Posterior Composites and the New Bulk-Fill Materials
Assessing the research behind this increasingly popular option
In 1990, 94% of dentists in the United States chose amalgam as their primary intracoronal posterior restorative material.1 By 2010, composite resin restorations had exceeded amalgam by a ratio of 2:1.2 In fact, it is estimated that one third of US dentists no longer use amalgam and those that do, report a steady decline.2
This is not an indictment of amalgam, a material that has served dentistry well for more than 100 years. There are many reasons for this relatively rapid and significant change in restorative dentistry. In this author’s opinion, the leading reasons are:
• individual patient desires for non-metal, natural looking restorations
• the less invasive nature of composite restorations3
• the significant improvement in composite resin material physical properties, leading to increased durability and longevity, which, according to recent clinical studies, can rival amalgam.4,5
Nevertheless, many dentists still complain that placing posterior composites is exacting, tedious, time consuming, and not always predictable.
Posterior Composite Predictability
Predictability seems to center on two main issues. The first is postoperative sensitivity. Some dentists believe postoperative sensitivity to chewing is caused by the composite material. However, when properly placed, this is very unlikely with today’s highly filled, low-shrink composite resins. If the patient states that the pain on chewing is sharp and occurs only when hitting a “certain” spot, the problem is almost always the result of an adhesive error (dentin not totally sealed), as opposed to a high restoration that hurts every time the patient chews on it. Thus, an intermittent type of postoperative sensitivity is, for the most part, iatrogenic.6
It should be noted that this problem, once erroneously ascribed to the use of the etch-and-rinse adhesive technique, has declined significantly in recent years.6 The majority of dentists now respect the precise nature of this particular category of adhesive and have learned how to execute the technique properly (ie, do not over-etch or under-prime dentin, dry primers thoroughly after application, and light cure adequately).
Additionally, there has been an increase in the use of the self-etch adhesives, with or without selective enamel etch. The author anticipates that the continued development of advanced universal adhesives will further reduce the incidence of this particular cause of postoperative sensitivity.
Sensitivity to cold following restoration placement is multifactorial and occurs with all kinds of operative procedures and materials. Because dentin is always sealed with an adhesive before placing composite resin, the incidence and duration of this problem should be less with posterior composite restorations compared with unbonded amalgam restorations.
The second major concern in terms of predictability is the lack of achieving a proper contact. Again, this is not a fault of the composite material but entirely a matricing factor. Fortunately, newly designed sectional matrix systems, introduced in the past few years (eg, Composi-Tight® 3D, Garrison Dental Solutions, https://garrisondental.com; Triodent V3 and V4 Systems, Ultradent, www.ultradent.com) as well as newly introduced circumferential and specialty matrices, used with or without a contact former (eg, PerForm™, Garrison Dental Solutions; Contact Pro 2, CEJ Dental, www.cejdental.com), have virtually eliminated this problem.
Faster Placement
Even with the predictability issue resolved, dentists still have to take the time and effort needed for the placement of the composite resin. Current composite resins now yield high physical properties of hardness, flexural strength, and fracture toughness, as well as low shrinkage and low wear. However, these highly filled, highly viscous materials can make it more difficult to achieve intimate adaptation to cavity walls and, because of low depth of cure, require multiple, separately adapted and cured layers. Manufacturers have begun to address this concern by introducing new composite resins and technologies specifically designed for posterior use that allow dentists to place restorations faster and easier. In short, these new bulk-fill products reduce the need for multiple layers when placing posterior composite restorations.
In addition, the flowable base materials (eg, SureFil® SDR® Flow, DENTSPLY Caulk, www.caulk.com; Filtek™ Bulk Fill Flowable Restorative, 3M ESPE, www.3mespe.com) as well as a highly-filled composite that is vibrated into the cavity (ie, SonicFill™, Kerr, www.kerrdental.com), allow excellent adaptation to cavity walls. This, in turn, results in fewer voids and seams when compared to traditional lined and layered composite placement techniques.7,8 These advanced materials have required modifications in resin chemistry (and in the case of SonicFill, the addition of sonic energy technology) to address depth of cure and shrinkage stress. They have also necessitated a re-examination of the science of light curing, polymerization kinetics, and shrinkage stress, especially considering the higher output of today’s curing lights.
Examining the Science
The discussion surrounding bulk-fill materials and whether they meet their manufacturers’ claims rightly leads clinicians to examine the available research. The common understanding is that composite resin should be placed and cured in separate 2-mm increments to reduce polymerization shrinkage stress, and there is a preponderance of literature supporting this approach.8-10 However, in 1996, a paper published in the Journal of Dental Research by Versluis and colleagues first questioned whether the incremental filling technique actually reduced overall polymerization shrinkage stress.11 The authors concluded that this technique actually increased shrinkage stress. Since then, there have been many published papers concluding that placing composite resins in posterior restorations using incremental layers does not appear to be clinically significant to the overall outcome of the restoration.12-17 A more recent article examining cuspal deflection published in the Journal of the American Dental Association also confirmed this premise.18 In addition, this paper further validated the trans-tooth illumination technique to improve depth of cure, which was espoused by Belvedere in 2001.19 However, 2-mm incremental layering is still recommended for non-bulk fill composites due to depth-of-cure limitations.
Regarding depth of cure of composite resins, there are two methods commonly used to measure depth of cure and extent of polymerization. One method, the International Standards Organization (ISO) standard 4049:2009, cures a column of composite from the top surface. The soft, uncured composite is subsequently scraped away from the bottom surface until reaching hard cured material. The depth of cure is then defined by dividing the remaining length of cured composite by two.
In an investigation by Tiba and colleagues into the depth of cure of several bulk-fill materials presented at the International Association of Dental Research (IADR) meeting in 2013, the authors concluded with the following statement: “This study shows some limitations of ISO 4049 for testing the depth of cure in relation to the more important hardness ratio for bulk fill composite materials.”20 In this author’s opinion, use of the ISO standard is limited to comparing relative “curability” of different composite resin materials. Since it does not measure how much composite resin has actually cured (carbon conversion) at any given depth, it is not specifically clinically relevant.20
A second method used by many investigators, known as the Knoop hardness test, defines the depth of cure as the distance from the top of a cured column of composite to a point where the microhardness value is at least 80% of the top surface value. Hardness has been shown to correlate to polymerization (80% bottom-to-top hardness equals 90% carbon conversion). In the author’s opinion, this makes this method for determining depth of cure clinically relevant.21
The same authors of the IADR paper mentioned above carried out an extensive investigation into the physical properties of several bulk-fill materials and compared the data to incrementally placed materials. This research was published by the American Dental Association (ADA) Laboratories.19 Using the hardness ratio method for depth of cure, all the products listed in Table 1 met their manufacturer's claims.19 X-tra base, x-tra fill (VOCO America, www.voco.com), and SonicFill significantly exceeded their respective manufacturer's claim. It should be pointed out that the values these authors obtained were different and lower when the ISO 4049 test method was used. Unfortunately, the opinion these same authors expressed in their IADR research regarding the “more important hardness ratio” did not appear. Although other investigators using the hardness ratio test have shown bulk-fill composites to meet or exceed manufacturer's claims,22,23 at least one paper published using a different protocol for hardness arrived at different results. So clinicians should pay close attention to various study methods.24
Another concern regarding placing large increments of bulk-fill composite resin into a cavity is the potential for creating high shrinkage stress. When the authors of the ADA study examined polymerization stress, the mean values for most of the bulk-fill products tested were not statistically different compared to the layered (non-bulk-fill) controls. The shrinkage stress value for SonicFill was the lowest and was lower by a statistically significant amount compared to the rest of the materials tested.25
Case Examples
The main objective of these new posterior composite resin materials is to reduce the cured layers of composite the dentist has to place, thereby increasing efficiency. The materials rely on high depth of cure (at least 4 mm) along with low shrinkage and controlled stress to allow most preparations to be rapidly filled in fewer increments.
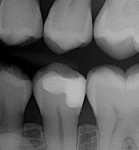
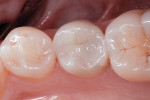
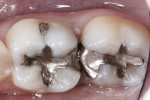
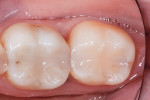
In the experience of the author, the rapid insertion along with the non-slumping, non-sticky sculptability of SonicFill makes placement time and effort rapid and easy, and similar to amalgam. This is something dentists have asked for since composite resins were first used for posterior restorations. In the case shown in Figure 1 through Figure 4, there is demonstrated adaptation to cavity walls without the need for a low-viscosity liner, which means this restoration was both clinically successful and more efficient to place. Figure 5 and Figure 6 show a case in which bulk-fill materials demonstrate both clinical success and longevity.
Final Thought
All of these newer bulk-fill composite resin materials are too new to have long-term clinical trial data; however, they are proving popular with dentists and they continue to grow in the marketplace. Given the number of posterior composites dentists place in practice, this growth would seem unlikely if these new posterior composite materials and technologies were not performing successfully.
Disclosure
Ronald D. Jackson, DDS, FACD, FAGD, FAACD, discloses that he acted as a consultant in the development of SonicFill and retains a financial interest.
References
1. In your dental practice, is dental amalgam still the restorative material of choice? J Am Dent Assoc. 2002;133(8):1046.
2. Jackson RD. Placing posterior composites: increasing efficiency. Dent Today. 2011;30(4):126-131.
3. Lynch CD, Frazier KB, McConnell RJ, et al. Minimally invasive management of dental caries. J Am Dent Assoc. 2011;142(6):612-620.
4. Opdam NJ, Bronkhorst EM, Roeters JM, Loomans BA. A restrospective clinical study on longevity of posterior composite and amalgam restorations. Dent Mater. 2007;23(1):2-8.
5. Opdam NJ, Bronkhorst EM, Loomans BA, Huysmans MC. 12-year survival of composite vs. amalgam restorations. J Dent Res. 2010;89(10):1063-1067.
6. Perdigao J, Geraldeli S, Hodges JS. Total-etch versus self-etch adhesive effect on post-operative sensitivity. J Am Dent Assoc. 2003;134(12):1621-1629.
7. Munoz-Viveros C, Yazici AR, Agarwal I, Campillo-Funollet M. Microleakage in Class II preparations restored with the SonicFill System. Presented at: AADR/CADR Annual Meeting & Exhibition; March 22, 2012; Tampa, FL.
8. Ferracane JL. Resin composite—state of the art. Dent Mater. 2011;27(1):29-38.
9. Meira JB, Bragga RR, Ballester RY, et al. Understanding contradictory data in contraction stress tests. J Dent Res. 2011;9(3):365-370.
10. Park J, Chang J, Ferracane J, Lee IB. How should composite be layered to reduce shrinkage stress incremental or bulk filling. Dent Mater. 2008;24 (11):1501-1505.
11. Versluis A, Douglas WH, Cross M, Sakaguchi RL. Does incremental filling technique reduce polymerization shrinkage stresses? J Dent Res. 1996;75(3): 871-878.
12. Neiva IF, deAndrada MA, Baratieri LN, et al. An in vitro study of the effect of restorative technique on marginal leakage in posterior composites. Oper Dent. 1998;23(6):282-289
13. Gallo JR 3rd, Bates ML, Burgess JO. Microleakage and adaptation of Class II packable resin-based composites using incremental or bulk filling techniques. Am J Dent. 2000;13(4):205-208.
14. Choi KK, Condon JR, Ferracane JL. The effects of adhesive thickness on polymerization contraction stress of composite. J Dent Res. 2000;79(3):812-817.
15. Leevailoj C, Cochran MA, Matis BA, et al. Microleakage of posterior packable resin composites with and without flowable liners. Oper Dent. 2001;26
(3):302-307.
16. Idriss S, Habib C, Abduljabbar T, Omar R. Marginal adaptation of Class II resin composite restorations using incremental and bulk placement techniques: an ESEM study. J Oral Rehabil. 2003;30(10):1000-1007.
17. Rees JS, Jagger DC, Williams DR, et al. A reappraisal of the incremental packing technique for light cured composite resins. J Oral Rehabil. 2004;
31(1):81-84.
18. Campodonico CE, Tantbirojn D, Olin PS, Versluis A. Cuspal deflection and depth of cure in resin-based composite restorations filled by using bulk, incremental and transtooth-illumination techniques. J Am Dent Assoc. 2011;142(10):1176 -1182.
19. Belvedere PC. Contemporary posterior direct composites using state-of-the-art techniques. Dent Clin North Am. 2001;45(1):49-70.
20. Tiba A, Hong A, Zeller G. Examining the depth of cure for bulk fill composite materials [abstract #2435]. Presented at: IADR General Session; March 22, 2013; Seattle, WA.
21. Bouschlicher MR, Rueggeberg FA, Wilson BM. Correlation of bottom-to-top surface microhardness and conversion ratios of various resin composite compositions. Oper Dent. 2004;29(6):698-704.
22. Yapp R, Baumann A, Powers JM. Comparative curing and thermal properties of demi ultra LED curing light. Research report–number 58. The Dental Advisor. 2014. www.dentaladvisor.com/publications/research-reports/comparative-curing-and-thermal-properties-of-demi-ultra-led-curing-light.pdf. Accessed July 10, 2014.
23. Christensen G. Advantages and challenges of bulk-fill resins. Clinician's Report. 2012;5(1):1-2.
24. Flury S, Hayoz S, Peutzfeldt A. Depth of cure of resin composites: Is the ISO 4049 method suitable for bulk fill materials? Dent Mater. 2012;28(5):
521-528.
25. Tiba A, Zeller GG, Estrich CG, Hong A. A Laboratory evaluation of bulk-fill versus traditional multi-increment-fill resin-based composites. ADA Professional Product Review. 2013;8(3):13-17.