Purposeful Exposure of a Polylactic Acid Barrier to Achieve Socket Preservation for Placement of Dental Implants: Case Series Report
Paul S. Rosen, DMD, MS; and Adam D. Rosen
Abstract:
This retrospective case series reports on the use of a polylactic acid barrier that was left exposed in the process of socket preparation for the placement of dental implants. A retrospective chart review found 43 patients with 48 extraction sockets that were treated in this manner. Teeth were removed and the sockets were thoroughly debrided, with 40 of them receiving a bone replacement graft covered by the polylactic acid barrier and the additional 8 receiving the membrane alone. Suturing left the barrier exposed, and the sites were re-entered on average at 23 weeks for the placement of a dental implant. All sites were able to receive a dental implant, demonstrating the ability to leave a polylactic acid barrier exposed and achieve successful guided bone regeneration (GBR) results. This ultimately helped avoid some of the negative sequelae of trying to achieve primary closure of the flaps at the time of tooth extraction.
The high success rate of dental implants has revolutionized the way clinicians treat patients today. Now more than ever, careful planning is given to how teeth should be extracted and their remaining sockets managed to enable enough bone volume for the placement of dental implants. Without appropriate forethought, the alveolar housing may assume a more palatal or lingual position, potentially impacting the esthetics, phonetics, and function for the implant that will be placed at the extraction site.
The key processes of tissue modeling and remodeling after tooth extraction have been well documented in both animals1 and humans.2,3 Horizontal buccal bone resorption has been shown to reach as high as 56% while lingual bone resorption has been reported up to 30%,4 and the overall reduction in width of the horizontal ridge has been seen to reach up to 50% following tooth extraction.5 Statistically, most hard-tissue loss occurs during the first month after removal of the tooth and can average up to 3 mm to 5 mm in width by 6 months. To limit these adverse physiologic sequelae following the removal of teeth, the placement of a graft material into a socket has been one proposed method to try to preserve the natural tissue contours at the extraction site, and prepare for the possible reconstruction with an implant-supported prosthesis.6 Moreover, combining the bone replacement graft with a barrier membrane material, or in some instances using a barrier material alone, has also been advocated to help stabilize the clot at the site of extraction and provide for optimal healing.7,8
Over the past several years, a number of meta-analyses have looked at socket preservation techniques.9-11 The scientific evidence from these systematic reviews appears to suggest that socket preservation techniques are of some benefit to reduce the amount of horizontal dimensional loss of the alveolar ridge. Whether this is consequential for the placement of a dental implant using a delayed approach, whether the results to such care hold up long term, whether this becomes more critical as the buccal plate becomes thinner, and which technique and/or material might provide the best results are still areas that require clarification. However, if bone width is crucial, then providing some type of treatment to the socket area—be that graft plus barrier or barrier alone—may be important.
A polylactic acid barrier material (Guidor®, Sunstar Americas, Inc., www.guidor.com) has recently been reintroduced to facilitate regeneration around teeth, dental implants, and socket areas. This device meets the criteria for an optimal barrier material as set forward by Scantlebury et al,12 which include tissue integration, cell-occlusivity, clinical manageability, space maintenance, and biocompatibility. Its design encompasses a double-layered matrix with two differently perforated layers, which allows for the exchange of fluids between the flap and underlying healing site, containment of any bone replacement graft material that may have been placed into the site, as well as tissue exclusion as the barrier allows for ingrowth into it while retarding tissue penetration into the healing site beneath it. Since the barrier is resorbed, the healing site need not be disturbed for its removal, sparing the patient a possible additional surgical procedure and potentially disturbing the developing clot. To maximize the site’s potential for healing, this membrane is supposed to be completely covered by the overlying flap throughout the postsurgical healing period. While this approach on its surface appears ideal, many circumstances would suggest otherwise. For example, if the extraction is located near a structure such as the mental nerve, achieving primary closure through coronal flap advancement may predispose the patient to temporary or permanent paresthesia or anesthesia. If the barrier could be left exposed to avoid such morbidities and maintain successful outcomes, this would be quite appealing for patient care. For this reason, this retrospective case series is being reported where a Guidor® membrane was purposefully left exposed at sites of tooth extraction for socket preservation. These sites were subsequently exposed at the time of implant placement where this approach could be evaluated for its success.
Case Series Treatment
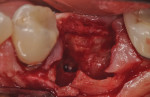
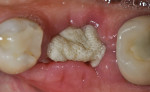
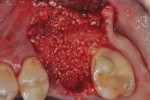
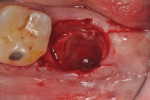
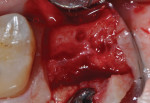
The patients were referred for the placement of dental implants to a clinical private practice limited to periodontics and dental implants. Informed consent was obtained prior to treatment. Anesthesia was obtained with local infiltration using articaine 4% with 1:100,000 epinephrine, except in those patients where this could not be tolerated and carbocaine 2% with 1:20,000 levonordefrin was used. Sulcular incisions were performed around the tooth to be extracted and were minimally extended to the adjacent teeth to mobilize the flap. Teeth were extracted using elevators and extraction forceps. In situations that were more challenging, a piezoelectric handpiece (Piezotome 2™, ACTEON North America, www.acteongroup.com) or dental high-speed handpiece were used to free up the root that was in the socket. Upon the tooth’s removal, the socket was curetted thoroughly to remove any residual soft tissue, and the socket was inspected to see whether it was intact (Figure 1). If the buccal or lingual plates were lost in any way, the flap was extended to provide better access to and visualization of these areas. The sockets were irrigated with povidone iodine for approximately 1 minute to further reduce any residual bacterial contamination of the site and then rinsed of this agent by copious use of sterile water.
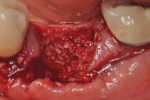
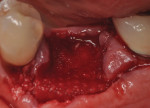
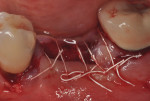
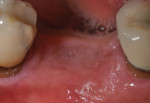
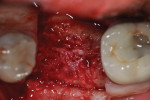
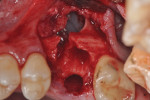
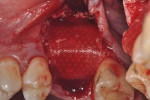
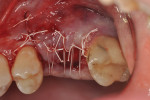
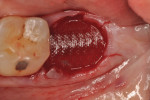
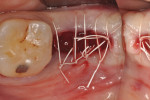
Each socket was treated in one of two manners. If the walls of the socket were determined to be thick and intact, with the diameter not larger than 7 mm to 8 mm, a barrier alone was trimmed to extend 3 mm beyond the socket and placed over the site. If the walls were thinner or if dehiscence or fenestrations were present at the socket, then a bone replacement graft of freeze-dried bone allograft (FDBA) (OraGraft®, LifeNet Health, Inc., www.accesslifenethealth.org) that was hydrated with either sterile water or recombinant platelet-derived growth factor-BB (rh-PDGF-BB) (Osteohealth, www.osteohealth.com) (Figure 2) was placed into the socket and was condensed with light incremental pressure to fill the socket. In two instances a bovine-derived xenograft (BioOss®, Osteohealth) was used mixed with rh-PDGF-BB, as a concurrent internal sinus lift was performed. The polylactic barrier was trimmed to fit the site (Figure 3) so that it also covered the areas where dehiscence or fenestrations were present in the buccal or lingual plate. Flaps were replaced back over the edges of the membrane without purposefully advancing them to achieve primary coverage. The site was sutured with 6-0 expanded polytetrafluoroethylene (ePTFE) (Gore-Tex®, W.L. Gore & Associates, www.gore.com) (Figure 4). For infection control, patients were prescribed amoxicillin 875 mg to be taken twice daily for 7 days, along with topically applying chlorhexidine 0.12% mouthrinse with a cotton swab to the site twice daily for 1 month. If a patient was allergic to amoxicillin, they were prescribed either clindamycin 150 mg four times daily for 7 days, or azithromycin 250 mg with two pills taken on the first day and then one per day for an additional 4 days. Ibuprofen 600 mg or acetominophen with codeine were used for analgesia. Patients were told to refrain from chewing on the site to avoid dislodging the barrier.
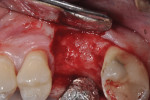
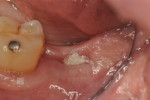
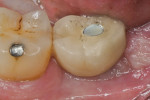
Patients returned at approximately 10 to 21 days, at which time the barrier was inspected to verify that it was intact, and sutures were removed if the site appeared to have adequately healed (Figure 5). Topical chlorhexidine was applied to debride the area of plaque. Postoperative instructions of not eating on the site were reinforced, and the patients were then seen at 3 to 4 weeks for further postoperative site inspection and debridement. If the sites were healing uneventfully, patients were then seen at 2 months and a determination of when implant placement would occur was made. Typically, if healing was uneventful, the site was re-entered for implant placement at 3 to 4 months (Figure 6 and Figure 7); a 6-month waiting period was pursued for those who had more advanced bone loss (Figure 8 through Figure 12).
The implant surgery was performed in a manner similar to tooth extraction. Local anesthesia was obtained and full-thickness flaps were elevated to inspect the areas and place a dental implant. Success was determined by whether adequate bone existed to place a dental implant of adequate length and width without further regenerative efforts being necessary for that placement. The bone quality type (I-IV) formed at the site was also determined at the time of site preparation for implant placement.
Results
This article reports on patients who were treated with socket preservation efforts to facilitate dental implant placements by a delayed approach. The information was obtained after an exhaustive chart review performed by one of the authors (ADR). A total of 43 consecutively treated patients were identified, which included 22 males and 21 females. Their ages ranged from 28-75 years (average 54.4). There were 39 nonsmokers and 4 smokers who indicated less than one pack per day usage. Their medical histories were unremarkable except for: three patients reporting a history of Type 2 diabetes, which was determined from their hemoglobin A1C levels to be well controlled; three with bone metabolic diseases (two with osteoporosis and one with osteopenia based upon Dexascan analysis); and four with an allergy to penicillin medications. A total of 48 extraction sites were treated that consisted of 23 maxillary molars (18 first molars/5 second molars), 3 maxillary premolars, and 22 mandibular molars (14 first molars/8 second molars). The reasons for treating these sites with a delayed approach were the inability to place an immediate implant with either a favorable position for its restoration, or the lack of adequate bone to allow for primary stability to be obtained. Bone inadequacy for anchorage stemmed from an absence of furcation bone, the size of the socket being too large, or severe loss/absence of the labial plate (11 instances), all of which precluded dental implant placement in these particular patients, in the opinion of the treating surgeon. Nine sites were treated with a barrier only, as the walls were quite thick and the diameter allowed for the barrier to be supported without need for the bone replacement graft (Figure 13 through Figure 18). All of the sites treated were able to receive an implant, as bone dimensions were adequate both in buccal-lingual/palatal and occlusal-apical directions. Table 1 summarizes the type of bone regenerated based upon clinical feel when preparing the osteotomies with the handpiece. Four of the sites had type I bone, 34 sites had type II, and 10 had type III.
A total of four adverse events were reported, none of which prohibited an implant being placed at the time of re-entry. In the context of overall sockets treated, this rate was less than 10% and in only one instance could this possibly be attributed to the barrier, bringing this figure to 2.1%. These events included deficiency on the labial at one site, a trifurcated molar having one of the sockets heal with limited bone, a labial piece of root being left that needed removal, and the loss of the barrier at 1 to 2 weeks. None of these events precluded proper implant placement, and only the former three required some added grafting. Moreover, when looking at time to barrier loss, the average was 4.8 weeks with a minimum of 1.5 weeks and a maximum of 8 weeks.
Discussion
Socket maintenance techniques have become a growing area of interest that has paralleled the need for and success of dental implant placements in everyday clinical practice. As teeth are deemed untreatable, many patients seek their replacement with implant-supported restorations. To optimize ridge volume for placing implants in three dimensions, clinicians have traditionally used barrier membranes either alone or in combination with a graft. Historically, these barrier membranes have been submerged when used and have stemmed from the early work with expanded polytetrafluorethylene (ePTFE), which had a tendency to become infected if the barrier became prematurely exposed.13 To avoid this, techniques like coating the ePTFE barriers with tetracycline were evaluated.14 Over time, polymer and collagen barriers were developed to avoid both this adverse scenario and also the need for membrane removal, which could potentially disturb the developing clot. Due to their composition and engineering, the need for primary closure was critical in order to avoid premature degradation of these membranes, which could lead to suboptimal results.15 The negatives to advancing a flap to gain primary closure include: the added morbidities of increased pain and swelling; altering the amount of keratinized tissue; altering soft-tissue landmarks, which could lead to diminished esthetics; or, quite possibly, causing anesthesia/paresthesia if near a vital structure such as the mental foramen. Studies by Cardaropoli and Cardaropoli16 and Engler-Hamm et al17 have demonstrated that exposure of bioabsorbable collagen, in the former study, and leaving a polylactic acid barrier exposed, in the latter, can still lead to successful outcomes. Moreover, Zubillaga et al18 found that despite their efforts to gain primary coverage with a polylactic acid barrier for ridge preservation, they still had exposure 45% of the time afterwards.
The results of this retrospective case series appear promising, because all the sites treated were able to successfully receive dental implants at the time of the second surgery. While critics may question the lack of numeric quantification in this case series, the sine qua non for the clinician is reaching the target goal, which in this case is preserving proper hard-tissue dimensions to allow for an implant to be appropriately placed with a low rate of complications. Other alveolar reconstructive studies that have provided quantitative information have discussed “successful” results while providing no clear definition for what “success” means.19 Moreover, meta-analyses of the available literature have demonstrated that ridge preservation techniques fail to completely stop ridge resorption.10,11 As such, the goal should be to limit resorption in an effort to place implants as optimally as possible.
The primary concerns with leaving a barrier exposed relate to the potential for site infection and premature barrier loss, which will lead to diminished success. The avoidance of site infection has been closely related to stringent infection control during healing using topical and/or systemic antimicrobials.14,20,21 In the current case series, there were no instances of infection at the extraction sites. Whether this could have been achieved by using the povidone iodine irrigation, the systemic antibiotic, or the topical application of the chlorhexidine alone without the other measures is unknown and would have to be determined through a controlled study. Currently, these measures have been employed by the surgeon in this report with great success. Regarding the premature loss of the barrier and diminished benefits, it appears that a period of approximately 3 to 4 weeks of an intact barrier for socket preservation efforts seems appropriate to facilitate adequate graft containment and clot stability to provide for favorable outcomes.
Conclusion
In summary, this case series from a clinical private practice adds to the growing body of evidence that resorbable barriers do not require their coverage to achieve favorable clinical outcomes at extraction sockets. The merits of limiting the advancement of a flap to achieve primary coverage of a barrier appear warranted given the potential for increasing the morbidity of pain and swelling at the site of an extraction.
References
1. Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
2. Pietrokovski J, Massler M. Alveolar ridge resorption following tooth extraction. J Prosthet Dent. 1967;17(1):21-27.
3. Amler MH, Johnson PL, Salman I. Histological and histochemical investigation of human alveolar socket healing in undisturbed extraction wounds. J Am Dent Assoc. 1960;61:32-44.
4. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
5. Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23(4):313-323.
6. Tarnow DP, Eskow RN, Zamzok J. Aesthetics and implant dentistry. Periodontol 2000. 1996;11:85-94.
7. Nevins M, Mellonig JT. Enhancement of the damaged edentulous ridge to receive dental implants: a combination of allograft and the Gore-Tex membrane. Int J Periodontics Restorative Dent. 1992;12(2):96-111.
8. Lekovic V, Kenney EB, Weinlaender M, et al. A bone regenerative approach to alveolar ridge maintenance following tooth extraction. Report of 10 cases. J Periodontol. 1997;68(6):563-570.
9. Ten Heggeler JM, Slot DE, Van der Weijden GA. Effect of socket preservation therapies following tooth extraction in non-molar regions in humans: a systematic review. Clin Oral Implants Res. 2011;22(8):779-788.
10. Vignoletti F, Matesanz P, Rodrigo D, et al. Surgical protocols for ridge preservation after tooth extraction. A systematic review. Clin Oral Implants Res. 2012;23(suppl 5):22-38.
11. Morjaria KR, Wilson R, Palmer RM. Bone Healing after Tooth Extraction with or without an Intervention: A Systematic Review of Randomized Controlled Trials. Clin Implant Dent Relat Res. 2012;Mar 8. doi:10.1111/j.1708-8208.2012.00450.x.
12. Scantlebury TV. 1982-1992: a decade of technology development for guided tissue regeneration. J Periodontol. 1993;64(11 suppl):1129-1137.
13. Machtei EE. The effect of membrane exposure on the outcome of regenerative procedures in humans: a meta-analysis. J Periodontol. 2001;72(4):512-516.
14. Zarkesh N, Nowzari H, Morrison JL, Slots J. Tetracycline-coated polytetrafluoroethylene barrier membranes in the treatment of intraosseous periodontal lesions. J Periodontol. 1999;70(9);1008-1016.
15. Nowzari H,London R, Slots J. The importance of periodontal pathogens in guided periodontal tissue regeneration and guided bone regeneration. Compend Contin Educ Dent. 1995;16(10):1042-1046.
16. Cardaropoli D, Cardaropoli G. Preservation of the postextraction alveolar ridge: A clinical and histologic study. Int J Periodontics Restorative Dent. 2008;28(5):469-477.
17. Engler-Hamm D, Cheung WS, Yen A, et al. Ridge preservation using a composite bone graft and a bioabsorbable membrane with and without primary wound closure: a comparative clinical trial. J Periodontol. 2011;82(3):377-387.
18. Zubillaga G, Von Hagen S, Simon BI, Deasy MJ. Changes in alveolar bone height and width following post-extraction ridge augmentation using a fixed bioabsorbable membrane and demineralized freeze-dried bone osteoinductive graft. J Periodontol. 2003;74(7):965-975.
19. Miyamoto I, Funaki K, Yamauchi K, et al. Alveolar ridge reconstruction with titanium mesh and autogenous particulate bone graft: computed tomography-based evaluations of augmented bone quality and quantity. Clin Implant Dent Relat Res. 2012;14(2):304-311.
20. Machtei EE, Oettinger-Barak O, Peleg M. Guided tissue regeneration in smokers: effect of aggressive anti-infective therapy in Class II furcation defects. J Periodontol. 2003;74(5):579-584.
21. Schallhorn RG, McClain PK. Combined osseous composite grafting, root conditioning, and guided tissue regeneration. Int J Periodontics Restorative Dent. 1988;8(4):8-31.
Disclosure
Paul S. Rosen, DMD, MS has received fees for lecturing on Guidor.
About the Authors
Paul S. Rosen, DMD, MS
Clinical Associate Professor of Periodontics
University of Maryland Dental School
Baltimore, Maryland;
Private Practice
Yardley, Pennsylvania
Adam D. Rosen
Dental Assistant
Private Practice
Yardley, Pennsylvania