of porcelain surface treatments and agents for composite-to-porcelain repair. J Prosthet Dent. 1996;76:119-124.
58. Hayakawa T, Horie K, Aida M, et al. The influence of surface conditions and silane agents on the bond of resin to dental porcelain. Dent Mater. 1992;8:238-240.
59. Spohr AM, Sobrinho LC, Consani S, et al. Influence of surface conditions and silane agent on the bond of resin to IPS Empress 2 ceramic. Int J Prosthodont. 2003;16: 277-282.
60. Thurmond JW, Barkmeier WW, Wilwerding TM. Effect of porcelain surface treatments on bond strengths of composite resin bonded to porcelain. J Prosthet Dent. 1994;72: 355-359.
61. Valandro LF, Della Bona A, Antonio Bottino M, et al. The effect of ceramic surface treatment on bonding to densely sintered alumina ceramic. J Prosthet Dent. 2005;93: 253-259.
62. Kern M, Thompson VP. Bonding to glass infiltrated alumina ceramic: adhesive methods and their durability. J Prosthet Dent. 1995;73: 240-249.
63. Ozcan M, Vallittu PK. Effect of surface conditioning methods on the bond strength of luting cements to ceramics. Dent Mater. 2003;19(8):725-731.
64. 3M ESPE. www.3mespe.com
65. Bouschlicher MR, Reinhardt JW, Vargas MA. Surface treatment techniques for resin composite repair. Am J Dent. 1997;10: 279-283.
66. Tezvergil A, Lassila LV, Yli-Urpo A, et al. Repair bond strength of restorative resin composite applied to fiber-reinforced composite substrate. Acta Odontol Scand. 2004;62:51-60.
67. dos Santos JG, Fonseca RG, Adabo GL, et al. Shear bond strength of metal-ceramic repair systems. J Prosthet Dent. 2006;96: 165-173.
68. Frankenberger R, Kramer N, Sindel J. Repair strength of etched vs silica-coated metal-ceramic and all-ceramic restorations. Oper Dent. 2000;25:209-215.
69. Cobb DS, Vargas MA, Fridrich TA, et al. Metal surface treatment: characterization and effect on composite-to-metal bond strength. Oper Dent. 2000;25:427-433.
70. Zhang Y, Lawn BR, Rekow ED, et al. Effect of sandblasting on the long-term performance of dental ceramics. J Biomed Mater Res B Appl Biomater. 2004;71:381-386.
71. Schulze KA, Marshall SJ, Gansky SA, et al. Color stability and hardness in dental composites after accelerated aging. Dent Mater. 2003;19:612-619.
72. Nathanson D, Banasr F. Color stability of resin cements an in vitro study. Pract Proced Aesthet Dent. 2002;14: 449-455.
73. Lu H, Powers JM. Color stability of resin cements after accelerated aging. Am J Dent. 2004;17:354-358.
a IPS Empress, Ivoclar Vivadent, Inc., Amherst, NY
b Filtek Z250 Universal Restorative, 3M ESPE, St. Paul, MN
c Fortress, Chameleon Dental, Kansas City, KS
d Ceramco II, Dentsply, York, PA
e Authentic, Jensen Industries, Inc., North Haven, CT
f IPS e.max CAD, Ivoclar Vivadent, Inc., Amherst, NY
g IPS e.max Press, Ivoclar Vivadent, Inc., Amherst, NY
h IPS Empress II, Ivoclar Vivadent, Inc., Amherst, NY
i Ceramco III, Dentsply, York, PA
j Omega 900, Vident, Brea, CA
k Ex-3, Noritake Co., Inc., Arlington Heights, IL
l IPS d.SIGN, Ivoclar Vivadent ,Inc., Amherst, NY
m Duceram Plus, Dentsply, York, PA
n Luxor
o VMK-68, Vident, Brea, CA
p Creation, Jensen Industries, North Haven, CT
q HF etching gel, Bisco, Inc., Schaumburg, IL
r HF etching gel, Ultradent Products, Inc., South Jordan, UT
s Synspar, Jeneric Pentron, Inc, Wallingford CT
t IPS d.SIGN, Ivoclar Vivadent, Inc., Amherst, NY
u OPC, Jeneric Pentron, Inc, Wallingford, CT
v Noritake, Noritake Dental Supply, Aichi, Japan
w Vintage, Shofu Dental Corp., San Marcos, CA
x Vita, Vident, Brea, CA
y Initial, GC America, Alsip, IL
z Etch it Kit, American Dental Supply, Easton, PA
aa Etch it Kit for Pressable Porcelain, American Dental Supply, Easton, PA
bb Silanit, Ivoclar Vivadent, Inc., Amherst, NY
cc In-Ceram, Vident, Brea, CA
dd Procera All Ceram, Nobel Biocare, Yorba Linda, CA
ee LAVA, 3M ESPE, St. Paul, MN
ff Wol-Ceram, XPdent Corp, Miami, FL
gg Cercon, Degudent, Frankfurt, Germany
hh CERAC InLab, Sirona Dental Systems, Charlotte, NC
ii Everest, KaVo Dental Corp., Lake Zurich, IL
jj CoJet Sand, 3M ESPE, St. Paul, MN
kk RelyX Luting Plus, 3M ESPE, St. Paul, MN
ll Fuji Cem, GC America, Inc., Alsip, IL
mm Porcelain Bonding Resin, Bisco, Inc., Schaumburg, IL
nn RelyX Veneer Cement, 3M ESPE, St. Paul, MN
oo Choice 2, Bisco, Inc., Schaumburg, IL
About the Author
Gary Alex, DMD
Co-Director
Long Island Center for Dental Esthetics and Occlusion
Long Island, New York
Private Practice
Huntington, New York