Advanced Periodontal Therapy—To Save or Replace the Questionable Tooth
Robert A. Horowitz, DDS
The accumulation of plaque at the gingival margin leads to gingivitis. According to the American Academy of Periodontology, gingivitis is inflammation of the gingival tissue without the loss of connective tissue. The clinical signs of gingivitis are increased formation of plaque at the gingival margin, bleeding, and the superficial signs of inflammation in the marginal tissues. As the effects of this condition are localized to the tissues above the alveolar crest, there is no loss of attachment of either gingiva or bone. Areas where this condition is present have no deeper pockets, although there may be bleeding on probing because of the edematous superficial tissues.1
When the appropriate number and type(s) of bacteria colonize the subgingival pocket, root, and gingival surfaces, their waste products can cause two phenomena to occur. One of these is that the acids released cause destruction of the osseous and soft tissues in the region. Additionally, the lipopolysaccharides on the cell walls of bacteria and other released products from the cells lead to an inflammatory cascade that also has destructive effects on the periodontal attachment apparatus. The combination of these effects brings about periodontal disease and the loss of bone and soft tissue attachment to the surface of the tooth.1
The definition of periodontal disease is an inflammatory process of the gingival tissues and/or periodontal membrane of the teeth, resulting in an abnormally deep gingival sulcus, possibly producing periodontal pockets and loss of supporting alveolar bone.1 This process has been documented as being site-specific and episodic in nature, leading to the diagnosis of patients who exhibit pocketing and bone loss in different regions of the mouth and at varied time points. Increased pocketing leads to greater deposits of calculus, bacteria, and biofilms on root surfaces below the level of the visible gum margin. Research has shown repeatedly that calculus deposits remain in deeper locations and at the margins of restorations even after non-surgical periodontal therapy.2,3 Even if the bacteria can be removed with instrumentation and kept out of the site for extended periods of time with adjunctive therapies, the roughened root surfaces where calculus has not been eliminated and the roots are planed will enable the colonization of bacteria and the attachment of biofilms.
The association between increased probing depths and periodontal bacteria and alterations of diabetic state, low birth weights, stroke, cardiovascular disease, and other systemic conditions has been shown in a number of articles. Beginning almost 50 years ago, the various relationships between periodontal disease and other diseases have been described. In an early medical article, there were discussions of interactions between diabetes and the periodontal condition.4 Later studies have used enzymatic markers to show that there is a link between a patient’s periodontal status and the level of diabetic control.5,6 In part, this may be caused by the specific bacteria Porphyromonas gingivalis.7 Specific actions with this bacteria and other factors may contribute to the increased risk of advanced periodontal bone loss in diabetic patients.8-10 Another study was performed to analyze the interrelationships between the severity of periodontal disease and the presence of symptoms of type I diabetes mellitus.11 Patients in this population that had symptoms of diabetes for longer than 5 years pre-sented with a higher number of missing teeth and greater average attachment loss than patients with diabetes for less than 5 years. In children, the severity of periodontal disease has been shown to be related to the amount of metabolic control of their diabetes.12 To help maintain these patients in a healthier periodontal state, careful monitoring and treatment of their diabetic condition is important.
A number of studies have been performed to attempt to identify the interaction between periodontal disease and pregnancy. One of these studies is a meta-analysis of published data and summary of research done to date.13 The effects of periodontitis were studied in relation to low birth weight, premature birth, miscarriage, and other adverse occurrences during pregnancy. Thirty of the analyzed studies showed a positive odds ratio between periodontitis and adverse pregnancy outcomes, ranging from 1 to 20 times the normal rate. When patients underwent prophylaxis and periodontal therapy, the studies showed a significant reduction in preterm, low-birth-weight babies. However, there was not a significant reduction in the rate of either preterm birth or low birth weight alone. While individual studies demonstrated these occurrences, the authors concluded that more rigorous studies need to be performed before there is a general consensus on the interactions between periodontal disease and pregnancy. Another recent study concluded that periodontal disease is correlated to having babies with a lower birth weight.14 A group of 450 pregnant Brazilian women was followed through their pregnancies. One group of patients with periodontal disease had periodontal treatment performed. Of those mothers who did not have the same therapy, 79% had preterm, low-birth-weight babies—a statistically significantly higher amount than those who had undergone dental treatments.
Certain systemic diseases may have an effect on the periodontal status of our patients. In patients suffering from rheumatoid arthritis, there are alterations in the capillary patterns.15 The changes in the number of elongated capillaries and decreased diameter of the capillaries were found in the periodontal tissues of patients with rheumatoid arthritis. These alterations may negatively affect the patient’s ability to fight periodontal disease, worsening the gingival and alveolar affects of this complex disease process. Another study determined that patients with periodontal disease have impaired function of their kidneys.16 In these individuals, antibodies of immunoglobulin G to periodontal bacteria, independent of other more common risk factors, were related to decreased renal function.
The purpose of this article is to present the restorative dentist with ideas of how to discuss the options of maintaining or replacing a tooth with significant bone loss. It is then up to the dentist to either treat the site or refer the patient to a practitioner with the latest knowledge of materials and techniques to accomplish this task. While the controversies of who should treat and whether the tooth should be saved or extracted draw many attendees to lectures at major symposia, the following cases and literature will help give dentists information to assist their decision-making process.
Case Reports
Patient 1
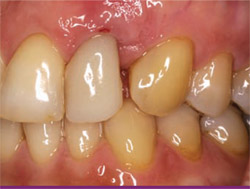
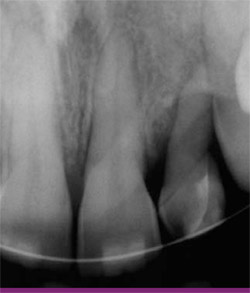
This patient presented with a non-maintainable maxillary lateral incisor. The tooth was short and exhibited Class 2 to Class 3 mobility, 4-mm to 6-mm pocket depths and was not in an esthetically acceptable location (Figure 1). There was no facial bone over the root of the tooth, which was quite short, and there was significant lateral bone loss on the radiograph (Figure 2). There was some gingival recession, causing an esthetic difference between the two sides of the patient’s mouth. The determination was made to perform forced eruptive orthodontic therapy in the incisal and facial directions before extraction. In this way, the amount of gingival tissue and residual alveolar bone could be maximally augmented with a non-surgical approach and then augmented as needed during one of the scheduled surgical procedures for extraction and implant placement. The active period of forced eruption took 3 months and was followed by a 6-week period of stabilization to enable maturation of gingival and osseous tissues.
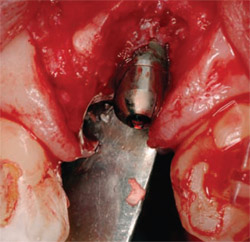
At the time of tooth extraction, no hard tissue regenerative therapy was performed, as there was sufficient alveolar bone volume for implant placement. The soft tissues were allowed to heal and keratinize for 2 months before surgical insertion of the implant (Figure 3). The implant was placed in an ideal position as a result of the amount of regenerated tissue (Figure 4). At that visit, the bone was augmented with a graft consisting of demineralized freeze-dried bone allograft (DFDBA), calcium sulfate with a binding material to improve graft handling and healing (CalMatrix™, Lifecore Dental, Chaska, MN) with a calcium-sulfate barrier over the site (CalForma™, Lifecore Dental) (Figure 5). Coronally, the implant used in this case had a laser-micro-grooved surface designed to prevent epithelial downgrowth and to support gingival tissue attachment to the implant surface (BioHorizons, Birmingham, AL).17 These surface modifications enhance connective tissue fiber attachment to the surface of the implant. At the same time, this enables optimal preservation of crestal hard and soft tissue. In this manner, the esthetic nature of the tissue sculpted around the final implant-borne restoration has been preserved for more than 3 years (Figure 6).
Patient 2
This patient was referred by her restorative dentist for evaluation of a lower left premolar. Clinical evaluation revealed probing depths of 8 mm around most of tooth No. 20 (Figure 7) and the appearance on the radiograph of a moat-shaped lesion. As the infrabony defect was deep, literature has shown that even scaling and root planing therapy would not predictably remove the accretions from the root surface. To resolve the defect by regeneration of bone rather than epithelial adhesion, the determination was made to treat the area with a surgical approach only.
At the time of surgery, the lesion measured deeper clinically than originally estimated during probing (Figure 8). There could be a number of reasons for this occurrence. When subgingival tissues are inflamed, often patients are too sensitive to enable accurate probing. Other factors that cause inaccurate probing depths are residual calculus on the root surface, narrow infrabony lesions, and probing into or beyond the base of the junctional epithelium when it is inflamed. The 10-mm probing depth at the disto-buccal surface was the beginning of the infrabony trough that passed under the disto-buccal contact point and ended as a 7-mm infrabony pocket at the mesio-lingual line angle (Figure 9). These areas were thoroughly debrided with hand and ultrasonic instruments to remove all remaining tissue tags and residual calcareous accretions. The roots were then treated with a mild citric acid solution to remove the smear layer, exposing the collagen fibrils at the distal ends of the dentinal tubules. The root surfaces were treated with enamel matrix derivative (Emdogain, Straumann USA LLC, Andover, MA) to enhance regeneration in the site.18,19 Because of the narrow nature of the infrabony pocket and relatively thick gingival tissue, no graft was placed in the defect and no barrier was placed over it. The soft tissues were sutured back to their original position and the area was allowed to heal.
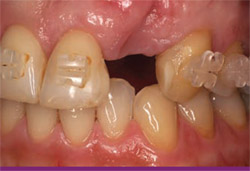
Over time, the area was seen to exhibit a complete elimination of the infrabony defect both from clinical measurements and radiographic interpretation. By the 6-month probing time, the pocket depths had been reduced to 3 mm on all surfaces of the tooth. This healed, shallow depth remained constant over the 6 years that the site was followed (Figure 10). Six years after the initial surgery, the area was opened at the time of placement of an adjacent dental implant. After flap elevation and gentle debridement of the coronal soft tissues, there appeared to be no residual infrabony lesion on any surface of the tooth (Figure 11). Compared to measurements that were taken at the time of the regenerative surgery, there was a gain of 5 mm to 9 mm of probing bone level around the tooth. The surgical application of enamel matrix derivative in this case enabled maintenance of the tooth for the patient’s lifetime in a healthy state.
Patient 3
This patient presented with a class III furcation involvement in the lower left second molar tooth (No. 18) (Figure 12 and Figure 13). As therapy to regenerate bone and periodontal attachment in this type of lesion is unpredictable20 and there was a tooth missing anterior to this tooth, the decision was made to extract the tooth, regenerate the missing alveolar bone and place two dental implants to support a fixed prosthetic device. Extraction of the tooth was performed after administration of local anesthesia and elevation of full-thickness facial and lingual flaps. The tooth was sectioned and the roots were extracted individually using periotomes and thin-bladed elevators and proximators. After socket debridement, a thin buccal plate remained that was deficient in buccal height (Figure 14). A bone graft material combination was placed in the defect to rebuild the alveolar height and width (Figure 15). The mixture consisted of DFDBA with calcium sulfate and a binding material (CalMatrix). This graft mixes like a putty and enables the bone replacement graft material to maintain its shape in a large defect. The material was covered by a putty-like mixture of calcium sulfate that sets in a bloody field (Cal-Forma). The facial and lingual flaps were repositioned to fully cover the graft material and barrier. Previous studies have shown the histologic advantage of this material compared to the use of bovine graft products.21
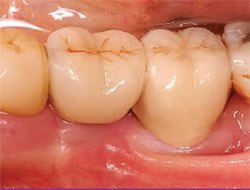
Six months postoperatively, the area was opened surgically to prepare for the placement of dental implants. Comparison of the initial (Figure 16) and final radiographs (Figure 17) shows that the grafted site is more dense than the surrounding bone. Full restoration of alveolar height and width had occurred (Figure 18), enabling the placement of two one-stage dental implants (Stage 1, Lifecore Dental) in ideal locations (Figure 19). Three months after implant insertion, solid abutments were torqued into place (Figure 20). After final impressions were taken, cementable crowns were fabricated and placed (Figure 21).
Discussion
Periodontal regeneration and/or formation of new bone in infrabony defects has been demonstrated in a number of studies.18,22-25 The use of combinations of grafts and the advent of growth enhancers is improving the predictability of these results. When patients have teeth with questionable prognoses, these techniques and materials can be used to regenerate bone in infrabony pockets. Histologic studies have shown that periodontal regeneration, including cementum and new periodontal ligament fibers, is possible with the technique shown in one of these cases and with other methods of therapy as well.24,26,27 This is the ideal therapy that can be offered to our patients when teeth missing periodontal bone support are to be saved. By performing periodontal regeneration, the complete attachment apparatus to the tooth can be restored, enabling the patient to maintain the tooth in a state of optimal health.
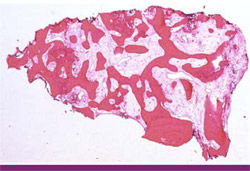
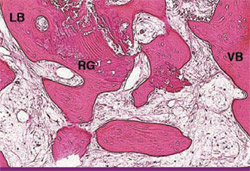
One goal of alveolar bone regeneration when teeth have been extracted should be the maximization of vital alveolar bone formation in the site. In the extraction case shown here, 55% bone was regenerated after this therapy (Figure 22). Other articles have shown the clinical and histologic advantages of mixing calcium sulfate with bone replacement graft materials.28-31 This is significantly more vital bone than has been shown with the use of other graft materials.32,33 The residual amount of graft material in the evaluated specimen was less than 3% (Figure 23) and the beginning of lamellar bone formation shows the advanced level of healing even at this early time period.
No matter what the future restorative needs are in a specific area, the volume of hard and soft tissue preserved or augmented should be maximized. In the case of fixed, non-implant-supported restorations and removable restorations, the amount of residual graft material in a site may not matter. When dental implants are planned for insertion into these sites, maximizing the percentage of vital alveolar bone can have some advantages. A denser recipient site will increase the initial stability of implants at the time of placement. When dental implants are placed in sites with a higher content of vital bone, there is greater initial bone-to-implant contact. The surfaces of the implant may then have greater resulting osseointegration because of osteoconduction from recruitment and migration of osteogenic cells to the implant surface.34 Initially, the osteogenic cells migrate through the residue of the peri-implant blood clot, followed by natural bone tissue being laid down on the implant surface.35
Either of these results will enable maximum preservation of alveolar bone around a tooth or dental implant. Minimizing the residual periodontal pocket through these surgical techniques, proper recare schedules, and home care by the patient will leave the patient with minimal chance of bacterial invasion through the bloodstream. In this way, patients will be able to speak and eat more properly and maintain implant-retained prostheses longer and, more importantly, remain in a better state of overall systemic health.
Acknowledgments
Special thanks to my colleagues and friends for pushing my search for the biologic rationale behind the work we do. Extra special thanks to Dr. Joan Phelan and Ms. Gloria Turner from the NYU College of Dentistry Department of Oral Pathology for assistance in preparation and analysis of the core sample discussed in this article. I would also like to thank Drs. Theo Mantzikos and Ilan Shamus for the orthodontic forced eruption of the teeth in Patient 1.
Disclosure
The author has received grant and research support and is a current consultant for Lifecore Dental.
References
1. American Academy of Periodontology. Current Procedural Terminology for Periodontitis. 11th ed. 2007; 90.
2. Waerhaug J. Presence or absence of plaque on subgingival restorations. Scand J Dent Res. 1975;83(1):193-201.
3. Caffesse RG, Sweeney PL, Smith BA. Scaling and root planing with and without periodontal flap surgery. J Clin Periodontol. 1986;13(3):205-210.
4. Williams RC Jr, Mahan CJ. Periodontal disease and diabetes in young adults. JAMA. 1960;172:776-778.
5. Thorstensson H, Hugoson A. Periodontal disease experience in adult long-duration insulin-dependent diabetics. J Clin Periodontol. 1993;20(5):352-358.
6. de Pommereau V, Dargent-Pare C, Robert JJ, Brion M. Periodontal status in insulin-dependent diabetic adolescents. J Clin Periodontol. 1992;19(9 Part 1):628-632.
7. Ojima M, Takeda M, Yoshioka H, et al. Relationship of periodontal bacterium genotypic variations with periodontitis in type 2 diabetic patients. Diabetes Care. 2005;28 (2):433-434.
8. Nelson RG, Shlossman M, Budding LM, et al. Periodontal disease and NIDDM in Pima Indians. Diabetes Care. 1990;13(8): 836-840.
9. Emrich L, Shlossman M, Genco RJ. Periodontal disease in non-insulin-dependent diabetes mellitus. J Periodontol. 1991;62(2): 123-131.
10. Taylor GW, Burt BA, Becker MP, et al. Non-insulin dependent diabetes mellitus and alveolar bone loss progression over 2 years. J Periodontol. 1998;69(1):76-83.
11. Al-Shammari KF, Al-Ansari JM, Moussa NM, et al. Association of periodontal disease severity with diabetes duration and diabetic complications in patients with type 1 diabetes mellitus. J Int Acad Periodontol. 2006;8(4):109-114.
12. Lalla E, Cheng B, Lal S, et al. Diabetes-related parameters and periodontal conditions in children. J Periodontal Res. 2007;42(4): 345-349.
13. Xiong X, Buekens P, Vastardis S, Yu SM. Periodontal disease and pregnancy outcomes: state-of-the-science. Obstet Gynecol Surv. 2007;62(9): 605-615.
14. Gazolla CM, Ribeiro A, Moyses MR, et al. Evaluation of the incidence of preterm low birth weight in patients undergoing periodontal therapy. J Periodontol. 2007;78(5):842-848.
15. Scardina GA, Messina P. Microvascular periodontal alterations: a possible relationship between periodontitis and rheumatoid arthritis. Clin Hemorheol Microcirc. 2007;37(3):229-235.
16. Kshirsagar AV, Offenbacher S, Moss KL, et al. Antibodies to periodontal organisms are associated with decreased kidney function. The Dental Atherosclerosis Risk In Communities study. Blood Purif. 2007;25(1):125-132.
17. Ricci JL, Charvet J, Frenkel SR, et al. Bone response to laser microtextured surfaces. In: Bone Engineering. Davies JE, ed. Toronto, Ont, Canada. Em2 Inc. 2000:282-294.
18. Cortellini P, Tonetti MS. A minimally invasive surgical technique with an enamel matrix derivative in the regenerative treatment of intra-bony defects: a novel approach to limit mor-bidity. J Clin Periodontol. 2007;34(1):87-93.
19. Pimentel SP, Sallum AW, Saldanha JB, et al. Enamel matrix derivative versus guided tissue regeneration in the presence of nicotine: a histomorphometric study in dogs. J Clin Periodontol. 2006;33(12):900-907.
20. Froum SJ, Tarnow D. Modified coronally positioned flap for obtaining new attachment in Class 2 and 3 furcation defects. Part I: Rationale and surgical technique. Int J Periodontics Restorative Dent. 1995;15(5):462-473.
21. Vance GS, Greenwell H, Miller RL, et al. Comparison of an allograft in an experimental putty carrier and a bovine-derived xenograft used in ridge preservation: a clinical and histologic study in humans. Int J Oral Maxillofac Implants. 2004;19(4):491-497.
22. Froum SJ, Weinberg MA, Rosenberg E, Tarnow, D. A comparative study utilizing open flap debridement with and without enamel matrix derivative in the treatment of periodontal intrabony defects: a 12-month re-entry study. J Periodontol. 2001;72(1):25-34.
23. Dereka XE, Markopoulou CE, Vrotsos IA. Role of growth factors on periodontal repair. Growth Factors. 2006;24(4):260-267.
24. Nevins M, Camelo M, Nevins ML, et al. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J Periodontol. 2003;74(9):1282-1292.
25. McGuire MK, Kao RT, Nevins M, Lynch SE. rhPDGF-BB promotes healing of periodontal defects: 24-month clinical and radiographic observations. Int J Periodontics Restorative Dent. 2006;26(3): 223-231.
26. Bowers GM, Chadroff B, Carnevale R, et al. Histologic evaluation of new attachment apparatus formation in humans. Part III. J Periodontol. 1989;60(12):683-693.
27. Yukna RA, Mellonig JT. Histologic evaluation of periodontal healing in humans following regenerative therapy with enamel matrix derivative. A 10-case series. J Periodontol. 2000;71(5):752-759.
28. al Ruhaimi KA. Effect of adding resorbable calcium sulfate to grafting materials on early bone regeneration in osseous defects in rabbits. Int J Oral Maxillofac Implants. 2000;15(6):859-864.
29. al Ruhaimi KA. Effect of calcium sulphate on the rate of osteogenesis in distracted bone. Int J Oral Maxillofac Surg.2001;30(3): 228-233.
30. Sottosanti J, Anson D. Using calcium sulfate as a graft enhancer and membrane barrier. [Interview]. Dent Implantol Update. 2003;14(1): 1-8.
31. Anson D. Maxillary anterior esthetic extractions with delayed single-stage implant placement. Compend Contin Educ Dent. 2002;23(9):829-838.
32. Artzi Z, Givol N, Rohrer MD, et al. Qualitative and quantitative expression of bovine bone mineral in experimental bone defects. Part 2: Morphometric analysis. J Periodontol. 2003;74(8):1153-1160.
33. Artzi Z, Tal H, Dayan D. Porous bovine bone mineral in healing of human extraction sockets. Part 1: histomorphometric evaluations at 9 months. J Periodontol. 2000;71(6): 1015-1023.
34. Davies JE. Understanding peri-implant endosseous healing. J Dent Educ. 2003;67(8):932-949.
35. Davies JE. Mechanisms of endosseous integration. Int J Prosthodont. 1998;11(5):391-401.
 |
 |
|
| Figure 1 Initial clinical presentation showing an esthetic problem. | ||
 |
||
| Figure 2 Periapical radiograph before therapy revealing significant bone loss around tooth No. 10. | ||
| Figure 3 Soft tissue healing 2 months after eruption and subsequent extraction of tooth No. 10. | ||
 |
 |
|
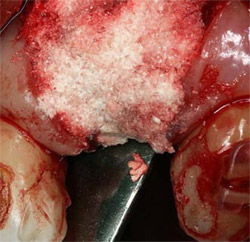
| Figure 4 Implant placed in ideal location, fully surrounded by alveolar bone. | Figure 5 Implant site augmented with a combination of DFDBA, calcium sulfate, and binder. | |
 |
 |
|
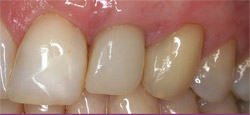
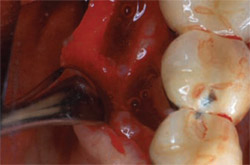
| Figure 6 This image shows the maintenance of esthetics and gingival contours 3 years after loading. | Figure 7 Initial clinical appearance of 7-mm to 8-mm trough-shaped infrabony defect. | |
 |
 |
|
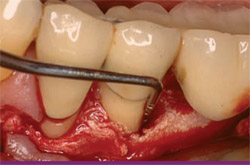
| Figure 8 After flap elevation, the full depth of the disto-buccal alveolar defect is realized. | Figure 9 Occlusal view of lingual trough after debridement of the infrabony defect. | |
 |
||
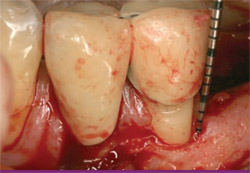
| Figure 10 Clinical view of the healed site 6 years after therapy with enamel matrix derivative. | ||
 |
 |
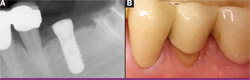
|
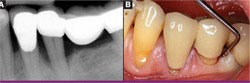
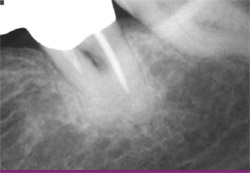
| Figure 11 View of healed bony site showing full restoration of alveolar bone. | Figure 12 Initial radiograph of tooth No. 19 showing class III furcation defect. | |
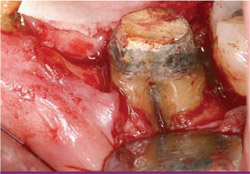 |
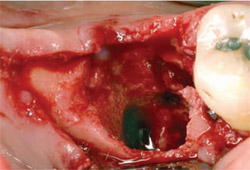 |
|
| Figure 13 After flap elevation, the fully exposed through-and-through furcation defect is seen. | Figure 14 The residual bone around the socket has a thin buccal plate and is deficient vertically. | |
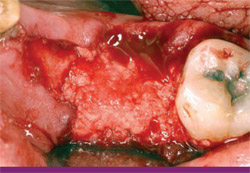 |
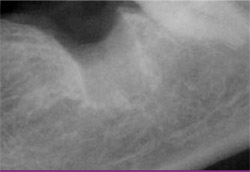 |
|
| Figure 15 A combination graft of DFDBA, calcium sulfate, and a binder has been inserted into the defect, filling it to ideal contour. | Figure 16 The immediate post-extraction radiograph shows the extent of bone loss after extraction. | |
 |
 |
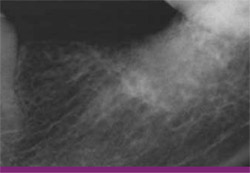
|
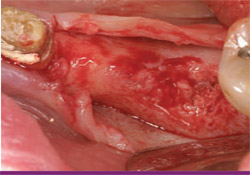
| Figure 17 This radiograph, taken on the day of implant placement, shows dense bone regenerated to the alveolar crest. | Figure 18 After flap elevation, a fully restored alveolar ridge is visualized. | |
 |
||
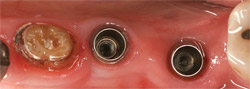
| Figure 19 Two Stage 1 dental implants are placed ideally in a wide, healed ridge. | ||
 |
 |
|
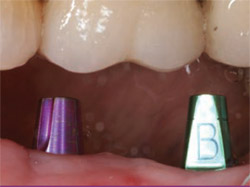
| Figure 20 Two color-coded abutments are torqued into place, simplifying impressions, temporization, and final restoration with cementable crowns. | Figure 21 Two single crowns are cemented in ideal locations with adequate bone and keratinized tissue. | |
 |
 |
|
| Figure 22 In this hematoxylin and eosin stained specimen, 55% bone is seen in the regenerated area. | Figure 23 Closer inspection of the sample reveals 97% of the regenerated material is vital bone (RG = residual graft, VB = vital bone, LB = lamellar bone). | |
| About the Author | ||
 Robert A. Horowitz, DDS Robert A. Horowitz, DDSAssistant Clinical Professor in Implant Dentistry and Periodontics College of Dentistry New York University New York, New York Private Practice in Periodontics and Implant Dentistry Scarsdale, New York |
||




