VELscope® Mucosal Examination System
The fact that oral cancer is the only form of cancer for which the morbidity rate has not decreased in the last 50 years is arguably one of the most embarrassing reflections on the dental profession. The fact that our screening technologies for this disease have not significantly changed during this same time is a reasonable, though not acceptable, justification. The signs and symptoms of oral cancer are similar to many benign conditions, such as cheek bites and denture irritation. Therefore, differential diagnosis is extremely important but has been difficult to accurately obtain. Studies have shown that dentists fail to recognize oral cancer in the majority of the cases presented to them (69%) and that they missed almost twice as many asymptomatic oral cancers as they found.1,2
Despite these facts, the discovery and diagnosis of oral cancer and oral mucosal diseases is primarily the responsibility of dentists. Physicians receive minimal training on the oral cavity and a routine part of their patients’ health assessment is to refer the patient to a general dentist for routine dental care and oral health examination. This removes them from the primary responsibility of detecting oral cancer until it reaches the later stages and has progressed beyond the oral cavity. The routine settlement against a dentist for failure to diagnosis oral cancer was $874,500 in 2002.3
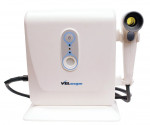
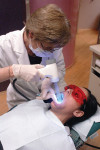
Fortunately, in the spring of 2006 LED Dental Inc (White Rock, BC, Canada) introduced the VELscope® Mucosal Examination System (Figure 1 and Figure 2). With its direct optical fluorescence visualization (FV) technology, we as dental professionals have an opportunity and the obligation to radically change the oral cancer morbidity statistics and, more importantly, change the disease state with a more thorough screening of the oral mucosa which may lead to accurate detection and early discovery. This in turn should lead to earlier diagnosis, treatment, and management, and hopefully decreases the disease’s progression and morbidity.
Oral cancer accounts for 2% to 4% of all cancers diagnosed annually in the United States, and only one half of those diagnosed are still alive 5 years after their diagnosis.4 The American Cancer Society estimates that in 2006 there will be almost 31,000 new cases of oral cancer in the United States. They also estimated that almost 8,000 people will die from oral cancer this year.5 There has been a 60% increase in oral cancer rates in adults under the age of 40 during the last 3 decades, and 25% of all cases do not fit the typical high-risk profile. All adults are at risk for developing oral cancer and should be screened routinely. Paradoxically, although tobacco is undeniably one of the two major etiological agents for oral cancer, it is not the tobacco-associated lesions that have the highest risk of malignant transformation, but those which appear to be idiopathic.6
It is surprising to many clinicians that the morbidity rate for oral cancer is higher than Hodgkin’s disease, malignant melanoma, and cancers of the cervix, testicles, brain, kidney, and ovaries. This is why it is often referred to as the “forgotten disease.” The relative 5-year rate for patients who are diagnosed with localized (early stage) oral cancer is 82%, but drops to 51% for patients with regional disease and 28% for patients whose cancer has metastasized to distant locations by the time of diagnosis. Note that “early diagnosis” is defined as stage I or stage II cancers, or “cases in which the tumor does not exceed four centimeters in the largest diameter and has not spread to adjacent structure or tissue, and in which there has been no metastasis to regional cervical lymph nodes or to other organs.”5 Unfortunately, as cited in Oral Health in America: A Report of the Surgeon General, 70% of oral cancer is diagnosed in the late stages at the present time.7
The National Institute of Dental and Craniofacial Research (NIDCR), part of the National Institutes of Health (NIH), has reported success using the VELscope System—with its customized optical device that allows clinicians to visualize in a completely new way whether a patient might have a developing oral cancer instead of a relatively benign condition at the early stages of the disease process.8 Like other cancers, this is extremely important as early diagnosis is essential because survival rates are significantly higher when the cancer is diagnosed at its earliest and precancerous stages.
This simple hand-held device emits a cone of a narrow band of blue light onto the mucosa of the oral cavity and excites the various molecules (fluorophores) within the cells. The technology allows the clinician to examine the mucosa by looking through its viewing handpiece with its specialized optical filters and observing changes in tissue fluorescent properties, which occurs at a cellular level. Remove the light, and the normal tissue fluorescence and its patterns are no longer visible to the human eye. This is because of the full-spectrum light stimulating the tissue and overshadowing the natural fluorescence and then compounding the absence of the required discriminating filtration.
This non-invasive but robust field-of-view technology enables the direct visualization of fluorescence in the context of surrounding normal tissue. Normal healthy cells absorb the light energy and re-emit a pale, apple-green, while potentially early tumors and dysplastic or cancerous cells appear very dark maroon to black.
One pilot study showed the efficacy of FV in detecting abnormal tissue by using histology as the gold standard to verify its findings. In this study, the device achieved a sensitivity and specificity of well over 95% when discriminating normal mucosa from severe dysplasia, carcinoma in situ, or invasive squamous cell carcinoma.9
This direct optical FV technology works without the need to use any rinses or stains during the screening, which helps to simplify the examination process. This allows easy integration into routine hygiene and examination appointments without interacting with antimicrobial medications, prophy pastes, fluoride applications, or interfering with other desired patient treatment or care. The absence of mouthrinses and stains means there is no patient discomfort or other objections caused by the taste of a mouthrinse or the effects and mess of a staining process that may linger for a prolonged period of time.
Conclusion
The discovery and diagnosis of oral cancer is the responsibility of dentists and the dental profession. We as dental professionals not only have this responsibility but also the obligation to our patients to examine and evaluate them for all oral diseases using the best diagnostic modalities available. The VELscope and its direct optical FV is rapidly becoming the standard of care in oral mucosal screening and evaluation. We are the gatekeepers of our patients’ oral health and are the health care specialists of the mouth. Routine, thorough oral mucosal exams will save lives.
REFERENCES
1. Pogerl MA. The dentist and oral cancer in North East Scotland. Br Dent J. 1974;137(1):15-20.
2. Coffin F. Cancer and the dental surgeon. Br Dent J. 1964;161: 199-202.
3. Lydiatt DD. Cancer of the oral cavity and medical malpractice. Laryngoscope. 2002;112(5):816-819.
4. Silverman S Jr. Demographics and occurrence of oral and pharyngeal cancers: The outcomes, the trends, the challenge. J Am Dent Assoc. 2001;132(Nov.): 7S-11S.
5. American Cancer Society. Cancer Facts and Figures 2006. Atlanta, Ga: 2006.
6. Oral Cancer, 2003. Shah, Johnson, & Batsakis (eds). Martin Duntiz, publisher.
7. Oral Health in America: A Report of the Surgeon General, 2000. Washington, DC: US Dept of Health and Human Services.
8. “Novel Device Shows Great Potential in Detecting Oral Cancer.” Washington, DC: National Institutes of Health. NIH News, April 10, 2006. Availalble at: https://www.nih.gov/news/pr/apr2006/nidcr-10.htm. Accessed June 8, 2006.
9. Lane PM, Gilhuly T, Whitehead P, et al. Simple device for the direct visualization of oral cavity tissue fluorescence. J Biomed Opt. Mar/Apr 2006,11(2).