The New STA Injection
Dynamic Pressure Sensing allows the clinician to know when the correct site has been found.
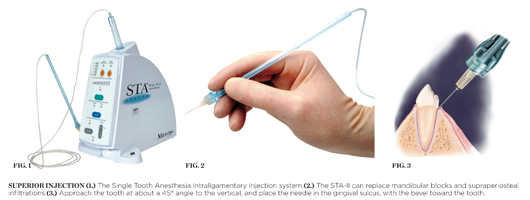
There are major differences that should be considered between the traditional periodontal ligament (PDL) injection delivered with a dental syringe and the Single Tooth Anesthesia Intraligamentary Injection (STA-II, Figure 1), such as:
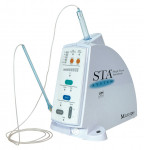
- The PDL injection is usually the last resort, when the mandibular block fails. The STA-II (Figure 2) should be the primary injection for any maxillary or mandibular tooth and can replace mandibular blocks and supraperiosteal infiltrations, which cause collateral numbness to the patient’s lip, face, and tongue.
- With the PDL injection, a small amount of anesthetic is injected under excessive pressure, which produces a short duration of anesthesia. The STA-II delivers a larger volume of anesthetic under minimal pressure resulting in longer duration (40 minutes).
- The PDL injection is difficult to administer and the flow rate depends on manual pressure. The STA-II is easy to administer and the flow rate is computer-controlled, consistent, and below the patient’s pain threshold.
- The PDL injection is painful on delivery, results in tissue damage and bone resorption, and can cause postoperative discomfort. The STA-II is a comfortable injection and a clinical study shows it causes no tissue damage or bone resorption and little or no postoperative discomfort.
- The STA, using Dynamic Pressure Sensing, allows the clinician to know when the correct site (the PDL space) has been arrived at for a successful intraligamentary injection; it also indicates if the site has been left and if the needle has been blocked by obstruction or pressure.
STA-II Technique Tips
Use a 30-gauge half-inch needle. For maxillary teeth, use the distal-buccal and mesial-buccal line angles. For mandibular teeth, use the distal-lingual and mesial-lingual line angles. It is very important to always start on the distal and then proceed to the mesial.
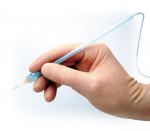
If anesthetizing a mandibular tooth, shorten the Wand STA Handpiece by pulling the micro-tubing out of the handpiece channel, turning the tubing away, and snapping off the plastic handpiece to a length of about 1.5 inches. The needle can also be slightly bent toward the bevel if necessary for better access. Approach the tooth at about a 45° angle to the vertical, and place the needle in the gingival sulcus, with the bevel toward the tooth (Figure 3). Initiate the Cruise Control by engaging the foot pedal until a voice prompts Cruise, and then release the foot pedal. A Control-Flo delivery of anesthetic continues throughout the entire injection. Slowly move the needle down the root of the tooth until there is resistance. Hold the needle in place without excessive pressure and wait for the sound and light prompts to signal the correct injection site. This may take up to 10 to 15 seconds. When the light prompt arrives at the middle of the Yellow Zone, there is a good chance that the needle is in the correct site; if the light prompt arrives at the Green Zone, there is an excellent chance that the needle is in the correct injection site. Hold the needle in place without excessive pressure, and deliver at least one half of a cartridge of lidocaine 2% with epinephrine 1:100,000 per site. Look for circumferential gingival blanching. Epinephrine is necessary for duration of anesthesia of approximately 45 minutes, but a concentration stronger than 1:100,000 should not be used. If using 4% articaine or prilocaine, add epinephrine 1:200,000 and best judgment with respect to a reduction in the volume of anesthetic delivered. As a general rule, about half of the amount of lidocaine 2% (a quarter of a cartridge per site) is recommended.
About the Author
Eugene R. Casagrande, DDS, FACD, FICD
Director of International & Professional Relations
Milestone Scientific, Inc.
For more information, contact:
Milestone Scientific, Inc.
Phone: 8008621125
Web: www.STAis4U.com
Disclaimer
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry. The preceding is not a warranty, endorsement, or approval for the aforementioned products or services or their effectiveness, quality, or safety on the part of Inside Dentistry or AEGIS Communications. The publisher disclaims responsibility for any injury to persons or property resulting from any ideas or products referred to in the preceding material.