Gingival Augmentation
“Periodontal plastic surgery” is gaining attention among dental patients, and it is incumbent for all dentists to be familiar with functional, structural, and cosmetic indications.
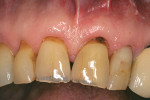
Many dental patients are interested in preserving and optimizing their overall physical health. They read about the interactions between oral health and diabetes, heart disease, and other systemic conditions. In addition to the desire for teeth which are free of caries (Figure 1), esthetically pleasing, and oriented in a proper arch form, “periodontal plastic surgery” is more widely discussed in treatment planning. This article will document a number of structural, maintenance, and cosmetic indications for enhancing facial periodontal support around teeth.
A Historical Look at “Keratinized Tissue”
Since the early 1970s, clinical periodontists and researchers have looked at the relationship between keratinized gingival tissue and the health of the tissue surrounding teeth and dental implants. Lang and Loe correlated the amount of keratinized tissue to gingival inflammation and, therefore, periodontal health.1 Their conclusion was that patients required a 2-mm zone of keratinized tissue with 1 mm of that tissue firmly attached to the dentoalveolar complex to maintain optimal oral health. This may be important in patients undergoing orthodontic therapy. A recent study determined that there was significantly more gingival recession after active orthodontic therapy.2 In this study, 20% of sites exhibited bleeding on probing. There was also more recession and higher plaque indices where there were bonded retainers left on the lingual surfaces of teeth. One out of every five patients in this study smoked. Combining the results of these studies should lead to more comprehensive periodontal evaluation of patients before undergoing orthodontic therapy. There have been debates in the literature based on both human and animal studies about whether or not keratinized tissue is necessary, and if so, how much. One study conducted in 1982 compared sites with recession that were treated with gingival grafting and contralateral sites that were maintained through a non-surgical approach.3 Over the 4-year time period that the patients were observed, the control areas did not lose any further attachment if the patients maintained optimal plaque control. The grafted sites, however, exhibited creeping reattachment over the previously denuded root surfaces.
Implant-supported restorations require keratinized tissue for the proper emergence profile of the restoration from the bone level. In addition to esthetically pleasing prostheses, keratinized tissue may be essential to the long-term maintenance of bone and gingival levels. In one study, Bouri and coworkers determined that the presence of a wider zone of keratinized tissue is beneficial to both the soft tissue and bone.4 Where there was less than 2 mm of keratinized tissue, there was a three times greater likelihood of finding bleeding on probing. Additionally, there was more mean crestal alveolar bone loss around the implants with minimal zones of keratinized tissue compared to implants that were restored in an area of adequate keratinized tissue height.
Periodontists may be called upon to restore facial keratinized tissue around dental implant-supported restorations. One recent study demonstrated that after initial high rates of success, the percentage of “recession” that remained covered 6 months after soft tissue grafting was only 66%.5 The authors concluded that coronal flap repositioning alone was insufficient to predictably correct gingival recession around dental implants.
Cosmetic Enhancement
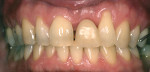
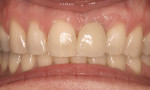
The primary rationale for root-coverage procedures being lumped together is to bring the facial gingival margin back to the location of the cementoenamel junction. Patients preparing for cosmetic enhancement of the coronal tooth structure do not wish to appear “long in the tooth.” Terms such as “the golden proportion” have been discussed by many authors in relation between length and ideal width of teeth in the esthetic zone (Figure 2). There have been a number of articles written over the years describing the relationship between the width and length of the coronal portions of a tooth across the “normal” spectrum of tooth sizes. According to the authors of these articles, average anterior teeth have standard relationships between their height and width. When the size of teeth appear in this range, they appear “normal.” 6 Studies have been done for more than 30 years correlating the average height and width of teeth,7 demonstrating repeated ratios describing length to width viewed from the anterior and from direct facial directions. These guidelines assist restorative dentists when fabricating full- or partial-coverage restorations, orthodontists when repositioning teeth around the arch, and periodontists when performing crown lengthening8 or gingival augmentation procedures.9
Factors Limiting Success
In attempting root coverage, one critical factor for successful augmentation is the location of the proximal bone and papilla height.10,11 This can limit the blood supply to the mobilized flap and barrier or graft tissue used. Patients who smoke have exhibited less predictable root coverage than non-smokers.12 This study showed clinical and histologic evidence of the effects of smoking on periodontal soft tissue regenerative surgery. The smokers had more gingival recession and significantly less root coverage after surgery than non-smokers. One cause of this and other possible “failures” from smoking could be a decrease in vascularity in the site. This study documented that smokers had more than 25% fewer blood vessels per field viewed under the microscope than non-smokers. Another study of gingival augmentation procedures led to the same conclusion that smokers had more gingival recession, lower clinical attachment levels, and more postoperative pocketing 2 years after the procedure than non-smokers.13
Materials Used for Augmentation
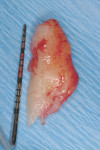
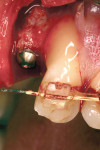
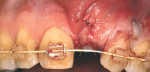
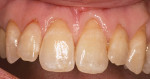
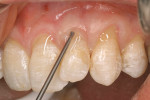
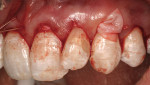
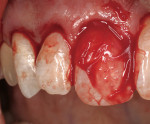
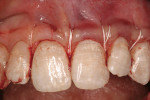
The use of the free gingival graft has been in the literature for more than 40 years. Its uses have incorporated pre-prosthetic surgery,14 frenectomy removal, vestibular deepening, and root coverage.15 Decreased trauma to the donor site, better color match, and more predictable healing results were seen with the subepithelial connective tissue graft16 (Figure 3). Over the past nearly 30 years, numerous studies have shown the effectiveness, predictability, and color match after root-coverage procedures performed with palatal connective tissue17 (Figure 4, Figure 5, Figure 6, Figure 7).
In areas where the palatal or tuberosity tissue is too thin to harvest autogenous tissue,18 allograft tissue can be used. Materials like pericardium and acellular dermal matrix have been incorporated into periodontal plastic surgical procedures19,20 (Figure 8 and Figure 9). Histology has demonstrated proof that both autogenous and allograft tissue heal and result in the formation of keratinized tissue in the recipient site. Improving the predictability of obtaining a successful graft by placing the materials between periosteum or mucosal tissue and the exposed root and bone or periosteum is a key factor in the success of these procedures.21 This technique, whether performed as a tunnel procedure or after full-flap elevation, enables the graft to be stabilized physically, preventing micromovement and eliminating dead space between the graft and recipient site (Figure 10). Additionally, there is a quicker perfusion of the graft/barrier complex when it is inserted between two blood supplies (Figure 11). Coronal positioning with PTFE sutures maintains the soft tissue flap in place over the graft.
Enhancing Augmentation with “Growth Factors”
The use of enamel matrix proteins has been documented for 20 years. Early studies showed root coverage, formation of acellular cementum, and insertion of connective tissue fibers completing regeneration of the soft tissue attachment to a previously denuded root surface.22 Continued studies have proven periodontal regeneration with elimination of the possibility of root resorption.23 Other research has shown increased predictability of root coverage when enamel matrix proteins are used as part of the surgical procedure.24 Fewer studies, still with promising results, have been done with other biologic modifiers like rh-PDGF.25
One novel allograft tissue (EpiFix, Snoasis Medical, www.snoasismedical.com) being used for periodontal augmentation is obtained from amnion tissue (Figure 12). The manner in which the graft material is harvested and processed leaves numerous components that aid tissue healing. In the basement membrane layer are type 3, 4, and 5 collagen and factors with bioactivity for cell adhesion (laminins and fibronectin)26 and angiogenesis.27 These binding factors contribute to success in terms of the percentage of the original defect that is covered by vital tissue. Additionally, the growth factors clinically appear to enhance the speed of healing in the recipient site.
Numerous options are available to the periodontist for treating patients who have root exposure. Treating these sites early in the process may prevent furcation involvement and caries formation. Use of biologically active grafts and/or enhancing factors (Figure 13) may lead to true regeneration and full structural reconstitution in the defect site. Proper flap management will assist in optimizing the results of the surgical procedure.28 Optimal home care and keeping patients off cigarettes and away from habits that can detrimentally affect the soft tissues should enable them to maintain the benefits from these procedures for their lifetime.
Disclosure
The author is a current consultant for Snoasis Medical.
References
1. Lang N, Loe H. The relationship between the width of keratinized gingival and gingival health. J Periodontol. 1972;43:623-627.
2. Levin L, Samorodnitzky-Naveh GR, Machtei EE. The association of orthodontic treatment and fixed retainers with gingival health. J Periodontol. 2008;79(11):2087-2092.
3. Dorfman HS, Kennedy JE, Bird WC. Longitudinal evaluation of free autogenous gingival grafts. A four year report. J Periodontol. 1982;53(6):349-352.
4. Bouri A Jr, Bissada N, al-Zahrani MS, et al. Width of keratinized gingiva and the health status of the supporting tissues around dental implants. Int J Oral Maxillofac Impl. 2008;23(2):323-326.
5. Burkhardt R, Joss A, Lang NP. Soft tissue dehiscence coverage around endosseous implants: a prospective cohort study. Clin Oral Implants Res. 2008;19(5):451-457.
6. Ali Fayyad M, Jamani KD, Agrabawi J. Geometric and mathematical proportions and their relations to maxillary anterior teeth. J Contemp Dent Pract. 2006;7(5):62-70.
7. Lombardi RE. The principles of visual perception and their clinical application to denture esthetics. J Prosthet Dent. 1973;29:358-382.
8. Sonick M. Esthetic crown lengthening for maxillary anterior teeth. Compend Contin Educ Dent. 1997;18(8):807-819.
9. Harris RJ. Gingival augmentation with an acellular dermal matrix: Human histologic evaluation of a case placement of the graft on periosteum. Int J Periodontics Restorative Dent. 2004;24:378-385.
10. Remya V, Kishore Kumar K, Sudharsan S, Arun KV. Free gingival graft in the treatment of class III gingival recession. Indian J Dent Res. 2008;19(3):247-252.
11. Miller PD Jr. Root coverage using free soft tissue autograft following citric acid application, III: A successful and predictable procedure in areas of deep-wide recession. Int J Periodontics Restorative Dent. 1985;5:14-37.
12. Souza SL, Macedo GO, Tunes RS, et al. Subepithelial connective tissue graft for root coverage in smokers and non-smokers: a clinical and histologic controlled study in humans. J Periodontol. 2008;79(6):1014-1021.
13. Andia DC, Martins AG, Casati MZ, et al. Root coverage outcome may be affected by heavy smoking: a 2-year follow-up study. J Periodontol. 2008;79(4):647-653.
14. Haggerty PC. The use of a free gingival graft to create a healthy environment for full crown preparation. Case history. Periodontics. 1966;4(6):329-331.
15. Douglass GL. Mucogingival repairs in periodontal surgery. Dent Clin North Am. 1976;20(1):107-130.
16. Langer B, Calagna L. The subepithelial connective tissue graft. J Prosth Dent. 1980;44(4):363-367.
17. Cortellini P, Tonetti M, Baldi C, et al. Does placement of a connective tissue graft improve the outcomes of coronally advanced flap for coverage of single gingival recessions in upper anterior teeth? A multi-centre, randomized, double-blind, clinical trial. J Clin Perio. 2009;36(1):68-79.
18. Studer SP, Allen EP, Rees TC, Kouba A. The thickness of masticatory mucosa in the human hard palate and tuberosity as potential donor sites for ridge augmentation procedures. J Periodontol. 1997;68(2):145-151.
19. Cummings LC, Kaldahl WB, Allen EP. Histologic evaluation of autogenous connective tissue and acellular dermal matrix grafts in humans. J Periodontol. 2005;76(2):178-186.
20. Yan JJ, Tsai AY, Wong MY, Hou LT. Comparison of acellular dermal graft and palatal autograft in the reconstruction of keratinized gingiva around dental implants: a case report. Int J Periodontics Restorative Dent. 2006;26(3):287-292.
21. Mele M, Zucchelli G, Montevecchi M, Checchi L. Bilaminar technique in the treatment of a deep cervical abrasion defect. Int J Periodontics Restorative Dent. 2008;28(1):63-71.
22. Heijl L. Periodontal regeneration with enamel matrix derivative in one human experimental defect. A case report. J Clin Perio. 1997;24(9 Pt 2):693-696.
23. McGuire MK, Cochran DL. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue. Part 2: Histological evaluation. J Periodontol. 2003;74(8):1126-1135.
24. Cheng YF, Chen JW, Lin SJ, Lu HK. Is coronally positioned flap procedure adjunct with enamel matrix derivative or root conditioning a relevant predictor for achieving root coverage? A systemic review. J Perio Res. 2007;42(5):474-485.
25. McGuire MK, Scheyer ET. Comparison of recombinant human platelet-derived growth factor-BB plus beta tricalcium phosphate and a collagen membrane to subepithelial connective tissue grafting for the treatment of recession defects: a case series. Int J Periodontics Restorative Dent. 2006;26(2):127-133.
26. Pakkala T, Virtanen I, Oksanen J, et al. Function of laminins and laminin-binding integrins in gingival epithelial cell adhesion. J Periodontol. 2002;73(7):709-719.
27. Koizumi NJ, Inatomi TJ, Sotozono CJ, et al. Growth factor mRNA and protein in preserved human amniotic membrane. Curr Eye Res. 2000;20(3):173-177.
28. Greenwell H, Vance G, Munninger B, Johnston H. Superficial-layer split-thickness flap for maximal flap release and coronal positioning: a surgical technique. Int J Periodontics Restorative Dent. 2004;24(6):521-527.
About the Author
Robert A. Horowitz, DDS
Assistant Clinical Professor in Implant Dentistry and Periodontics
College of Dentistry New York University
New York, New York
Private Practice in Periodontics and Implant Dentistry
Scarsdale, New York