Screening for Oral Cancer: A Matter of Life and Death
Michael A. Kahn, DDS
Reprinted with permission from the Journal of the Massachusetts Dental Society.
During dental school admission interviews, applicants often mention that one of the appeals of choosing dentistry over medicine is the avoidance of a life and death situation. However, if a dentist is a vigilant clinician and an astute and knowledgeable diagnostician, then he or she may be involved in the early detection of one of the deadliest forms of cancer. The American Cancer Society (ACS) has estimated that 29,370 new cases of oral cavity/oropharyngeal cancer occurred in the United States during the year 2005.1 This incidence is nearly three times the amount of estimated cervical carcinoma cases during the same time period and nearly 5,000 more than the estimated new cases of thyroid cancer. Although the ACS estimates that both this past year’s cases of leukemia and cutaneous melanoma occurred more often—34,810 and 59,580, respectively—oral cancer continues to represent a significant unknown, initially silent, and subsequently painful, morbid disease.1
Therefore, it is incumbent that each licensed American dentist and the remainder of the dental health care delivery team be regularly reminded of the importance of oral cancer screening as well as refreshed in the proper head and neck soft tissue examination technique. The purpose of this article is to update the dental clinician on the importance and proper technique of oral cancer screening as well as review some important information about oral premalignant lesions.
SQUAMOUS CELL CARCINOMA
Overwhelmingly in the United States, the most frequent type of oral cancer is squamous cell carcinoma, which arises from the stratified squamous epithelial mucosal lining of the oral cavity.2-5 Epidemiologic studies have shown that approximately 95% of oral cancer is of this type, with the remaining 5% distributed among soft and hard tissue sarcomas, primary or metastatic melanoma, and salivary gland tumors.2,4-6 For this reason, unless specifically noted, the remainder of this article will deal with squamous cell carcinoma of the oralcavity proper and the adjacent skin and vermilion border of the lips.
Factors Affecting Oral Cancer
Every hour in the United States, three additional oral cancer cases are diagnosed and, during that same hour, one person dies of oral cancer. Both of these sobering facts have held constant for nearly the past 30 years. The male-to-female incidence ratio of oral cancer in the United States is currently 2:1, an increase from the 3:1 ratio of a generation ago, which is primarily a result of the increase in tobacco use by women.4,5,7-9 Despite these disturbing statistics, there is one glimmer of hope during the past year. The ACS and the American Dental Association (ADA) have reported that for the first time in nearly 30 years, the relative 5-year overall survival rate improved to 59% (all races combined), which is a small, but significant positive change from the 50% to 55% cited for the past 20 years.1 This encouraging trend may be a result of earlier detection of lesions by the dental health care delivery team with subsequent, timely surgical biopsies. Despite this promising news, there remains a glaring disparity of 5-year survival rates when they are stratified by race (eg, white vs black), with white people having a 61% 5-year survival rate vs a 39% 5-year survival rate for black people.1
Tobacco, whether smoke (eg, cigarettes, cigars, pipe) or smokeless (eg, chewing tobacco, snuff), continues to be the number one etiologic factor of oral cancer. Alcohol is considered an important and significant synergistic cofactor (the aldehyde component in alcohol is thought to be the primary culprit).2,5,7-9 If a person smokes cigarettes, it is estimates that he or she is 8 to more than 20 times more likely to develop squamous cell carcinoma. If that same person also overindulges in the use of ethanol (beverage alcohol), then the likelihood of oral cancer can rise to more than 80-fold.7
Other known etiologic or influencing factors of oral cancer include therapeutic radiation or, in the case of lip cancer, ultraviolet radiation. Certain viruses, such as herpes simplex type VIII, Epstein-Barr virus and, particularly, human papillomavirus are thought to be, at times, oncogenic.10,11 The immune status of the individual is also important because when the immune system is compromised, oral cancer is much more likely.4 Diet and nutrition remain significant factors and recent molecular biological studies show that sensitivity to mutagens, metabolic enzymes polymorphisms, errors or lack of DNA repair, and certain cancer proto-oncogenes and tumor suppressor genes may play a significant role.12
Concept and Degree of Dysplasia
All dentists should refresh their knowledge about the concept and degree of premalignant oral lesions exhibiting various amounts of dysplasia the interruption of the normal maturation pattern of the oral mucosa’s epithelium. Subjective amounts of dysplasia are categorized as mild, moderate, severe, or in-situ carcinoma depending on the amount of epithelial thickness that exhibits dysplasia. Mild dysplasia is generally described as involving only the lower one fourth of the epithelium’s thickness, moderate dysplasia involves one half, severe dysplasia (Figure 1) three fourths, and in-situ carcinoma (Figure 2) has top-to-bottom involvement but without invasion of the underlying lamina propria (ie, connective tissue) via violation of the basement membrane zone.
It is well known that a woman’s cervical epithelium may have similar degrees of dysplasia; however, the process of progression is slower and consistently sequential. Unfortunately, many dentists do not realize that early, mild dysplasia of the oral epithelium does not necessarily progress through the more involved stages of dysplasia, but may invade at any time. Therefore, it is imperative that dentists continue to screen their patients for early premalignant lesions and have them thoroughly removed, even if only mild dysplasia is present.
Warning Signs and Symptoms
The dental team should always be cognizant of the classic clinical warning signs of oral cancer: difficulty in chewing or speaking; a lump or thickening in the mucosa, glands, or lymph node area; an ulceration that does not heal; abnormal bleeding; and unexplained pain or paresthesia.13 Although this article does not discuss intrabony (ie, jaw) cancer, the clinician should remember the classic suspicious-for-malignancy radiographic interpretation signs: asymmetry, irregular borders, focal and symmetrical widening of the periodontal ligament, apical root resorption with a “spike or nail” appearance, and, in young persons, superior displacement of erupting teeth.5
It is vitally important that the dental health care provider be familiar with the nature and appearance of surface changes that indicate possible premalignant or malignant change. Head and neck cutaneous surface changes in color, texture, and morphology should always be investigated and the patient informed. Lip cancer of the skin and vermilion border as a result of chronic ultraviolet exposure may occur in any geographical area of the United States despite the belief that this condition is unique to the sunbelt states. The classic visual warning signs are an uneven coloration, splotchiness, development of a scale or crust, or a blurring of the usual distinct border between the vermilion and skin or labial mucosa.4
Within the oral cavity proper, the most common premalignant clinical changes are leukoplakia or erythroplakia (Figure 3A and Figure 3B). These terms are solely clinical descriptions of a white or red plaque (slightly elevated above the plane of the mucosa) or patch that does not wipe off and cannot receive an alternative specific diagnosis. It is imperative to remember that there is no correlation between the size, color, or texture of a leukoplakia or erythroplakia and its final histological diagnosis. Rarely, a combination of the two, erythroleukoplakia (speckled leukoplakia), is also seen (Figure 4). Ideally, a biopsy of these clinical lesions must be taken as soon as possible unless a specific etiology (eg, defective restoration, denture clasp) is suspected, eliminated, and the lesion resolves within 2 weeks.
Eighty-five percent of oral premalignancies or malignancies are leukoplakias.4,6,13,14 They typically occur in a man with an average age of 60 years. Eighty-percent of patients with leukoplakias are cigarette smokers and those persons who smoke frequently have more and larger lesions. Waldron and Shafer reported that when biopsies of leukoplakias are taken and subsequently examined microscopically, only 20% have hyperkeratosis with some degree of dysplasia.14 Interestingly, the least common sites for leukoplakia, the tongue and floor of the mouth, are the most likely to be dysplastic, 25% and 50%, respectively.14
Although rarer, erythroplakias are clinically more significant. Shafer and Waldron found that 91% prove to have severe dysplasia or worse.15 They, too, are seen most often in elderly men with an average age of 70 years. The most common intraoral sites for erythroplakia are the lateral tongue, floor of mouth, soft palate, and alveolar ridge.
Classic oral pathology studies of the 1970s and 1980s found and validated certain oral and paraoral areas as the highest risk sites for premalignant and malignant oral cancer.7,14,15 The most common extraoral site is the skin and vermilion of the lower lip. Within the oral cavity proper, the lateral and ventral surfaces of the tongue are the most common with the floor of mouth the second most common (Figure 5). The soft palate complex is also a common site of involvement, specifically the uvula, soft palate proper, anteriortonsillar pillar, and lingual retromolar trigone (Figure 6).
Head and Neck Examination
During the past few years, numerous dental journals and publications have emphasized to all dentists the importance of performing a standard, reproducible, and frequent soft tissue head and neck examination that can be competently performed in 11/2 to 3 minutes once a thorough and complete review of the patient’s medical, dental, and social history is accomplished.3,13,19-20 The two basic components of the head and neck examination, visualization and palpation, are performed adhering to a few basic principles. Namely, a properly focused and dispersed incandescent operatory light should be available and, with the patient properly positioned, the normally wet mucosal surfaces of the oral cavity proper should be dried, particularly the floor of the mouth and buccal mucosa. Palpation is particularly important to detect oral cancer does not originate from the surface epithelium and to help the clinician assess the amount of possible induration present in more advanced squamous cell carcinomas. There is no specific screening sequence that must be followed as long as the clinician can systematically repeat it in each patient. Table 1 lists a suggested screening order of the extraoral and intraoral head and neck soft tissues, with emphasis on detection of lack of symmetry or changes in texture, color, function, and morphology.
Recently, a disturbing trend has been reported in the dental literature.Tongue cancer incidence has doubled in the last 50 years, most notably in increasing numbers of patients in the age range of 20 to 40 with no contributory social history (eg, tobacco usage, ethanol abuse, etc.).21,22 Equally disturbing is that these young adult patients seem to have a very biologically active, aggressive type of squamous cell carcinoma (the average age of survival once diagnosed is often less than 1 year).
CONCLUSION
Whether the patient is young or old, there is no doubt that early detection of oral cancer is the key to the reduction of morbidity and a longer span of survival. It is absolutely imperative that each dentist develop his or her own standard soft tissue head and neck examination and make it a frequent, repeatable part of the dental appointment routine. Obviously, patients with any of the clinical and/or social factors mentioned should undergo particular scrutiny. For too long, the survivability statistics of oral cancer have languished in the realm of mediocrity. Licensed American dentists should accept the responsibility to ensure that their patients not only receive the best esthetic and functional restorative procedures and palliative care but also screening and treatment, if necessary, of carcinomatous transformation at its earliest stages.
References
1. Cancer statistics, 2005. CA: A Cancer Journal for Clinicians. 2005;55(1):10-14.2. Bouquot JE. Epidemiology. In Gnepp DR: Pathology of the Head and Neck. New York, NY: Churchill Livingstone; 1988:263-314.
3. Detecting oral cancer: a guide for health professionals. Bethesda, MD: National Institutes of Health, national Institute of Dental and Craniofacial Research; 1995.
4. Neville BW, Damm DD, Allen CM, et al. Oral and Maxillofacial Pathology, 2nd edition. Philadelphia, PA: W.B. Saunders Co; 2002.
5. Silverman S. Oral Cancer, fifth edition. Hamilton, Ontario, Canada: American Cancer Society Atlas of Clinical Oncology, BC Decker, Inc.; 2003.
6. Kleinman DV, Crossett LS, Gloeckler Ries LA, et al. Cancer of the oral cavity and pharynx: a statistics review monograph, 1973-1987, NIH Monograph. Bethesda, MD, National Institute of Dental Research; 1992.
7. Blot WJ, McLaughlin JK, Winn DM, et al. Smoking and drinking in relation to oral and pharyngeal cancer. Cancer Res. 1988;48(11): 3282-3287.
8. Choi SY, Kahyo H. Effect of cigarette smoking and alcohol consumption in the aetiology of cancer of the oral cavity, pharynx and larynx. Int J Epidemiol. 1991;20(4): 878-885.
9. Schmidt BL, Dierks EJ, Homer L, et al. Tobacco smoking history and presentation of oral squamous cell carcinoma. J Oral Maxillofac Surg. 2004;62(9):1055-1058.
10. Cox MF, Scully C, Maitland N. Viruses in the aetiology of oral carcinoma? Examination of the evidence. Br J Oral Maxillofac Surg. 1991;29(6):381-387.
11. Niv A, Sion-Vardi N, Gatot A, et al. Identification and typing of human papillomavirus (HPV) in squamous cell carcinoma of the oral cavity and oropharynx. J Laryngol Otol. 2000;114(1): 41-46.
12. Notani PN, Jayant K. Role of diet in upper aerodigestive tract cancers. Nutr Cancer. 1987;10(1-2):103-113.
13. Mashberg A, Samit AM. Early diagnosis of asymptomatic oral and oropharyngeal squamouscancer. CA Cancer J Clin. 1995;45(6): 328-351.
14. Waldron CA, Shafer WG. Leukoplakia revisited: a clinicopathologic study of 3256 oral leukoplakias. Cancer. 1975;36(4): 1386-1392.
15. Shafer WG, Waldron CA. Erythroplakia of the oral cavity. Cancer. 1975;36(3):1021-1028.
16. Glazer HS. Spotting trouble. AGD Impact. 2003;18-19.
17. Horowitz AM, Alfano MC. Perform a death-defying act. J Am Dent Assoc. 2001;132(suppl):5S-6S.
18. Mages M. Missing pieces: why aren’t more dentists performing oral cancer exams? AGD Impact. 2002;16-18.
19. Opportunistic oral cancer screening: a management strategy for dental practice. British Dental Association. 2000;6: 1-36.
20. Sciubba JJ. Oral cancer and its detection. History-taking and the diagnostic phase of management. J Am Dent Assoc. 2001;132(suppl): 12S-18S.
21. Corcoran TP, Whiston DA. Oral cancer in young adults. J Am Dent Assoc. 2000;131(6): 726.
22. Gorsky M, Epstein JB, Oakley C, et al. Carcinoma of the tongue: a case series analysis of clinical presentation, risk factors, staging and outcome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;98(5):546-552.
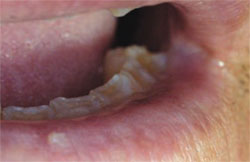 | 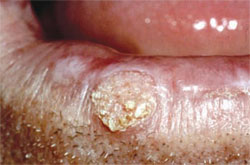 | |
| Figure 1. Sun-damaged left lower lip vermilion with subsequent biopsy demonstrating actinic keratosis with severe dysplasia. | Figure 2. Biopsy-confirmed squamous cell carcinoma of the lower lip and actinic keratosis with in situ carcinoma. | |
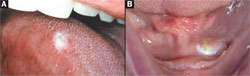 | 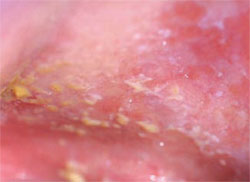 | |
| Figures 3A and 3B. An asymptomatic leukoplakia of right lateral tongue (A) and left anterior floor of mouth (B) discovered during routine oral screening. | Figure 4. Asymptomatic speckled erythroplakia of the left soft palate complex. | |
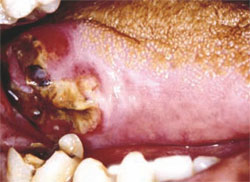 | 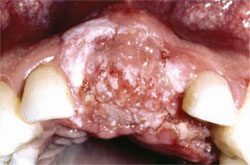 | |
| Figure 5. Squamous cell carcinoma of the right posterior lateral tongue. The lesion was initially thought to be a traumatic ulceration due to the adjacent tooth's fractured crown. | Figure 6. Papillary, exophytic, and ulcerative squamous cell carcinoma of the anterior maxillary gingiva | |
 | ||
| About the Author | ||
| Michael A. Kahn, DDS Professor Department of Oral and Maxillofacial Pathology Director Division of Professional Services Tufts University School of Dental Medicine Boston, Massachusetts | ||



