Root-End Filling with a New Bioactive Cement
Biodentine can be used in both the root and crown.
Biodentine™ (Septodont USA, www.septodontusa.com) was recently introduced to the dental market. This new bioactive cement has dentin-like mechanical properties and can be used as a root-end filling material, as well as a repair material for root perforations and resorptions. Biodentine can be used in both the root and crown. It can be used for root and pulpal floor perforations, internal and external resorption, apexification, and retrograde root-canal obturation. Biodentine fulfills the requirements for a suitable root-end filling material in that it exhibits biocompatibility, long-term sealing of the cavity, antimicrobial properties, and the ability to induce hard-tissue regeneration; it is also stable, insoluble, non-resorbable, hydrophilic, and easy to prepare and place.
In the case presented in this article, Biodentine was used as a retrofilling material in a 61-year-old patient after apicoectomy on tooth No. 16. The root-end surgery was necessary to remove an apical lesion and a separated root. Seven months after the operation (Figure 1 through Figure 7), tooth No. 16 was clinically normal and tested negative for percussion. The final radiograph showed a complete healing of the apical bone defect without any pathological findings (Figure 8).
Because of its material properties, the author considers Biodentine to be an interesting alternative to conventional root-end filling materials that would benefit from improved radiopacity.
Material Background
A root-end surgery subsequent to a root canal treatment is sometimes the last option considered to conserve a tooth. This apicoectomy results in an exposure of root dentin and root-canal filling material (gutta-percha).1 The placing of a root-end filling after root-end resection is mandatory, because it is well known that the orthograde gutta-percha filling alone—due to its material properties—is insufficient to support bone regeneration. Teeth treated with a root-end filling material (eg, mineral trioxide aggregate [MTA]) after apicoectomy showed significantly better healing than teeth treated by smoothing of the orthograde gutta-percha root filling only.2
The goal of a root-end filling is to seal the root-end cavity and prevent microleakage. Characteristics of a suitable root-end filling material include:
- biocompatibility.
- the ability to seal all margins, preferably by a molecular bonding to the dentinal walls.
- bacteriostatic properties, or not encouraging bacterial growth.
- the ability to induce hard-tissue regeneration.
- stability, insolubility, non-resorbability, and hydrophilicity.
- being easy to prepare and place.
- radiographically visible.1,3,4
An ideal retrofilling material with high-quality apical root canal fillings for endodontic surgeries has yet to be found.3,4 In the past, several different materials such as amalgam, reinforced zinc oxide eugenol cements (interim restorative material [IRM], super ethoxy benzoic acid (EBA]), glass-ionomer cement, and composite resin were used.3-5 Recently, MTA, a refined “Portland cement,”6 was found to have less cytotoxic effects and better results with biocompatibility and microleakage protection, giving it more clinical success over traditional root-end filling materials.5,7 Nevertheless, MTA has some drawbacks: handling is difficult at times, setting time is long, compressive and flexural strengths are much lower than those of dentin, and it is quite costly.
Biodentine, in contrast, offers similar properties to those of MTA, but it consists of powder in a capsule and liquid in a pipette. The powder mainly contains tricalcium silicate, calcium carbonate, and dicalcium silicate, the principal components of MTA. Zirconium oxide serves as the radiopacifier. The liquid consists of calcium chloride in aqueous solution with an admixture of polycarboxylate. The powder is mixed with the liquid in a capsule in a triturator for 30 seconds, and once mixed, its setting time is around 12 minutes. During the setting of the cement, calcium hydroxide is formed. The consistency of Biodentine is similar to phosphate cement. Its crown uses include pulp protection, temporary closure, deep-caries management, cervical filling, direct and indirect pulp capping, and pulpotomy.
The following case illustrates the use of Biodentine for retrograde root-end filling after apicoectomy.
Case Report
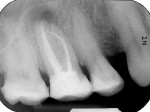
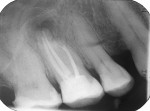
A 61-year-old patient complained of pain and swelling (approximately 1 cm in diameter) in the vestibule of the first molar in the right maxilla. Tooth No. 16 tested negative to sensitivity and positive to percussion. The patient underwent root canal treatment at another location 1 year previously. The radiographs taken (Figure 1) show an apical translucency and a separated root canal instrument that extruded out of the mesio-buccal root. The filling of the four root canals was radiopaque. There were no signs of voids or porosities in the root canal walls. For terminal anesthesia, the patient was injected with 1 mL anesthetic (Septanest, sold in the United States as Septocaine®, Septodont USA, septodontusa.com). For pain therapy, a surgical incision was performed (infection was found). The wound was irrigated and an iodoform stripe was applied. Five days later, the patient came back for wound irrigation and replacement of the iodoform stripe. The patient reported neither pain nor other symptoms.
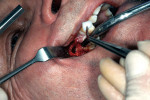
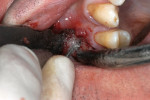
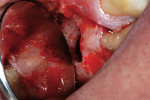
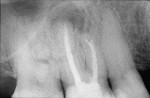
One week later, the patient came in for an apicoectomy. The anesthetic Septanest was again injected for terminal anesthesia, and a trapeze-like gingival flap was prepared in the region of tooth No. 16. The bone was removed to display the mesio-buccal root (Figure 2). An apicoectomy was performed, and granulation tissue was removed and collected for a histological examination. With an ultrasonic device (VDW.ULTRA®, VDW GmbH, www.vdw-dental.com), both mesio-buccal root canals were prepared for the retrograde (Figure 3), enabling the visibility of the separated root canal instrument in the first mesio-buccal root canal (Figure 4). The instrument was removed and the mesio-buccal root canals were cleaned and shaped with an ultrasonic tip approximately 3 mm from the retrograde. Biodentine was chosen as the filling material, and it was applied into the cavity (Figure 5). The gingival flap was replaced and sutured. Postoperative radiographs showed a complete removal of the separated root canal instrument and the bone defect (Figure 6).
The suture was removed after 1 week with optimal results. The patient was pain- and symptom-free. The histological examination confirmed the diagnosis of an apical granuloma with an accumulation of granulocytes and foam cells. There were no signs of a cyst-forming epithelium or malignant cells.
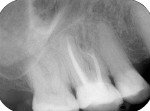
Tooth No. 16 tested negative to sensitivity and percussion at 3 and 7 months. Radiographs showed the beginning of bone regeneration at 3 months (Figure 7) and complete regeneration after 7 months. Figure 8 shows post-apicoectomy and retrograde root-end filling with Biodentine.
Conclusion
In this case, bone regeneration was visible 6 months after the operation. As a treatment alternative, an orthograde retreatment may be possible, but the apical surgery was preferred due to the root canal filling. In addition, it was questionable if the root canal instrument could be removed through an orthograde approach.
Acknowledgment
The author would like to thank Dr. Sebastian Bürklein, Central Interdisciplinary Ambulance in School of Dentistry, Westphalian Wilhelms-University, Münster, Germany, for his kind help with the apicoectomy.
Disclosure
Dr. Dammaschke received honoraria from Septodont USA for writing this article.
References
1. Pitt Ford TR. Surgical treatment of apical periodontitis. In: Ørstavik D, Pitt Ford TR, eds. Essential Endodontology. Oxford, UK: Blackwell; 2003:278-308.
2. Christiansen R, Kirkevang LL, Hørsted-Bindslev P, Wenzel A. Randomized clinical trial of root-end resection followed by root-end filling with mineral trioxide aggregate or smoothing of the orthograde gutta-percha root filling—1-year follow-up. Int Endod J. 2009;42(2):105-114.
3. Carr GB. Surgical endodontics. In: Cohen S, Burns RC (eds.) Pathways of the Pulp. 6th ed. Mosby, St. Louis, Missouri: Mosby; 1994: 531-567.
4. Stropko JJ. Micro-surgical endodontics. In: Castellucci A (ed.) Endodontics. Vol. III. Edizioni Odontoiatriche Il Tridente, Florence; 2009:1118-1125.
5. Bodrumlu E. Biocompatibility of retrograde root filling materials: A review. Aust Endod J. 2008;34(1):30-35.
6. Dammaschke T, Gerth HUV, Züchner H, Schäfer E. Chemical and physical surface and bulk material characterization of white ProRoot MTA and two Portland cements. Dent Mater. 2005;21(8):731-738.
7. Roberts HW, Toth JM, Berzins DW, Charlton DG. Mineral trioxide aggregate material use in endodontic treatment: A review of the literature. Dent Mater. 2008;24(2):149-164.
About the Author
Till Dammaschke, DMD
Assistant Professor
Department of Operative Dentistry
University of Münster
Münster, Germany