Remineralization Strategies
Dental caries’ slow progression offers dental professionals an opportunity for early intervention.
Dental caries is the most prevalent chronic disease in the United States. Early recognition and intervention of the disease process (before cavitation), can stop and even reverse the disease (remineralization of the non-cavitated lesion). Saliva’s natural remineralization process cannot keep pace with altered biological and behavioral factors. Fluoride has been proven to prevent dental carious lesions; but when the caries challenge is severe, fluoride alone may not be sufficient. Various technologies have been developed to supplement calcium and phosphate, maintain homeostasis, and restore the caries balance. This article highlights some available techniques and materials to enhance remineralization and reduce demineralization.
Caries
The slow progression of dental caries through enamel of up to 73 months, coupled with earlier detection methods and recent treatment advances, gives the dental profession the opportunity to remineralize early carious lesions.1,2 Dental caries is a complex, multifactorial, infectious, and chronic disease process with both biological and/or behavioral risk factors that is initiated when acid dissolves mineral from teeth (demineralization). Despite the dramatic decline in the prevalence of dental caries in the United States over the past few decades, dental caries still affects 91% of the adult population and is the primary cause of tooth loss.3 This high level of disease may be attributed to the interaction of several factors such as lack of knowledge/education, dietary habits, certain medical conditions, medication intake, access to dental care, genetics, tooth morphology, oral hygiene habits, exposure to community water fluoridation, and previous caries experience.
Carious lesions occur when there is an imbalance of protective and pathological factors, thus creating a disturbance in the cyclical demineralization and remineralization process of coronal and root surfaces. The formation of plaque biofilm enables the proliferation of cariogenic bacteria which produces acid as a by-product of fermentable carbohydrates and consequently reduces salivary pH, dissolving the mineralized structure of the teeth.2,3-13 The lower the pH, the more pathogenic the flora becomes. Early lesions appear as a result of the loss of calcium, phosphate, and carbonate, forming a partially demineralized subsurface lesion, which is seen as “white spots” especially in areas of plaque accumulation (Figure 1). The early stages of this process can be reversed by decreasing pathological factors such as plaque biofilm and diet and increasing some of the protective factors discussed below.
Saliva
Saliva has the principal role in maintaining oral homeostasis. Normal saliva maintains the physiologic pH and the integrity of the dentition through its ability to replace or restore tooth’s minerals, and prevent demineralization.14 Saliva clears debris, lubricates, and provides antimicrobial protection. Any qualitative and quantitative loss of saliva (due to radiation to head and neck, Sjögren’s syndrome, or medication-induced salivary hypofunction, etc) results in an increase in cariogenic bacterial population (ie, Streptococcus mutans and Lactobacillus) at the expense of non-cariogenic bacteria present in normal saliva, consequently upsetting the caries balance.15,16
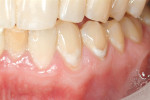
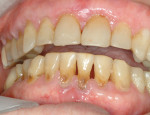
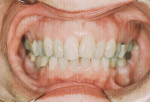
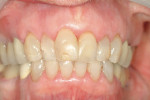
The frequent consumption of fermentable carbohydrates and the presence of orthodontic brackets, partial dentures, crowns, bridges, or multiple restorations also contribute to plaque development and retention. When acidic challenges persist and demineralization continues, the intact enamel surface will eventually collapse and result in a cavity (Figure 2, Figure 3, Figure 4).4 However, carious lesions can be remineralized chemically before surface cavitation occurs and more advanced lesions can be remineralized to allow retention of restorations.3-5
Fluoride
The main mechanism of action of fluoride is remineralization of teeth after their eruption.3 It is well documented that fluoride concentrated in plaque and saliva simultaneously inhibits demineralization and catalyzes remineralization, which promotes the formation of fluoroapatite. Fluoroapatite is more resistant to acid attack than the original hydroxyapatite. Fluoride can be bacteriostatic or bacteriocidal, depending on the concentration. 17 The normal remineralization process utilizes the re-uptake of calcium and phosphate ions from saliva in the presence of fluoride.
Community water fluoridation and fluoride-containing dentifrices (250 ppm to 1,500 ppm) have proven to be the most effective vehicles for the delivery of fluoride and are responsible for the major decline in carious lesions worldwide.3 Daily use of fluoride oral rinses (230 ppm to 920 ppm) has been demonstrated to be a clinically effective adjunct to brushing with fluoride-containing dentifrices. A multitude of over-the-counter and professionally applied fluoride products are also available. The most common forms of fluoride used in commercial dentifrices are stannous fluoride, sodium fluoride, and sodium monofluorophosphate.18 Professional or prescription-strength fluoride treatments include pastes, gels, and foams (up to 5,000 ppm), rinses (233 ppm to 920 ppm), and varnishes (22,600 ppm). Treatment with prescription-strength fluoride combined with professional fluoride treatment (preferably fluoride varnish) at 3- to 6-month intervals has become the standard of care for those at high risk for developing caries.12,16 Though fluoride dentifrices have been very successful in reducing the incidence of coronal caries worldwide, root caries and erosion are an increasing problem13
Calcium and Phosphate
When there are elevated cariogenic challenges such as salivary hypofunction, fluoride alone is not enough to prevent dental caries. Saliva is supersaturated with calcium and phosphate ions, creating a concentration gradient that facilitates the diffusion of ions into the teeth to maintain homeostasis. Concomitant therapy with calcium, phosphate, and prescription-strength fluoride is strongly recommended to overcome the challenge of salivary hypofunction.19-21
Treatments
For many decades, attempts to formulate the delivery of calcium, phosphate, and fluoride remained a biocompatibility and bioavailability challenge.20 Current technological advancements have overcome these challenges and many remineralization systems are now commercially available or in development. The following treatments may have the potential to be effective for the management of early carious lesions by promoting remineralization and preventing demineralization.
Age Defying® (Arm and Hammer, https://www.armandhammer.com) was previously called Enamelon®, and is a remineralizing toothpaste that contains amorphous calcium phosphate/ACP along with 1,100 ppm of fluoride.21 A 1-year randomized clinical trial of caries in head-and-neck cancer patients compared the efficacy of this dual-phase remineralizing toothpaste to that of a fluoride-only toothpaste and showed an increase in reversals of early carious lesions and a significant reduction in root surface lesions.21
Caphosol® (EUSA Pharma, https://www.eusapharma.com) is a supersaturated remineralizing rinse of soluble calcium and phosphate ions. In a xerostomic group, a regimen of Caphosol used daily with 1.1% sodium fluoride dentifrice, and fluoride varnish treatments every 3 months was effective in preventing the progression of both root and coronal caries and significantly increased net reversals or remineralization.22 Figure 4 and Figure 5 show the successful caries management of a woman 22 years post-radiation due to a submandibular gland tumor. Her regimen included daily use of Caphosol, 1.1% sodium fluoride trays, and then toothpaste once it became available, as well as fluoride varnish applications every 3 months.
Recaldent® (Bonlac Foods Limited, https://www.fonterra.com), MI Paste® (GC America, https://www.gcamerica.com), and Trident White® Chewing Gums (Cadbury Adams USA, https://www.cadburyadams.com) are all casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) products that increase the levels of calcium and phosphate ions to remineralize enamel subsurface lesions. Evidence supports the clinical use of the CPP-ACP as an adjunct to fluoride treatment for the management of early carious lesions.23,24 In a randomized, controlled clinical trial of sugar-free gum containing CPP-ACP, it was shown to promote the regression of early interproximal caries radiographically.25 CPP-ACP and fluoride products work best if used at different times of the day.26 Adding ACP as filler in sealants and composites may help with remineralization of both enamel and dentin.22-24
NovaMin® (Sultan Healthcare, https://www.sultanhealthcare.com) is made with calcium sodium phosphosilicate, a nanosized, bioactive glass which interacts with oral fluids, releasing sodium, calcium, and phosphate, creating a pH gradient to deposit hydroxycarbonate apatite (HCA) to the surface of both enamel and dentin. Originally used for the treatment of bony defects, in vivo and vitro experiments have shown that NovaMin enhances remineralization and prevents demineralization of enamel and dentinal lesions, as well as lesions due to erosion.27-31 Novamin re-hardened root lesions that prescription strength fluoride alone could not.27 A prospective randomized clinical trial is currently in progress to establish the evidence for this product.
A recent study comparing the effects of resin-based sealants, fluoride-containing sealants, fluoride varnish, and a glass-ionomer cement on stabilizing or reversing incipient caries concluded that glass-ionomer was the most effective in reducing carious areas and the most efficient at inhibiting new caries lesions and demineralization of intact enamel adjacent to sites where it was placed.8 Using glass ionomers as a restoration, sealant, or cavity liner is a good option for high-risk patients because of their fluoride-releasing property. When exposed to the fluoride contained in saliva, dentifrices, rinses, etc, glass ionomers, compomers, and resin-modified glass ionomers are recharged with fluoride which is released over time, enhancing remineralization. Glass ionomers are reinforced with resins to overcome wear under the forces of attrition. Nanocomposites with good stress-bearing and caries-inhibiting capabilities are under development.32
Chlorhexidine, an antimicrobial agent, has not been found to be effective for caries reduction as a rinse but, when used as a 10% coating it has shown a 25% reduction in caries in an adult high-risk population.33,34 A current multicenter clinical trial is now in progress to determine its effectiveness for the prevention of adult caries.
Conclusion
Guidelines and definitions of risk categories for dental caries are available from a number of resources, including the ADA Council on Scientific Affairs.26,35,36 It is important to assess the level of caries risk of a patient to choose the appropriate products as an adjunct to fluoride treatments to assist in remineralization. Further randomized clinical trials are needed to strengthen the level of evidence for these remineralizing agents.
Disclosure
Dr. Papas has served as a consultant to EUSA Pharma.
References
1. Berkey CS, Douglass CW, Valachovic RW, Chauncey H. HTI: Longitudinal radiographic analysis of carious lesion progression. Comm Dent Oral Epidemiol. 1988:1683-1690.
2. Featherstone JD. Remineralization, the natural caries repair process—the need for new approaches. Adv Dent Res. 2010;21:1-2.
3. Centers for Disease Control and Prevention. Surveillance for dental caries, dental sealants, tooth retention, edentulism, and enamel fluorosis—United States, 1988-1994 and 1999-2002. In: Surveillance Summaries, August 26, 2005. MMWR. 2005;54(No. SS-3):5-7.
4. Featherstone JD. Dental caries: a dynamic disease process. Aust Dent J. 2008;53(3):286-291.
5. Choo-Smith LP, Dong CCS, Cleghorn B, et al. Shedding new light on early cavities detection. J Can Dent Assoc. 2008;74(10):913-918.
6. Jenson L, Budenz AW, Featherstone JD, et al. Clinical protocols for caries management by risk assessment. J Calif Dent Assoc. 2007;35(10):714-723.
7. Fontana M, Zero DT. Assessing patients’ caries risk. J Am Dent Assoc. 2006;137(9):1231-1239.
8. Ferreira JMS, Silva MFA, Oliveira AFB, et al. Evaluation of different methods for monitoring incipient carious lesions in smooth surfaces under fluoride varnish therapy. Int J Paediatr Dent. 2008;18:300-305.
9. Trairatvorakul C, Kladkaew S, Songsiripradabboon S. Active management of incipient caries and choice of materials. J Dent Res. 2008;87(3):228-232.
10. Horowitz AM. A report on the NIH consensus development conference on diagnosis and management of dental caries throughout life. J Dent Res. 2004;83:C15-C17.
11. Longbottom C, Ek strand K, Zero D. Traditional preventive treatment options. Monogr Oral Sci. 2010;21:149-155.
12. American Dental Association Council on Scientific Affairs. Professionally applied topical fluoride: evidence-based clinical recommendations. J Am Dent Assoc. 2006;137(8):1151-1159.
13. Winn DM, Brunelle JA, Selwitz RH, et al. Coronal and root caries in the dentition of adults in the United States, 1988-1991. J Dent Res. 1996;75:642-651.
14. Atkinson JC, Grisius M, Massey W. Salivary hypofunction and xerostomia: diagnosis and treatment. Dent Clin North Am. 2005;49: 309-326.
15. Keene HJ, Fleming TJ. Prevalence of caries-associated microflora after radiotherapy in patients with cancer of the head and neck. Oral Surg Oral Med Oral Pathol. 1987;64:421-426.
16. Zero DT, Fontana M, Martínez-Mier EA, et al.The biology, prevention, diagnosis and treatment of dental caries. J Am Dent Assoc. 2010;140:(Suppl 1):25S-34S.
17. Camosci DA, Tinanoff N. Anti-bacterial determinants of stannous fluoride. J Dent Res. 1984;63:1121-1125.
18. Zero DT. Dentifrices, mouthwashes, and remineralization/caries arrestment strategies. BMC Oral Health. 2006;6(Suppl 1):S9.
19. Biesbrock AR, Faller RV, Bartizek RD, et al. Reversal of incipient and radiographic caries through the use of sodium and stannous fluoride dentifrices in a clinical trial. J Clin Dent. 1998; 9:5-10.
20. Hara AT, Karlinsey RL, Zero DT. Dentine remineralisation by simulated saliva formulations with different Ca and P in contents. Caries Res. 2008;42:51-56.
21. Papas A, Russell D, Singh M, et al. Caries clinical trial of a remineralizing toothpaste in radiation patients. Gerodontology. 2008;25:76-88.
22. Singh ML, Papas AS. Long term clinical observation of dental caries in salivary hypofunction patients using supersaturated calcium phosphate remineralizing rinse. J Clin Dent. 2010;20:87-92.
23. Reynolds EC. Casien phosphopeptide-amorphous calcium phosphate: the scientific evidence. Adv Dent Res. 2010;21:25-29.
24. Cochrane NJ, Saranathan S, Cai F, et al. Enamel subsurface lesion remineralisation with casein phosphopeptide stabilised solutions of calcium, phosphate and fluoride. Caries Res. 2008;42(2):88-97.
25. Morgan MY, Adams GG, Bailey DL, et al. The anticariogenic effect of sugar-free gum containing CPP-ACP nanocomplexes on approximal caries determined using digital bitewing radiography. Caries Res. 2008:42:171-184.
26. Young DA, Featherstone J, Roth JR, et al. Consensus statement. Caries management by risk assessment: implementation guidelines. CDA Journal. 2007;35(11):799-805.
27. Burwell AK, Likowski LJ, Greenspan DC. Calcium sodium phosphosilicate (Novamin®) remineralization potential. Adv Dent Res. 2010;21:35-39.
28. Karlinsey RC, MacKay AC, Stookey GK, Pfarrer AM. In vitro assessments of experimental NaF dentifrices containing a prospective calcium phosphate technology. Am J Dent. 2010;22:180-184.
29. Trairatvorakul C, Kladkaew S, Songsiripradabboon S. Active management of incipient caries and choice of materials. J Dent Res. 2008;87(3):228-232.
30. Pfarrer AM, Karlinsey. Challenges of implementing new remineralization technologies. Adv Dent Res. 2010;21:79-82.
31. Wefel JS. NovaMin® : Likely clinical success. Adv Dent Res. 2010;21:40-43.
32. Xu H, Moreau JL, Laurence LS, Sun C. Strength and fluoride release characteristics of a calcium fluoride based dental nanocomposite. Biomaterials. 2008;29: 4261-4267.
33. Wyatt CC, Maupome G, Hujoel PP, et al. Chlorhexidine and preservation of sound tooth structure in older adults. A placebo-controlled trial. Caries Res. 2007;41:93-101.
34. Banting DW, Papas A, Clark DC, et al. The effectiveness of 10% chlorhexidine varnish treatment on dental caries incidence in adults with dry mouth. Gerodontology. 2000;17:67-76.
35. Horowitz AM. A report on the NIH consensus development conference on diagnosis and management of dental caries throughout life. J Dent Res. 2004;83:C15-C17.
36. American Dental Association Council on Scientific Affairs. Professionally applied topical fluoride: evidence-based clinical recommendations. J Am Dent Assoc. 2006;137(8):1151-1159.
About the Authors
Athena S. Papas, DMD, PhD
Tufts University
Boston, Massachusetts
Mabi L. Singh, DMD, MS
Tufts University
Boston, Massachusetts