Oral Cancer Screening
The emphasis on oral cancer screening aims to increase public awareness and encourage patients to visit a dental professional on a regular basis.
Health history combined with extraoral and intraoral examinations are integral parts of the evaluation of patients who are new to a dental practice as well as the yearly recall dental examination. In addition to caries and periodontal disease, conventional oral examination (COE) detects various other developmental, infectious, immunologically mediated, neoplastic, or reactive conditions. Malignant neoplasia is responsible for the most serious oral disease consequences. Squamous cell carcinoma (SCC) is the most common oral malignancy and accounts for 94% of cases.1 Of course, extraoral and intraoral examinations are not limited to detection of oral SCC, but recently emphasis has been placed on “oral cancer screening.”
The emphasis on oral cancer screening is designed to increase public awareness of the disease and to encourage patients to regularly visit a dental professional. In addition, emphasis on oral cancer screening has also focused healthcare professionals’ awareness of the clinical manifestations of oral cancer. The impetus for increasing awareness of oral cancer screening is the example of successful medical screening programs such as the prostate specific antigen (PSA) test for prostate cancer, PAP smears to detect cervical cancer, and mammography to detect breast cancer. The wide implementation of screening tests has resulted in markedly improved survival statistics for these diseases.2
The overall 5-year survival rate for patients with oral SCC is 60%.3 This dismal statistic has not significantly improved over the last 30 years and results from diagnosis of the majority of oral SCC cases at an advanced stage (59%), rather than at a localized stage (41%).4 Diagnosis of oral SCC in a localized stage results in a 5-year survival rate of 82%, while diagnosis at an advanced stage with local and regional invasion results in a 5-year survival rate of 53%. Advanced oral SCC with distant metastasis at the time of diagnosis results in a 5-year survival rate of only 28%.3 These rates are variable depending on the anatomic location of the tumor, patient gender, and ethnicity.4 Additional factors affecting prognosis are histopathologic findings, such as pattern of invasion5 and HPV-16 status of the tumor tissue.6
Aside from mortality, the stage of oral SCC at diagnosis has a strong impact on patient quality of life after treatment. Early stage (Stage I and II) localized oral SCC is treated with surgical excision and radiotherapy is optional depending on the case. Locoregionally advanced (Stage III, IVA, and IVB) disease with spread to deeper structures and local lymph nodes is treated with surgical resection, dissection of neck lymph nodes, radiotherapy, and chemotherapy. Surgical resection requires grafts or removable prostheses to replace missing tissues. Metastatic (Stage IVC) disease is treated with chemotherapy.7 Advanced-stage oral SCC patients treated with radiation and chemotherapy experience oral mucositis, trismus, xerostomia, increased susceptibility to dental caries, and osteoradionecrosis.8,9 Anatomically, oral cancer is defined as SCC of the lip (skin and vermilion), oral cavity proper, and oropharynx, which are readily accessible for visual inspection and digital palpation. However, the advanced stage at diagnosis of the majority of oral SCC cases does not reflect this accessibility.
Screening detects disease early in asymptomatic people. Thus, strictly speaking, screening is a process that is must be applied to every patient to detect disease. On the other hand, “case finding” is a process that is applied to a patient with signs or symptoms of disease in order to determine what type of disease an abnormality represents. Examples of case-finding methods in oral cancer are toluidine blue staining,10 brush cytology,11 and incisional or excisional biopsy.12
History-taking is an important first step in every patient assessment, including oral cancer screening. Chronic exposure to tobacco, alcohol, and ultraviolet light are the major risk factors for oral SCC; however, about 25% of oral SCC cases arise in people who are considered “low-risk.”13 Oral SCC can, and does, occur in “low-risk” populations, such as children, adolescents, and young adults, presenting an extra challenge to the clinician.14,15 Demographics are relevant; male gender, African-American ethnicity, and increasing age are all factors that increase the risk of oral SCC. The mean age at diagnosis is 63 years and 96% of cases occur in people older than age 40.4 Other risk factors include: betel (areca nut) use, reduced host defenses, and nutritional and genetic factors.16 Certain oral clinical conditions are considered premalignant, eg, actinic cheilosis, smokeless tobacco keratosis, oral submucous fibrosis, proliferative verrucous leukoplakia, and oral erosive lichen planus, although it should be noted that the malignant transformation potential of oral erosive lichen planus is controversial.17 Patients with these conditions, immunosuppression, or a history of epithelial dysplasia or oral SCC should have more frequent screening examinations on a sliding scale of frequency depending on the progression, or lack thereof, of clinical lesions. Generally, it is prudent to examine most patients within 1 month of the initial evaluation, then at 3, 6, and 12 months for the first year after the initial evaluation. If the clinical and radiographic findings remain unchanged, then the period between recall appointments can be lengthened to 6 months, then 12 months.18 A history of previous oral SCC is significant because of evidence that carcinogenic substances affect all of the mucosal lining of the upper aerodigestive tract, not just the area that first became malignant, in a process known as “field cancerization.”19,20 Some evidence to support this theory is the fact that 35% of oral SCC survivors develop at least one second primary tumor during the 5-year follow-up after treatment.21
Recently developed screening devices are being marketed to dental professionals as adjuncts to COE for detection of oral premalignant lesions and SCC. None of these devices has undergone rigorous clinical trials to establish their sensitivity and specificity as screening tools used in a general dental practice.22 Sensitivity is a measure of the proportion of the subjects with the disease that test positive, while specificity determines the proportion without the disease who test negative. In general, a desirable test has both high specificity (few false positives) and high sensitivity (few false negatives).22 FDA approval for the devices was obtained through a 510(k) Exempt FDA Device clearance, which gives a Class I approval through the reasoning that “a non-invasive device may receive FDA approval if it has been shown to be efficacious in another bodily site.”22 The currently available oral cancer screening devices operate by either of two major mechanisms, tissue fluorescence imaging or tissue reflectance (Table 1).
Tissue Fluorescence Imaging
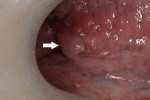
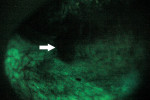
This method was originally developed as a screening tool in lung cancer, where numerous studies have shown its effectiveness. The technology was adapted for the oral cavity, and two devices using this technology have been brought to market, the VELscope® (LED Dental, Inc, https://www.leddental.com) and the Identafi® 3000 ultra (Trimira™ LLC, https://www.trimira.net). These devices rely on production of a particular wavelength (λ) of light, which is shined on the oral mucosa where it excites tissue fluorophores that emit a characteristic fluorescence when viewed through proprietary filters. An abnormality is noted when the tissue does not fluoresce and is noted as a dark area (Figure 1 and Figure 2).
VELscope® is an acronym for “visually enhanced lesion scope.” The device was introduced in 2006 and consists of a portable light-source unit that produces a blue light (λ 400 nm to 460 nm) that is connected by a fiber-optic light-guide cable to a handpiece through which the light is emitted. Disposable plastic barrier sheaths for the handpiece are available for infection control. After stimulation by the 400-nm to 460-nm light, tissue fluorescence is viewed through a replaceable proprietary filter on the handpiece. Normal tissue is visualized as a bright green fluorescence, while abnormal tissue shows a loss of fluorescence, seen as a dark green or a deep-maroon color.
The large area of illumination by the VELscope provides a wide field of view. There also is a radiometer feature that is used each time the unit is turned on to ensure that the machine is outputting the correct wavelength of light. However, because of the fixed angle of the light beam perpendicular to the handle, it is difficult to use the VELscope to visualize the base of the tongue, the palatine tonsils, and tonsillar pillars of the oropharynx. These are sites of oropharyngeal cancer linked to the sexually transmitted, oncogenic types of human papilloma virus (HPV).23 Another limitation is that the VELscope light source unit is substantial in size, somewhat limiting its portability, and it requires an accessible electrical outlet to use.
0vice emits light from the end of a slim handheld, wand-like, battery-operated cordless handpiece fitted with a disposable clear, hard, plastic barrier sheath for infection control. An additional disposable clear, hard, plastic barrier sheath is available with an angled mirror at the end. The operator uses special filtering goggles and turns a knurled knob on the wand to choose one of three different wavelengths of light to use for the examination. The white light is useful as an adjunct to the regular dental chair lamp. The violet-spectrum light (λ 405 nm) is used to excite tissue fluorophores, and the emitted fluorescence of normal tissue is visualized as a blue color, while abnormal tissue shows a loss of fluorescence seen as a dark area. A third option of green-amber illumination is available to enhance the fine visualization of vasculature at and near the tissue surface of areas that are abnormal under violet light.
The Identafi™ 3000 ultra is compact and highly portable and does not require an accessible electrical outlet. The disposable angled mirror is useful to better visualize areas such as the base of the tongue, the palatine tonsils, and tonsillar pillars of the oropharynx. However, the field of illumination of the Identafi™ 3000 ultra is much smaller than the VELscope,thus the time required to visualize the entire oral cavity is longer. There is no feature to ensure that the device is outputting the intended λ 405-nm of violet light. Another limitation is that the batteries will eventually need to be replaced with a freshly recharged set or a set of non-rechargeable AA batteries, if used for a long period of time, such as in a community oral cancer-screening program.
Tissue autofluorescence imaging devices do not require patients to use an acetic-acid rinse, which increases patient acceptability. Manufacturers recommend that room lights should be dimmed to minimize their impact on the visualization of autofluorescence. Thus, these devices are not acceptable for use in an environment where the lighting cannot be controlled. Tissue autofluorescence imaging devices do not do away with the need for clinical judgment. Loss of tissue autofluorescence is not exclusive to premalignant or malignant lesions. The manufacturers recommend blanching tissue that exhibits a loss of fluorescence. If the autofluorescence returns on blanching, a vascular or inflammatory lesion origin is suggested. However, it is awkward, if not impossible, for lesions located in certain anatomic sites, such as the oropharynx, tonsils, or base of the tongue, to be blanched while simultaneously holding the light-emitting handpiece. If an area with a loss of fluorescence is potentially of inflammatory or traumatic etiology, it is recommended that patients return in 2 weeks for a re-examination to see if the loss of fluorescence is maintained.
In the peer-reviewed literature, tissue fluorescence imaging is useful in the hands of oral medicine specialists to determine margins during surgical resection of oral SCC24 and to detect clinically occult areas adjacent to areas of previously treated severe dysplasia or carcinoma-in-situ.25,26 However, these studies were conducted on patients with tissues known to be at high risk of being affected by the field alterations20 that gave rise to the original lesions. Because the Identafi is so recently introduced, there is only a single case report in the literature that describes its utility as a case-finding tool in an oral metastasis from an unknown primary.27 However, tissue fluorescence imaging devices are being marketed to general dentists as oral cancer-screening adjuncts for the vast majority of patients without a history of dysplasia, carcinoma-in-situ, or oral SCC. There is no current evidence that using tissue fluorescence in a general dental practice saves lives, as the marketing claims. It is acknowledged that the use of tissue fluorescence requires training, experience, and a firm knowledge of oral mucosal diseases.28,29
Tissue Reflectance
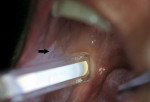
This type of screening tool was originally developed in clinical trials to detect cervical cancer and its preceding dysplastic changes. Abnormal squamous epithelium in the cervical complex appears distinctly white after washing the cervix with a dilute acetic-acid solution and viewed under chemiluminescent light (Speculite®). The technology was adapted for the oral cavity, and three devices using tissue reflectance are currently available on the market. Components common to all of the systems are a rinse agent and a light source. Patients swish with the flavored 1% acetic-acid rinse solution for 1 minute. The rinse is to remove the glycoprotein barrier and slightly desiccate the oral mucosa. Abnormal tissue appears “acetowhite” with illumination by the light source (Figure 3).
ViziLite Plus® TBlue630 (Zila Pharmaceuticals, https://www.vizilite.com) is a system for oral lesion identification and marking that was introduced in 2002. ViziLite is used after the COE is completed and the patient has rinsed with the acetic-acid solution. The light stick is activated by bending the flexible plastic capsule containing acetyl salicylic acid to break a fragile inner glass vial containing hydrogen peroxide, which produces a chemiluminescent blue/white light source (λ 490 nm to 510 nm) that is placed into a disposable plastic holder and used to visually inspect the mucosal surfaces. Normal tissues appear slightly blue, while abnormal squamous epithelium appears “acetowhite” with brighter, sharper, and more distinct margins. TBlue630 Oral Lesion Marking System is a patented, pharmaceutical-grade phenothiazine metachromatic dye packaged in an easy-to-use three-swab system. TBlue630 is used to mark ViziLite-identified lesions. Lesions that uptake and accumulate toluidine blue are thought to have increased amounts of nucleic acids such as DNA and RNA, providing the deep-blue staining that allows ViziLite-identified lesions to be seen clearly under normal light.
One study of the ViziLite system concluded that the overall detection rate as compared to COE was not improved and the chemiluminescent light produced reflections that made lesion visualization more difficult.30 Another study of patients with previous history of oral cancer or premalignant lesions showed that sensitivity for Vizilite and tolonium chloride was 100% and 70.3%, respectively; and specificity was 14.2% for Vizilite and 25% for tolonium chloride, although the majority of the lesions were clinically suspicious by COE.31 Another study that correlated the results of COE ViziLite and TBlue630 with histologic findings showed that chemiluminescence increased the sharpness of lesion margins and 100% of serious lesions retained the TBlue630 stain. The authors acknowledged that the study was performed in both a referral center for mucosal disease and a cancer center, thus the results cannot be generalized to screenings performed in a general dental practice.32
Microlux/DL™ (AdDent, Inc, https://www.addent.com) is a 2-in-1 battery-powered handheld device that uses fiber optic illumination with two different light guides: a diffuse white-light oral lesion screening instrument, and a transilluminator to detect caries or tooth fractures. The device produces a diffuse blue/white light that is used after the COE is completed and the patient has rinsed with the acetic-acid solution. The light is used to visually inspect the mucosal surfaces. Normal tissues appear slightly blue, while abnormal squamous epithelium appears “acetowhite” with brighter, sharper, and more distinct margins. The only study of Microlux/DL™in the peer-reviewed literature showed an enhanced visibility of lesions that were also identified by COE and concluded that it is a poor discriminator for inflammatory, traumatic, and malignant lesions.33
Orascoptic DK (Orascoptic, https://www.orascoptic.com) is a 3-in-1 battery-powered device that employs a battery-powered handheld LED light source with interchangeable diagnostic elements: an oral lesion screening instrument, a transillumination instrument to help visualize crown fractures and proximal caries in teeth, and a lighted mirror to increase visual acuity in hard-to-see places within the oral cavity. There have been no studies reported in the peer-reviewed literature regarding the efficacy of the Orascoptic DK as an adjunct to COE.
Tissue reflectance devices require patients to use an acetic-acid rinse, which some patients may find objectionable. In addition, tissue reflectance is recommended for use in detection of white lesions (leukoplakia) which are much less likely to be histologically premalignant or early invasive SCC than red (erythroplakia) or ulcerated lesions.34,35 A systematic review of the literature concluded that high-quality studies to show efficacy of use of tissue-reflectance devices to detect oral premalignant lesions is sparse and it is unclear what benefit they would provide to the practicing clinician.22
Conclusion
A busy general dentistry practice may see 3,000 patients per year. In the United States, the yearly incidence of oral cancer is 10 cases per 100,000 persons. Statistically, only one case of invasive squamous cell carcinoma will be discovered in an ordinary practice population in 3 years. Some clinicians have recommended the use of adjunctive screening technologies as a practice builder and some device manufacturers are marketing their products in direct-to-consumer advertising.36 The issue of positive financial impact on the dental practice is not inconsequential as there are CDT codes that allow an extra fee for adjunctive screening methods. However, several recent systematic reviews of visually based oral examination adjuncts have concluded that the evidence to support their use is still lacking.22,37-39 This begs the question: is it ethical to charge for a test that has not undergone rigorous studies or been shown to improve the likelihood of oral SCC diagnosis in the general population?
It is important to understand that an adjunctive oral cancer screening device is not a “magic wand” that can be applied to any oral abnormality to give unequivocal information regarding the benign or malignant nature of the tissue, whatever the marketing claims. These techniques should be considered an adjunct to the “gold standard” of oral cancer screening: health history, extraoral and intraoral visual examination with incandescent light, and manual palpation. All of the adjunctive tests are useless if a good history is not taken and the patient does not receive a thorough examination by an educated healthcare professional. Many sources of information on oral cancer screening are available and it is imperative that clinicians keep up to date through continuing education, online resources such as the Oral Cancer Foundation Web site (https://www.oralcancerfoundation.org/dental/screening.htm), and review of the literature.13,40,41 An educational CD, “Oral Cancer Screening” is available through the American College of Prosthodontists at https://www.prosthodontics.org/products/ACPProducts.asp. It is also important to realize that these adjunctive devices cannot diagnose oral cancer.42 A positive finding with an adjunctive screening device requires a definitive diagnosis. The “gold standard” for diagnosis is surgical biopsy and microscopic examination.12
References
1. Kramer IR, Lucas RB, Pindborg JJ, Sobin LH. Definition of leukoplakia and related lesions: an aid to studies on oral precancer. Oral Surg Oral Med Oral Pathol. 1978;46(4):518-539.
2. Cancer Facts and Figures. American Cancer Society. Available at: https://www.cancer.org/docroot/STT/stt_0.asp.
3. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4): 225-249.
4. Rhodus NL. Oral cancer and precancer: improving outcomes. Compend Contin Educ Dent. 2009;30(8):486-498.
5. Brandwein-Gensler M, Teixeira MS, Lewis CM, et al. Oral squamous cell carcinoma: histologic risk assessment, but not margin status, is strongly predictive of local disease-free and overall survival. Am J Surg Pathol. 2005;29(2):167-178.
6. Glazer CA, Chang SS, Ha PK, Califano JA. Applying the molecular biology and epigenetics of head and neck cancer in everyday clinical practice. Oral Oncol. 2009;45(4-5):440-446.
7. Choong N, Vokes E. Expanding role of the medical oncologist in the management of head and neck cancer. CA Cancer J Clin. 2008;58(1):32-53.
8. Vikram B. Adjuvant therapy in head and neck cancer. CA Cancer J Clin. 1998;48(4):199-209.
9. Whitmyer CC, Esposito SJ, Terezhalmy GT. Radiotherapy for head and neck neoplasms. Gen Dent. 1997;45(4):363-370.
10. Martin IC, Kerawala CJ, Reed M. The application of toluidine blue as a diagnostic adjunct in the detection of epithelial dysplasia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(4):444-446.
11. Sciubba JJ. Improving detection of precancerous and cancerous oral lesions. Computer-assisted analysis of the oral brush biopsy. U.S. Collaborative OralCDx Study Group. J Am Dent Assoc. 1999;130(10):1445-1457.
12. Melrose RJ, Handlers JP, Kerpel S, et al. The use of biopsy in dental practice. The position of the American Academy of Oral and Maxillofacial Pathology. Gen Dent. 2007;55(5):457-461.
13. Weinberg MA, Estefan DJ. Assessing oral malignancies. Am Fam Physician. 2002;65(7):1379-1384.
14. Jepsen SA, Closmann JJ. The insidious nature and presentation of oral squamous cell carcinoma in the low-risk population. Gen Dent. 2008;56(1):78-82.
15. Woo VL, Kelsch RD, Su L, et al. Gingival squamous cell carcinoma in adolescence. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107(1):92-99.
16. Scully C, Bagan J. Oral squamous cell carcinoma: overview of current understanding of aetiopathogenesis and clinical implications. Oral Dis. 2009;15(6):388-399.
17. Eisen D. The clinical features, malignant potential, and systemic associations of oral lichen planus: a study of 723 patients. J Am Acad Dermatol 2002 Feb;46(2):207-14.
18. Alexander RE, Wright JM, Thiebaud S. Evaluating, documenting and following up oral pathological conditions. A suggested protocol. J Am Dent Assoc. 2001;132(3):329-335.
19. Slaughter DP, Southwick HW, Smejkal W. Field cancerization in oral stratified squamous epithelium; clinical implications of multicentric origin. Cancer. 1953;6(5):963-968.
20. Thomson PJ. Field change and oral cancer: new evidence for widespread carcinogenesis? Int J Oral Maxillofac Surg. 2002;31(3):262-266.
21. Lingen MW. Cause-related marketing: the uneasy alliance between altruism and corporate greed. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(4):407-409.
22. Lingen MW, Kalmar JR, Karrison T, Speight PM. Critical evaluation of diagnostic aids for the detection of oral cancer. Oral Oncol. 2008;44(1):10-22.
23. Herrero R, Castellsague X, Pawlita M, et al. Human Papillomavirus and Oral Cancer: The International Agency for Research on Cancer Multicenter Study. Journal of the National Cancer Institute. 2003;95(23):1772-1783.
24. Poh CF, Zhang L, Anderson DW, et al. Fluorescence visualization detection of field alterations in tumor margins of oral cancer patients. Clin Cancer Res. 2006;12(22):6716-6722.
25. Lane PM, Gilhuly T, Whitehead P, et al. Simple device for the direct visualization of oral-cavity tissue fluorescence. J Biomed Opt. 2006; 11(2):024006.
26. Poh CF, Ng SP, Williams PM, et al. Direct fluorescence visualization of clinically occult high-risk oral premalignant disease using a simple hand-held device. Head Neck. 2007;29(1):71-76.
27. Vigneswaran N, Koh S, Gillenwater A. Incidental detection of an occult oral malignancy with autofluorescence imaging: a case report. Head Neck Oncol. 2009;1(1):37.
28. Balevi B. Evidence-based decision-making: should the general dentist adopt the use of the velscope for routine screening for oral cancer? J Can Dent Assoc. 2007;73(7):603-606.
29. Laronde DM, Poh CF, Williams PM, et al. A magic wand for the community dental office? Observations from the British Columbia Oral Cancer Prevention Program. J Can Dent Assoc. 2007;73(7):607-609.
30. Oh ES, Laskin DM. Efficacy of the ViziLite system in the identification of oral lesions. J Oral Maxillofac Surg. 2007;65(3):424-426.
31. Ram S, Siar CH. Chemiluminescence as a diagnostic aid in the detection of oral cancer and potentially malignant epithelial lesions. Int J Oral Maxillofac Surg. 2005;34(5):521-527.
32. Epstein JB, Silverman S Jr, Epstein JD, et al. Analysis of oral lesion biopsies identified and evaluated by visual examination, chemiluminescence and toluidine blue. Oral Oncol. 2008;44(6):538-544.
33. McIntosh L, McCullough MJ, Farah CS. The assessment of diffused light illumination and acetic acid rinse (Microlux/DL) in the visualisation of oral mucosal lesions. Oral Oncol. 2009;45(12):227-231.
34. Melrose RJ. Premalignant oral mucosal diseases. J Calif Dent Assoc. 2001;29(8):593-600.
35. van der Waal I, Axell T. Oral leukoplakia: a proposal for uniform reporting. Oral Oncol. 2002;38(6):521-526.
36. Lingen MW. Direct-to-consumer advertising for oral cancer screening devices. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107(3):299-300.
37. Thomson PJ, Hamadah O. Cancerisation within the oral cavity: the use of ‘field mapping biopsies’ in clinical management. Oral Oncol. 2007;43(1):20-26.
38. Patton LL, Epstein JB, Kerr AR. Adjunctive techniques for oral cancer examination and lesion diagnosis: a systematic review of the literature. J Am Dent Assoc. 2008;139(7):896-905.
39. Fedele S. Diagnostic aids in the screening of oral cancer. Head Neck Oncol. 2009;1(1):5.
40. Horowitz AM, Alfano MC. Performing a death-defying act. J Am Dent Assoc. 2001;132 (Suppl):5S-6S.
41. Lynch DP. Oral cancer risk and detection: The importance of screening technology. Dental Economics. 2007;97(9):1-9.
42. Scully C, Bagan JV, Hopper C, Epstein JB. Oral cancer: current and future diagnostic techniques. Am J Dent. 2008;21(4):199-209.
About the Author
Lynn W. Solomon, DDS, MS
Associate Professor
Department of Oral Pathology
Tufts University School of Dental Medicine
Boston, Massachusetts