Chairside CAD/CAM
Lithium disilicate restoration for anterior teeth made simple.
Gregg A. Helvey, DDS
In 1904, Dr. Hart J. Goslee from Chicago, Illinois, presented his paper entitled, “The Mechanical Requirements and Cosmetic Possibilities of Modern Artificial Crown Work” at the Fourth International Dental Congress in St. Louis, Missouri.1 Dr. Goslee said, “Since it is the very highest achievement of true art to copy nature, we have contributed liberally thereto by the development of procedures which enable us in our particular field to so closely simulate nature in her perfections and imperfections as to be able to substitute the natural with the artificial in such a manner as to preclude detection.” He concluded his paper with, “the special advantages to be obtained from the application of porcelain crowns lie in the artistic manner and facility with which the natural conditions and varying characteristics may be closely simulated, together with the increased possibilities for securing a degree of strength exceeding any other style of construction.”
Imagine what Dr Goslee’s reaction would have been if someone at that meeting had told him that in 105 years a small portable machine would take a picture of an anterior tooth and then mill the exact shape and contour from a ceramic block. Then the clinician would stain it and cement it. The all-ceramic restoration would be stronger than any porcelain crown he could fabricate, the whole process taking under 2 hours. Dr. Goslee probably would have smiled at the idea because he directed at the end of his paper to, “attempt to stimulate others to aid in the elevation of the profession in general, and of dental art and dental mechanics in particular, to a sphere beyond that of mere artisan, and more in accord with the higher degree of art which modern possibilities offer.” He went on to say such possibilities, however, are not the product of the day, but are an outgrowth of a gradual development to achieve results beyond the ordinary.2
Esthetic dentistry has come full circle since Dr. Goslee presented his paper in 1904. One of the other points he read in his paper was that metal was used indiscriminately in the fabrication of anterior crowns. The all-porcelain “jacket” crown was first introduced by Charles H. Land and patented in 1889.2 The restoration was improved by Dr. E. B. Spaulding of Detroit, and later was extensively used and publicized by Dr. W. A. Capon of Philadelphia.2 In 1909, Dr. R. H. Riethmueller described a method of making an all-porcelain jacket crown using platinum foil in Dental Cosmos.2The porcelain “jacket” crown was not known for its strength but was nevertheless used quite extensively up to the 1950s.
The technique of bonding feldspathic porcelain to a metal framework was invented in the late1950s by Dr. Abraham Weinstein.3 Using the “lost wax” casting technique, gold was used to create a metal coping onto which porcelain was fused. The fit was much better than the porcelain jacket crown, which employed a platinum foil as a matrix during fabrication. The marginal integrity of the porcelain jacket crown was always questionable because after completion of the crown the foil was removed, thus creating a marginal gap.
After Buonocore’s discovery of enamel etching in 1955,4 all-ceramic restorations had a re-emergence into the marketplace. The most popular all-ceramic restoration was IPS Empress® by Ivoclar Vivadent (www.ivoclarvivadent.us). Enamel and dentin bonding helped to increase the popularity of this restoration.
Then computer-aided design/computer-aided manufacturing (CAD/CAM) entered dentistry with CEREC® (Sirona Dental Systems, www.sirona.com) in the early 1990s. As the computer software technology grew, so did the milling hardware capability. That has brought dentistry to digital impressions (iTero™, Cadent, www.cadentinc.com; and Lava™ C.O.S., 3M ESPE, www.3mespe.com) and single-visit chairside restorations using CEREC® AC (Sirona) and E4D Dentist™ (D4D Technologies, www.d4dtech.com). Several manufacturers have contributed to the development of the restorative materials used by the single-visit CAD/CAM systems. 3M ESPE manufactures the Paradigm™ MZ100 composite block, Vident (www.vident.com) provides the Vitablocs® Mark II ceramic block, and Ivoclar Vivadent offers the IPS Empress ceramic and their newest lithium disilicate ceramic additions, the CAD LT and CAD HT blocks. The use of the CAD LT ceramic block will be illustrated in the following single-visit case study.
Lithium Disilicate
To increase the strength, thermal expansion, and contraction behavior of ceramics, manufacturers have added crystalline filler particles.5 Other types of filler additions include particles of high-melting glasses that are stable at the firing temperature of the ceramic.6 Kelly5 refers to a ceramic as a “glass-ceramic” when the filler particles are added mechanically during manufacturing precipitate within the starting glass by special nucleation and growth heating treatments. Two examples of glass ceramics that contain high concentrations of lithium disilicate are IPS e.max® Press and IPS e.max® CAD (Ivoclar Vivadent). The press form is used in the laboratory, whereas the CAD form is used in conjunction with CAD/CAM technology for chairside and laboratory settings. Both materials can be pressed or milled to full contour, then stained and glazed or cut back and layered with the appropriate layering ceramics.
Lithium disilicate was first introduced in 1998 as IPS Empress® 2 (Ivoclar Vivadent).7 It was developed in response to a high demand for all-ceramic fixed partial dentures (FPDs).8 It was used mainly as a single-core or FPD framework to which a veneering ceramic was applied.
Today, the lithium disilicate CAD blocks come partially crystallized. In this state, the lithium metasilicate crystals provide sufficient strength for milling and try-in purposes. The manufacturing process uses a glass technology that provides a material void of any defects and an even distribution of color pigments. The grain sizes of lithium metasilicate crystals range from 0.2 µm to 1 µm, rendering a flexural strength of 130 MPa to this material. This is comparable to the other mill-ready leucite-reinforced CAD/CAM (ProCAD, Ivoclar Vivadent) blocks and the feldspathic CAD/CAM blocks (Vitabloc Mark II).9
The standard 27-minute crystallization process reaches a temperature of 840ºC to 850ºC (1,544°F to 1,562°F) in a two-stage ceramic furnace. A shorter firing cycle (under 20 minutes) can be used with this type of oven, providing no more than two restorations at a time and the spray-type glaze is used rather than the paste version. During the firing cycle, the restoration shrinks by a factor of 0.2%. The computer software takes this shrinkage into account during the milling process.
During the crystallization cycle there is a controlled growth of the grain size (0.5 µm to 5 µm). This transformation leads to a glass ceramic that is made up of prismatic lithium disilicate dispersed in a glassy matrix.10 This alteration increases the flexural strength of the restoration to 360 MPa,11 an increase of 170%. A random orientation of small interlocking plate-like crystals makes up the lithium-disilicate restoration. The orientation and size of the crystals can account for crack deflection and blunting which, in turn, accounts for the increase in fracture toughness over leucite-reinforced cermics.12
There are two basic fabrication methods. The first method is to mill the restoration to full anatomical contour. Before crystallization, the incisal edge is preserved by creating a silicone index.13 The incisal edge is cut back, creating mamelons and layered with the appropriate incisal porcelains back to the original contour using the silicone index as the guide. The restoration is then crystallized in the furnace using the standard firing program. A variation of this technique is crystallizing before the layering steps. This method allows the operator to see the color of the restoration before application of the layering ceramics. This does require a wash coat firing of the layering ceramic before the build-up ceramic is applied.
The second method is to mill the crown to full contour, then stain, glaze, and crystallize. This method also has a variation that includes applying the stain and glaze after the crystallization step. This allows the operator to see the final color of the crown while applying the stains. It may be easier to apply the stains, but it involves a second 12-minute firing cycle.
Case Study
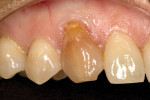
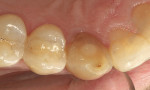
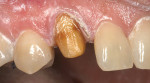
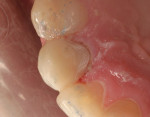
A 43-year-old woman presented with a darkened right maxillary canine that had been endodontically treated 10 years earlier (Figure 1 ). The patient experienced blunt trauma as a result of a fall and the tooth became non-vital some time after the incident. The color of the tooth darkened progressively. The tooth had now become her chief complaint. Radiographic examination was negative. Clinical examination revealed a large lingual composite that was placed where endodontic access had been made (Figure 2 ) and wear on the incisal edge was present. Various treatment options were discussed, which led to using a full-coverage all-ceramic crown fabricated using CEREC technology.
The patient requested to duplicate the same shape but change the color to match the adjacent teeth. The first step was to select the correct shade. Shade selection should always be made before any powdering or tooth preparation. The manufacturer supplies a shade guide but the shade tabs are plastic and not made of lithium disilicate. Selecting the shade with a different material than that of the final restoration can result in the wrong selection.
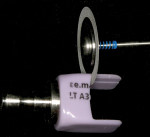
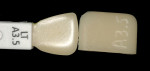
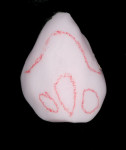
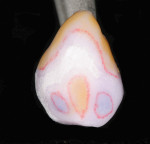
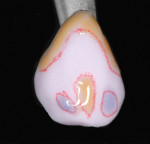
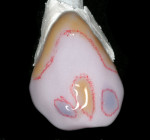
A customized shade guide can easily be fabricated. There are, at times, side portions of the CAD block remaining after the restoration is milled. These unmilled portions can be separated from the remainder of the block with a diamond disk (Figure 3) and inscribed using a small diamond bur with the appropriate shade name (A2, A3 etc) (Figure 4 ) and then crystallized during a firing cycle. This will create an exact shade tab of the actual lithium disilicate block material, which helps in eliminating inconsistencies in shade selection (Figure 5 ).
The correlation mode was selected in the CEREC computer program, which makes a copy of the existing tooth. To capture the tooth, a reflective surface must be created using titanium dioxide that is evenly powdered over the entire tooth and the adjacent teeth. Five preoperative images were taken to preserve a copy of the unprepared tooth.
The tooth was then prepared using a 6847KR 018 modified shoulder diamond bur (Brasseler USA, www.brasseler.com). The minimum amount of reduction is 1 mm at the cervical margin, 1.2 mm of axial reduction in the facial and lingual mid-body area, and 1.5 mm of incisal reduction. In cases with a dark stump shades, increasing the facial reduction to 2 mm will increase the ceramic thickness and provide a greater masking effect of the underlying shade of the prepared tooth. The modified shoulder will create a flat 1-mm depth at the cervical margin with a slightly rounded internal line angle. Sharp line angles are areas of higher stress concentrations and should always be avoided with all-ceramic restorations. Rounded internal line angles minimize stress concentrations.14
The last step in the preparation process was to use a 10839 014 end-cutting diamond bur (Brasseler) to remove any reverse or troughed edges on the margin, which are contraindicated in all-ceramic restorations.15 Creating a 90° exit angle will facilitate the scanning process and prevent thinning of the ceramic margins. Flat, defined margins also will enhance the accuracy of the margin tracing step during that phase of the computer program. A diode laser was used on the mesial portion to expose the margin (Figure 6).
The prepared tooth was once again powdered with titanium dioxide. Several images were taken, which included the adjacent teeth. A common anatomical landmark in both the preoperative and postoperative images is necessary for the software to merge or “stitch” the images together creating a virtual model (Figure 7 ).
Once the design process was completed, an IPS e.max CAD LT block in shade A-2 was selected. Recently, the manufacturer has introduced a new version of the CAD material with a higher level of translucency referred to as the CAD HT block.
The appropriate shade block (LT version) was inserted into the milling machine. Selecting the correct milling program is important in achieving the proper physical properties of the material. There are ways of “fooling” the software into thinking that a different ceramic material is in the milling machine, which will allow the operator to use a “fast-milling” mode. The difference in milling modes may save 10 minutes of mill time but the adverse effect on the lithium disilicate may be beyond healing during the crystallization cycle (written personal communication with manufacturer’s ceramic engineer). The manufacturer strongly advises not to use this accelerated milling program. Not only are there adverse effects on the material to consider, there is a decrease in longevity of the diamond milling burs.
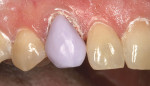
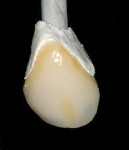
After the milling process was completed, the small remaining milling sprue was removed. The crown was steam-cleaned to remove the milling slurry film. This film also can be removed using an ultrasonic bath. The crown was then tried in to confirm the fit and the bite. Next, the crown was removed and the marginal areas were finished and polished using a pink and grey Dialite diamond-impregnated wheel (Brasseler). The crown was then re-inserted for staining (Figure 8).
Staining Technique
Chairside staining is an art form. For the beginner, it can be very difficult to determine the correct stain, how much to use, where to place it, and how to keep the various stains from “bleeding” into each other. The first step is to observe the matching tooth, whether it is contralateral or adjacent. The color in teeth has to be looked for, and even we, as clinicians, overlook what makes up a natural tooth. As well-respected dentist/ceramist Dr. Robert Stein once said to the author at a lecture in 1979, “Your eyes see the color but your brain does not perceive it.” It takes a conscientious effort to observe the color in teeth. Nakagawa’s study found that anterior teeth have 4 different modes of color. The most frequent occurrence is when there are different colors in the cervical, mid-body, and incisal. Having no color change is second, followed by color change in the cervical area only. Least frequent is a color change only in the mid-body of the tooth.16 Because the tri-colored mode has the greatest frequency, the clinician should initially look for this mode.
To find the correct stain, a thinned out amount of stain can be applied to the matching tooth to see if there is a blending. This process can be repeated until the stain and the color of the matching tooth are the same. The chroma or saturation of the stain can also be determined with this exercise. The next step is to determine exactly where to place the stain and keep it there. One method is to use a red wax margin liner pencil that is used for marking the margins on the dies of working models. Using the wax pencil, areas can be drawn on the surface of the crown where the different stains are to be applied (Figure 9). These areas should have the polished surface removed with a fine slow-speed diamond or green stone. The wax keeps the stains in place and allows the operator to place numerous stains in a small area. During the crystallization firing, the wax pencil lines burn off and leave no traces. This method can be referred to as “staining within the lines” similar to our early grammar school exercise of “coloring within the lines.”
In this case it was determined that the contralateral tooth had an increased chroma in the cervical area where the Khaki stain matched. The mid-body contour ridge also had an increase in chroma where the Khaki stain matched. It was also determined that the mesial and distal depressions had more translucency and this was duplicated using Incisal 1 stain. After the stains were applied, the crown was carefully removed from the patient and secured from the lingual aspect of the crown with college pliers (Figure 10 ).
Lithium-Disilicate Crystallization
The next step is the crystallization process, where the crown is placed on a silver nitride firing tray by means of an IPS e.max CAD crystallization pin. There are several different sizes of pins. The largest pin that does not contact the internal walls of the crown is selected. The crown is first filled with IPS Object Fix Putty or Flow material. This material fixes the crown to the crystallization pin and protects the margins from rounding off during the 840°C firing cycle. Any excess putty is then smoothed with a porcelain or plastic spatula and removed from the sides of the crown with a wet brush (Figure 11). While holding the crown, the IPS e.max CAD Crystall./Glaze Spray is applied to the entire crown. Holding the spray can about 4 inches away and short bursts, spray glaze is applied to the crown, allowing each coat to dry before applying the next (Figure 12). A paste type glaze is also available.
It is important that no other type of firing tray, such as a honeycomb tray, be used during this crystallization process. The rate of cooling after the crystallization process is critical in preserving the physical properties of the lithium disilicate. The silver nitride tray and pin material hold the heat longer, so the rate of cooling is slower. It is critical that this step not be hurried (Figure 13).
After the crystallization step was completed and the crown cooled to the touch, the crystallization pin was removed. The putty material was removed using a steam cleaner. An alternative method of removing the putty material can be accomplished with an ultrasonic bath.
Cementing Materials
What is unique about the IPS e.max CAD lithium disilicate ceramic is having the ability to insert a restoration using either adhesive, self-etching adhesive or conventional cementing methods. The question then should be: Which cementing medium produces the best result, or are they equal in terms of retention, fracture resistance, and sealing ability? There are a number of studies that find that resin-based cementing materials show similar results compared to glass-ionomer cement. Al-Wahadni et al,17 in their study, found no significant difference in fracture resistance when premolar lithium-disilicate crowns were cemented with glass-ionomer and resin cement. A study was conducted on anterior and posterior 3-unit lithium-disilicate fixed partial dentures (FPDs) cemented with glass-ionomer and resin cement. Wolfart et al18 found that the 8-year survival rate according to Kaplan-Meier was 93%. They concluded that short-span, crown-retained, three-unit FDPs made from lithium-disilicate glass-ceramic can be used clinically irrespective of an adhesive or conventional cementation.
Zhang et al19 reported that the fracture mode of monolithic dental ceramics is thickness-dependent and fracture resistance was strongly dependent on the support material. The less stiff support material resulted in flexure of the cement side of the ceramic, increasing tensile stresses that produced radial fractures. Radial fractures can be initiated by sandblasting or grinding from a dental bur. They concluded that high elastic-modulus cement reduced the risk of fractures. Fleming et al20 studied the effect of resin cement increasing the performance of all-ceramic crowns. They found the combination of ceramic surface pre-treatment and cement moved the fracture origin from the ceramic/cement interface to the cement surface. Two previous studies that had been proposed by Marquis21 (crack healing) and Nathanson22 (resin-polymerization shrinkage-strengthened ceramic) were based on the presence of defects on the ceramic surface. Fleming found ceramic strengthening was independent of the defect population. Dong and Darvell23 found that generation of critical tensile stress was dependent on the elastic modulus mismatch between the ceramic, the cement, and the supporting material. The greater bond strength between the ceramic and the supporting material prevented delamination and allowed for more stress transfer between the ceramic and the tooth. Increasing the bond strength (more stress transfer) was more advantageous than increasing the flexure strength of the ceramic. By increasing the cement elastic modulus, the ceramic-cement-dentin complex became stiffer, therefore, decreasing the stress in the ceramic. Lee et al24 found greater resistance to fracture of bonded ceramics when comparing the stiffness of the adhesive cement. Rekow et al25 reported that the thickness and type of ceramic were the determinants of ceramic strength, and those factors having some influence included cement elastic modulus, the position and angle of the applied load, and the supporting tooth core.
A monolithic IPS e.max CAD lithium-disilicate crown, with a flexural strength of 360 MPa, which is seconded only by the pressed form, is the strongest ceramic available. If the tooth preparation provides the required ceramic thickness, then, according to Rekow et al,25 using adhesive cement with a high elastic modulus will contribute to the over-all strength of the restoration. The use of an adhesive cement with a higher elastic modulus would correlate with the findings of Zhang et al,19 Dong and Darvell,23 and Lee et al.24
Ceramic Surface Preparation and Insertion
Conditioning or treatment of the bonding surface of all-ceramic crowns is necessary to create a strong ceramic-to-resin bond. Peumans et al26 reported a significant difference in microtensile bond strength between ceramic specimens treated with hydrofluoric acid, silane, and resin versus specimens that either had no treatment or were treated with phosphoric acid only. In their study, Öztürk et al27 tested the bond strength of two different luting cement systems to lithium-disilicate and leucite-reinforced ceramic inlays. One system used hydrofluoric-acid conditioning for 60 seconds then neutralized (Ceramic Etchant Neutralizer, DENTSPLY Ceramco, www.ceramco.com) followed with the application of silane (Monobond-S, Ivoclar Vivadent) for 60 seconds. The specimens were then cemented with Variolink II with its respective bonding agent. The other cement system used phosphoric acid for 5 seconds to condition the cementing surface, then was rinsed and air-dried followed by the application of Clearfil™ Ceramic Primer (Kuraray Dental) for 60 seconds. The specimens were then cemented using Panavia F ED and its respective bonding agent. Microtensile bond strength testing showed no differences between the two systems. In fact, Krishnan et al28 found lower bond strengths of hydrofluoric-acid-etched IPS Empress ceramic than untreated ceramic surface when using Clearfil Ceramic Primer.
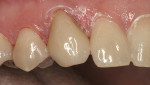
In this case, the Clearfil Esthetic Cement with DC Bond (Kuraray) system was selected for its high elastic modulus. The internal bonding surface was conditioned with phosphoric acid for 5 seconds (manufacturer’s recommendation), thoroughly rinsed with water, air-dried, and followed with an application of Clearfil Ceramic Primer. The primer was immediately air-dried. The two-bottle DC Bond adhesive material was mixed and applied to the tooth for 20 seconds followed by thorough drying with the air-water syringe. The clear-shade esthetic cement was dispensed with the automix tip and placed in the crown. The crown was then seated followed by light polymerization for 20 seconds on each side. The excess was then removed and the occlusion was verified (Figure 14 and Figure 15 ).
Conclusion
In the 105 years since Dr. Goslee presented his paper, the art and mechanics of “artificial crown work” have made tremendous strides. The use of an all-ceramic material milled from a computer program and inserted at the same appointment would have most likely impressed him. It is hard to imagine the technology that will be used to restore that same canine 105 years from now.
Acknowledgment
Special thanks to Ruth Egli, RDH, for her editorial contribution.
References
1. Kirk EC, Litch WF, Endelman E. Transactions of the Fourth International Dental Congress. St. Louis, Mo; August 29–September 3, 1904. Dental Cosmos. 1905:174-180.
2. Taylor JA. History of Dentistry: A Practical Treatise for the Use of Dental Students and Practitioners. Lea and Febiger, New York; 1922.
3. Asgar K. Casting metals in dentistry: Past, present, future. Adv Dent Res. 1988;2:33-43.
4. Touati B, Miara P, Nathanson D. Esthetic Dentistry and Ceramic Restorations. Martin Dunitz Ltd, London; 1999:10.
5. Kelly JR. Dental ceramics: What is this stuff anyway? J Am Dent Assoc. 2008:139:4S-7S.
6. Denry IL. Recent advances in ceramics for dentistry. Crit Rev Oral Bio Med. 1996;7(2):134-143.
7. Giordano R. Materials for chairside CAD/CAM-produced restorations. J Am Dent Assoc. 2006;137(1 Suppl):14S-21S.
8. Powers JM, Sakaguchi RL. Craig’s Restorative Dental Materials. 12th ed. Mosby Elsevier, St. Louis, Mo; 2006:469.
9. Seghi RR, Sorensen JA. Relative flexural strength of six new ceramic materials. Int J Prosthodont. 1995;8:239-246.
10. van Noort R. Introduction to Dental Materials. Elsevier Health Sciences, Philadelphia, Pa; 2002:244.
11. Helvey GA. Retro-fitting an existing crown adjacent to a removable partial denture in a single visit. Inside Dentistry . 2009; 5(3):34-41.
12. Mansour YF, Al-Omiri MK, Khader YS, Al-Wahadni AM. Clinical performace of IPS-Empress 2 ceramic crowns inserted by general dental practitioners. J Comp Dent Pract. 2008;9(1):1-11.
13. Taskonak B, Sertgöz A. Two-year clinical evaluation of lithia-disilicate-based all-ceramic crowns and fixed partial dentures. Dent Mater. 2005;22:1008-1013.
14. McLaren EA, White SN. Glass-infilrated airconia/alumina-based ceramic for crowns and fixed partial dentures: Clinical and laboratory guidelines. Quintessence. 2000:63-76.
15. Gürel G. The Science and Art of Porcelain Laminate Veneers. Quintessence Publishing, Chicago; 2003:267-268.
16. Nakagawa Y. Analysis of natural tooth color. ShiKai Tenbo. 1975;46:527.
17. Wahadni AM, Hussey DL, Grey N, Hatamieh MM. Fracture resistance of aluminum oxide and lithium disilicate-based crowns using different luting cements: An in vitro study. J Contemp Dent Pract. 2009;10(2):51-58.
18. Wolfart S, Eschbach S, Scherrer S, Kern M. Clinical outcome of three-unit lithium-disilicate glass-ceramic fixed dental protheses: Up to 8 years results. Dent Mater. 2009 (in press).
19. Zhang Y, Kim JW, Bhowmick S, et al. Competition of fracture mechanisms in monolithic dental ceramics: Flat model systems. J Biomed Mater Res. Part B: Appl Biomater 88B 2009:402-411.
20. Fleming GJP, Maquire FR, Bhamra G, et al. The strengthening mechanism of resin cements on porcelain surfaces. J Dent Res. 2006;85(3):272-276.
21. Marquis PM. The influence of cementson the mechanical performance of dental ceramics. Bioceramics. 1992;5:317-324.
22. Nathanson D. Principles of porcelain use as an inlay/onlay material. In: Garber DA, Goldstein RE, eds. Porcelain and Composite Inlays and Onlays: Esthetic Posterior Restorations. 1993; Chicago, Ill; Quintessence. 23-32.
23. Dong XD, Darvel BW. Stress distribution and failure mode of dental ceramic structures under Hertzian indebtation. Dent Mater. 2003;19:542-551.
24. Lee JJ-W, Wang Y, Lloyd IK, Lawn BR. Joining veneers to ceramic cores and dentition with adhesive interlayers. J Dent Res. 2007;86(8):745-748.
25. Rekow ED, Harsono M, Janal M, et al. Factorial analysis of variables influencing stress in all-ceramic crowns. Dent Mater. 2006;22(2):125-132.
26. Peumans M, Hikita K, DeMunch J, et al. Effects of ceramic surface treatments on bond strength of an adhesive luting agent to CAD/CAM ceramic. J Dent. 2007;35(4):282-288.
27. Öztürk AN, Inan Ö, Inan E, Öztürk B. Microtensile bond strength of CAD/CAM and pressed-ceramic inlays to dentin. Euro J Dent. 2007;1:91-96.
28.Krishnan G, Yapp R, Powers J. Bond strength of Clearfil Esthetic cement and Clearfil Ceramic Primer to treated and untreated ceramics. Dent Advisor. 2008(18):3-4.
About the Author
Gregg A. Helvey, DDS
Adjunct Associate Professor
Virginia Commonwealth University School of Dentistry
Richmond, Virginia
Private Practice
Middleburg, Virginia