Bisphosphonate-Related Osteonecrosis of the Jaws—A Case Report
Abstract
Bisphosphonates are commonly prescribed for the treatment of osteoporosis and bone cancers. These drugs come in both intravenous and oral forms. Because these drugs have become more prevalent, there have been increased reports of bisphosphonate-related osteonecrosis of the jaws (BRONJ). This case presentation focuses on a 50-year-old woman with a 10-year history of monthly Zometa (zoledronic acid) use for treatment of multiple myeloma, resulting in BRONJ with a very unfavorable outcome. Its purpose is to depict how the chronic use of intravenous (IV) bisphosphonates can lead to bisphosphonate-related osteonecrosis of the jaw (BRONJ), and to present treatment options, including conservative measures, the dentist may be able to employ in the early phases of the disease, along with the indications for jaw resection. Osteonecrosis of the jaws in association with bisphosphonate therapy is a very serious condition with which all dentists should be familiar. When the osteonecrosis begins, it can be very painful to the patient, difficult to treat, and can lead to very deleterious sequelae.
Bisphosphonates suppress bone turnover by disrupting osteoclastic signal transduction, maturation, and longevity, thus interfering with bone remodeling. The overall effect is a decrease in bone turnover and inhibition of the bone’s reparative ability. It has been hypothesized that suppressed turnover can impair oral wound healing, which, in some cases, leads to bisphosphonate-related osteonecrosis of the jaws (BRONJ), an osteopetrosis-like jaw necrosis.1
Intravenous (IV) bisphosphonates are primarily used and are effective in the management of certain cancer-related conditions. These include hypercalcemia of malignancy, skeletal-related events associated with bone metastases in the context of solid tumors and in the management of lytic lesions in the setting of multiple myeloma. IV bisphosphonates are effective in preventing and reducing hypercalcemia, stabilizing bony pathology, and preventing pathologic fractures in cases of skeletal involvement. While they have not been shown to improve cancer-specific survival, they have had a significant improvement on the quality of life for patients with advanced cancer that involves the skeletal system.
ZOMETA® (zoledronic acid injection) (Novartis Oncology, www.zometa.com) is an IV bisphosphonate used in the treatment of hypercalcemia of malignancy and to reduce and delay bone complications due to multiple myeloma and bone metastases from solid tumors. The manufacturer of Zometa has issued a box warning that states:
"Osteonecrosis of the jaw (ONJ) has been reported mainly in cancer patients treated with intravenous bisphosphonates, including ZOMETA.
* If you have advanced breast cancer or a type of cancer called multiple myeloma, or if you have had dental extraction, periodontal disease, local trauma, including poorly fitting dentures, you may be at greater risk of getting ONJ.
* You should maintain good oral hygiene and have a dental examination with preventive dentistry prior to beginning ZOMETA. While on treatment, avoid invasive dental procedures, if possible, as recovery may take longer.”
Case Report
Dental and Medical History
A 50-year-old woman was referred to the oral and maxillofacial surgery clinic in January 2010 for evaluation of mouth sores and draining facial fistulas during her inpatient admission for schizoaffective disorder. Her history of the present dental illness revealed that she had an extraction of a left mandibular molar approximately 3 years previously and that soon thereafter she noticed pieces of bone extruding from the extraction site, with no pain and poor healing of the socket. One year after the extraction she sought treatment at another institution for the non-healing socket. A bone biopsy was done, but she did not follow-up with her appointments.
Her past medical history was significant for multiple myeloma diagnosed in 2000 and which had been treated with monthly doses of Zometa for 10 years, with the last Zometa treatment in October 2009. She also had a history of schizoaffective disorder, hypertension, and asthma. Her medications at the time of presentation were: metoprolol, albuterol, and divalproex sodium delayed-release tablets (Depakote, Abbott Laboratories, www.abbott.com).
Diagnostic Findings
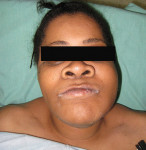
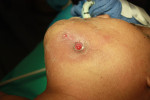
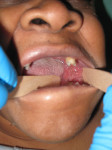
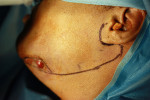
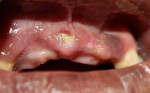
On the initial clinical examination, she appeared to be a well-nourished African-American woman in mild distress. There was moderate edema of the left face (Figure 1), which was indurated, and there were two actively draining orocutaneous fistulas located in the vicinity of the inferior border of the left mandible (Figure 2). Intraorally, the patient was partially edentulous with exposed bone in the left posterior mandible with purulent discharge on palpation (Figure 3).
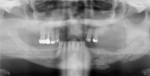
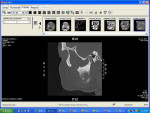
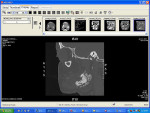
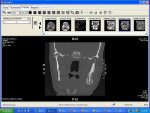
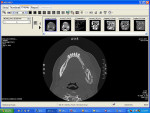
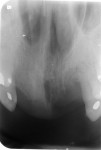
Sensory testing of the lip revealed profound left-sided V3 parasthesia. Radiographic examination showed sclerotic changes in the horizontal ramus of the left mandible. Serial panorex revealed rapid destruction with pathologic fractures appearing within a matter of 6 weeks (Figure 4 and Figure 5). A maxillofacial CT scan revealed extensive, progressive destruction and also revealed a pathologic fracture at the left angle of the mandible (Figure 6 through Figure 9).
Hospital Course
It was decided to transfer the patient to the medical floor for management by the infectious disease service in conjunction with the oral and maxillofacial surgery service.
Diagnostic Tests: At the initial clinical visit, the fistula was cultured, the specimen was submitted for cytology, a fine needle aspiration (FNA) was performed, and pieces of bone were sent for histopathology to rule out multiple myeloma lesions. The cultures revealed moderate growth of Streptococcus viridans, gram-negative rods, and gram-positive cocci. Cytology revealed inflammatory cells, with predominant neutrophils. The FNA revealed inflammatory cells—predominantly atypical lymphocytes and neutrophils—and also was positive for actinomyces. The bone scan demonstrated increased bone activity involving the ascending ramus and angle of the left mandible with less intense irregular bone activity along the anterior aspect of the body of the mandible.
Diagnosis: The authors’ impression was that it was probable partially treated or chronic osteomyelitis involving the left mandible, a diagnosis that was supported by a follow-up gallium scan, which demonstrated intense gallium uptake involving the ascending ramus with less intense activity in the anterior third of the body of the mandible. A diagnosis of BRONJ/osteomyelitis was thus made.
Treatment/Outcome: The infectious disease department recommendation was for IV penicillin G (PenG) for 6 weeks. The patient, however, did not respond significantly to the antimicrobial therapy, and there was significant radiologic evidence of continuous osteonecrosis. It was decided to do a surgical resection of the diseased bone with immediate reconstruction with a bone plate.
Surgical Procedure
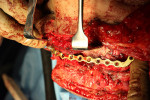
Due to the progressive nature of the osteonecrosis, the extent of the destruction, and the patient’s failure to respond to antibiotic therapy, the decision was made for her to undergo surgical resection. The surgical procedure involved a left hemimandibulectomy with reconstruction plate placement via preauricular and submandibular approaches (Figure 10 through Figure 14).
Discussion
As BRONJ is a fairly new entity in the dental and medical literature, the authors used the American Association of Oral and Maxillofacial Surgeons (AAOMS) definition of BRONJ to make their diagnosis.2
Patients may be considered to have BRONJ if all of the following three characteristics are present:
1. Current or previous treatment with a bisphosphonate;
2. Exposed bone in the maxillofacial region that has persisted for more than 8 weeks; and
3. No history of radiation therapy to the jaws.
Pathogenesis
BRONJ is a condition found in patients who have received IV bisphosphonate therapy for various bone-related conditions. Its incidence is 0.8% to 12% with IV bisphosphonates, and 0.01% to 0.04% with oral bisphosphonates.3 BRONJ manifests as exposed, nonvital bone involving the maxillofacial structures. BRONJ is thought to be caused by trauma to dentoalveolar structures that have a limited capacity for bone healing due to the effects of bisphosphonate therapy. That there is relatively greater bone turnover in jaws explains why there is greater bisphosphonate deposition in the jaws and an increased risk of BRONJ there versus extragnathic sites. Other criteria for diagnosing BRONJ: there may be asymptomatic or painful exposed bone on the jaws, with a 2:1 incidence in the mandible versus the maxilla, and it can be spontaneous or at the site of trauma. BRONJ may become secondarily infected, have soft-tissue swelling, and cause loosening of teeth.
Microbiology
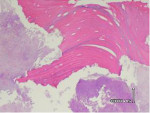
On microscopic examination, specimens typically show areas of partially or completely necrotic bone with debris and fibrinous exudates accompanied by an inflammatory infiltrate formed by neutrophils, histiocytes, eosinophils, and plasma cells. The non-vital bone exhibits loss of osteocytes from the lacunae, peripheral resorption, and bacterial colonization. Scattered sequestra and pockets of abscess formation are also commonly seen. Superficial sequestrum almost always shows necrotic bone surrounded by many bacterial colonies; utilizing special stains such as Periodic Acid-Schiff (PAS) and gram-staining can be useful to further confirm Actinomyces colonies. Studies have shown a strikingly high presence of Actinomyces in necrotic bone areas and considered these pathogens to be involved in the chronic, non-healing process of BRONJ.4 Actinomyces are commonly present as commensals in the oral cavity; thus their presence within the necrotic lesions is likely not an etiological factor in the pathogenesis of BRONJ, but rather a secondary infection of the necrotic tissues.
Treatment Suggestions
There are various methodologies—both surgical and nonsurgical—used in the management of BRONJ. Nonsurgical management of BRONJ may consist of the following and is based on the stage the patient is in:
- Antimicrobial rinses
- Systemic antibiotics
- Systemic or topical antifungals
- Discontinuation of bisphosphonate therapy
- No dental therapy or minimally invasive dental therapy (eg, root canal therapy instead of extraction)
- Hyperbaric oxygen (HBO) therapy: still undergoing clinical trials and research to establish efficacy
Surgical intervention for BRONJ remains limited because of the impaired ability of the bone to heal. Because no long-term or controlled studies on the management of BRONJ have been published, the article from AAOMS, which is based on the consensus of a panel discussion, is the best available guide to therapy.
In order to direct rational treatment guidelines and collect data to assess the prognosis in patients who have used either IV or oral bisphosphonates, the AAOMS proposes use of the following staging categories and their respective treatments.
- BRONJ Stage 0: Nonspecific findings: dull aching bone pain in the body of the mandible, odontalgia with no odontogenic cause, sinus pain, periodontally involved teeth not explained by chronic periodontitis, and periapical/periodontal fistula not associated with pulpal caries. Treatment: symptomatic treatment, with conservative management of local factors, including chronic pain management and possible antibiotic therapy.
- BRONJ Stage I: Findings: exposed/necrotic bone, asymptomatic, no evidence of infection. Treatment: antimicrobial rinses and no surgical intervention.
- BRONJ Stage II: Findings: exposed/necrotic bone, pain, erythema, +/- purulent drainage. Treatment: antimicrobial rinses, systemic antibiotics or antifungals, analgesics.
- BRONJ Stage III: Findings: exposed/necrotic bone, pain/infection, pathologic fracture, extraoral fistula, osteolysis to inferior border. Treatment: antimicrobial rinses, systemic antibiotics or antifungals, analgesics, surgical debridement or resection.
Patient Staging and Treatment
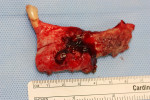
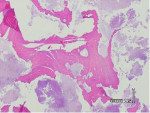
The patient described was classified to be in Stage III (exposed/necrotic bone, pain/infection, pathologic fracture, extraoral fistula, osteolysis to inferior border) of BRONJ. Histology revealed acute osteomyelitis showing necrotic bone and acute inflammatory infiltrate and acute and chronic inflammation of the soft tissue (Figure 15 and Figure 16). Therefore, the plan was to do a surgical resection of the diseased bone with immediate reconstruction with a bone plate.
Postoperative panorex revealed reconstruction plate in place status post hemimandibulectomy (Figure 17). At 8 weeks post-surgical resection, the patient was doing well. She completed her 6 weeks of IV PenG and was put on an oral regimen of PenVK 500 mg every 6 hours for 6 months as per infectious disease protocol, and was maintained with chlorhexidine rinses three times daily.5 The patient was meticulously followed for evidence of new bony exposures, and two new exposures surfaced in the anterior and left maxillary regions (Figure 18 and Figure 19).
Indications for Jaw Resection
Surgical resection is indicated for patients with Stage III BRONJ.6 These patients benefit from resection and debridement in combination with antibiotic therapy, which may offer long-term palliation with resolution of acute infection and pain. It is imperative to maintain careful handling of soft tissues to help in the healing of soft tissues; bone resection should be performed as atraumatically as possible, with smoothing out of rough edges; and, lastly, a tension-free wound closure should be maintained to enhance soft-tissue healing.
Conclusion
In conclusion, IV and oral bisphosphonate use is on the rise. Therefore, to prevent BRONJ, it is imperative to stress prevention protocols to healthcare practitioners. Practitioners may stress the importance of obtaining comprehensive oral/dental examination, prophylaxis, and treatment prior to starting bisphosphonate treatment. The patient should understand that maintaining good oral/dental health is extremely important. The risk for invasive procedures should be based on dose/duration of bisphosphonate use. Invasive procedures such as exodontia, deep scaling and root planning, periodontal surgery, surgical endodontics, and dental implants are to be avoided if possible.
References
1. Freiberger J. Utility of hyperbaric oxygen in treatment of bisphosphonate-related osteonecrosis of the jaws. J Oral Maxillofac Surg. 2009;67(5 Suppl);96-106.
2. American Association of Oral and Maxillofacial Surgeons Position Paper on Bisphosphonate-Related Osteonecrosis of the Jaw—2009 Update.
3. Mavrokokki T, Cheng A, Stein B, Goss A. Nature and frequency of bisphosphonate-associated osteonecrosis of the jaws in Australia. J Oral Maxillofac Surg. 2007;65(3):415-423.
4. Ficarra G, Beninati F. Bisphosphonate-related osteonecrosis of the jaws: an update on clinical, pathological and management aspects. Head Neck Pathol. 2007;1(2):132-140.
5. Ruggiero SL, Woo SB. Bisphosphonate-related osteonecrosis of the jaws. Dent Clin North Am. 2008;52:111-128.
6. Carlson ER, Basile JD. The role of surgical resection in the management of bisphosphonate-related osteonecrosis of the jaws. J Oral Maxillofac Surg. 2009;67(5 Suppl):85-95.
About the Authors
Amandip K. Kamoh, DDS
Chief Resident
Division of Oral and Maxillofacial Surgery
Woodhull Hospital
Brooklyn, New York
Orrett Ogle, DDS
Chief
Division of Oral and Maxillofacial Surger
Director of Residency Training Program in Oral and Maxillofacial Surgery
Woodhull Hospital
Brooklyn, New York