G•U•M® PerioShield™ Oral Health Rinse
This oral health rinse has received FDA approval as a medical device and is available over the counter.
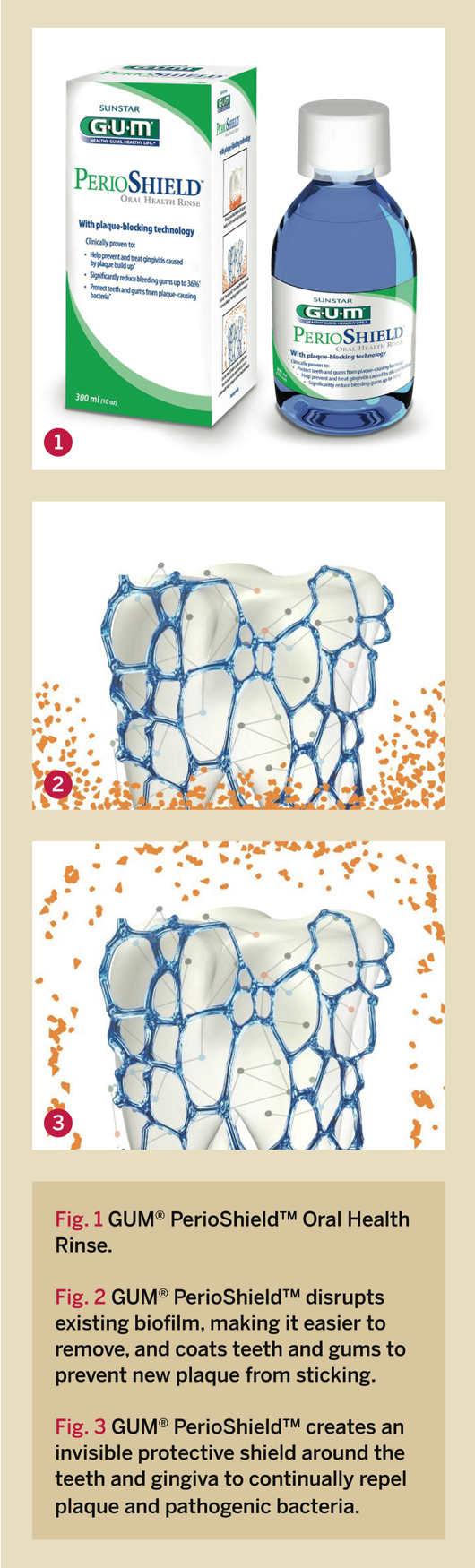
Sunstar Americas, Inc., has recently launched a unique oral health rinse, GUM® PerioShield™ Oral Health Rinse, a new and innovative aid for the prevention and treatment of gingivitis, which can lead to periodontitis (Figure 1). With 29 scientific studies backing the product, this launch further establishes Sunstar’s role in providing clinically proven solutions to dental professionals.
What makes PerioShield unique? It works by disrupting existing biofilm, making it easier to remove, and coating the teeth and gums to prevent new plaque from sticking. It has a proprietary ingredient, delmopinol hydrochloride 0.2%, and a unique mechanism of action that helps prevent the formation of new plaque. Delmopinol (a morpholinoethanol derivative) is an amine alcohol that functions as a surface-active agent shown to interact with pellicle constituents and inhibit glucan synthesis by Streptococcus mutans.2 It has little or no demonstrable effect on the bacteria, but it interferes with plaque/biofilm matrix formation, which makes it easier to remove. The interference with plaque matrix formation inhibits the ability of bacteria to produce polysaccharides, which is the “glue” and nourishment necessary for plaque bacteria to thrive and proliferate (Figure 2 and Figure 3).
With this unique mechanism of action, PerioShield received FDA approval as a medical device and is available over the counter. The rinse creates an invisible protective shield around the teeth and gingiva to continually repel plaque and pathogenic bacteria—thus the name PerioShield.
Because the rinse is not bactericidal, but disrupts the development and maturation of the harmful biofilm while maintaining a good oral flora, it is a very attractive proposition for patients concerned about antimicrobial resistance and maintaining equilibrium of the oral ecosystem.
While PerioShield can be used by any patient group, the ideal candidates are patients with gingivitis who can benefit from stopping the progression of periodontal disease at its earliest stages. It can also be used as a relapse prevention tool for post-chlorhexidine treatment and at-home maintenance therapy after scaling and root planing. PerioShield is also right for any patient who can benefit by maintaining a healthy, balanced oral flora.
References
1. DiMatteo MR, Giordani PJ, Lepper HS, Croghan TW. Patient adherence and medical treatment outcomes: a meta-analysis. Med Care. 2002;40:794-811.
2. Rundegren J, Simonsson T, Petersson L, et al. Effect of delmopinol on the cohesion of glucan-containing plaque formed by Streptococcus mutans in a flow cell system. J Dent Res. 1992:71(11):1792-1796.
For more information, contact:
Sunstar Americas, Inc.
Phone: 800-528-8537
Web: perioshield.gumbrand.com
Disclaimer
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dental Assisting.