Diode Laser Offers Minimal Benefit for Periodontal Therapy
Charles M. Cobb, DDS, MS, PhD; Mark S. Blue, DDS; Nabil E. Beaini, DDS; Michael R. Umaki, DDS; and Keerthana M. Satheesh, DDS, MS
Abstract
Current evidence indicates that use of diode lasers in the treatment of periodontitis—either as a monotherapy or adjunctive to traditional therapy—offers minimal benefit. Further, subgingival application of the diode laser during nonsurgical periodontal therapy can result in undesired outcomes, even when using manufacturer-recommended parameters.
Diode lasers may offer significant biologic benefit with respect to treatment of dentinal hypersensitivity.1-3 In addition, in vitro studies have reported that diode low-level light therapy (LLLT) decreases cell apoptosis and levels of tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β) in cultures of diabetic and hypoxic cells.4 Further, animal studies have shown that LLLT appears to accelerate wound healing through biostimulatory effects that promote fibroblast development and proliferation.5-7
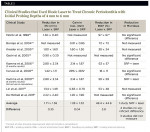
Somewhat in contrast to the beneficial applications noted above, the use of a diode laser in photodynamic therapy (PDT) for treatment of periodontal disease has yielded inconclusive results. Currently, there are 15 published human clinical studies.8-22 The aggregate of studies show little differences between scaling and root planing (SRP) alone versus SRP + PDT with respect to reductions in probing depth, bleeding on probing (BOP), and subgingival bacterial loads, and gains in clinical attachment level (CAL) (Table 1).
Obviously, many questions remain to be answered regarding use of the diode laser as a monotherapy or as an adjunct for the treatment of periodontitis. Indeed, well-designed studies are limited, and the overall quality of the body of evidence is insufficient to support use of lasers for routine periodontal therapy. In fact, one can argue that deriving an evidence-based treatment decision from published studies would be overtly speculative, as the body of evidence is weak and often confusing.23-28
A recent article by Christopher and Blackburn29 discussed how the wavelength of the various diode lasers and the neodymium-doped yttrium aluminium garnet (Nd:YAG) are related to selective destruction of black/brown-pigment–producing bacteria. This frequently stated concept caused the authors of this paper to re-examine the laser-periodontal therapy conundrum. Consequently, they present their views on two issues—both basic to laser periodontal therapy: 1) the benefit of laser subgingival curettage; and 2) efficacy of laser-mediated reductions in subgingival bacterial loads. The authors also present a case with an adverse outcome following subgingival diode laser application.
Subgingival Curettage
Laser-mediated periodontal therapy is based on the perceived benefits of subgingival curettage and a significant decrease in subgingival bacterial load. In addition, two laser manufacturers have declared protocol-specific benefits: 1) one company received FDA 510(K) clearance for its “assisted new attachment procedure” protocol, ie, reducing pocket probing depth by a cementum-mediated periodontal ligament (PDL) new attachment to a previously diseased root surface in the absence of a long junctional epithelium; and 2) the other manufacturer has also received FDA 510(K) clearance for its “laser assisted new attachment procedure” protocol that can regenerate tooth-supporting tissues lost as a result of periodontitis, ie, alveolar bone, cementum, and PDL.
More than 25 years ago, clinical research demonstrated that curettage plus scaling and root planing (SRP) had no advantage over SRP alone.30-35 This view was supported by the 1989 Proceedings of the World Workshop on Clinical Periodontics36 and, again, in the 1993 Proceedings of the 1st European Workshop on Periodontology.37 Indeed, Kalkwarf,36 in the 1989 Workshop, stated that, “Gingival curettage has no justifiable application during the active therapy stages for chronic periodontitis.”
Over the years, subgingival curettage has been accomplished with manual curettes, chemicals,38,39 conservative gingival flap surgery,40,41 and lasers. Interestingly, the excisional new attachment procedure (ENAP) has been described as “curettage with a scalpel,” since a primary goal of this surgery is the removal of pocket epithelium.40,41 Given the overwhelming evidence that subgingival curettage offers little to no benefit over what can be achieved by SRP alone, the authors question the sense of submitting patients to such a procedure, ie, laser-assisted periodontal therapy. Indeed, it should be noted that because of a lack of supporting evidence, the code for curettage has been omitted from the CDT-4 code listing.42
Currently, there are only nine published clinical studies using various diode lasers in the treatment of periodontitis (Table 2 ).43-51 It should be noted that two reports by Kamma et al47,49 can be considered a single study, with 3- and 6-month post-treatment data published at different times. Only data from the 3-month report47 was used in the calculated averages noted in Table 2 of this article. Due to variations in wavelength, parameters, outcome measures, initial pocket probing depths (PD), and lack of examiner blinding and calibration, it is impossible to perform a valid meta-analysis of the nine published clinical studies. However, if one calculates a simple difference between averages on selected clinical parameters, a trend can be established. In this regard, a comparison of SRP + diode laser versus SRP alone is revealing. The average differences in reduction in PD for the nine studies (0.35 mm), gain in CAL (0.04 mm), reduction in BOP (3.8%) (equivalent to 6/168 sites), and reduction in microbial load favors the combined therapy in one study, whereas four studies showed no significant difference between treatment groups, and three studies did not measure microbial reduction.
Reduction in Subgingival Bacterial Load
Lasers, as a group, have inconsistently demonstrated the ability to reduce microorganisms within a periodontal pocket (Table 1).42 Yet marketing statements continue to be made regarding “pocket sterilization” and assertions that diode and Nd:YAG laser wavelengths selectively destroy black-pigment–producing subgingival anaerobic bacteria.29
The dark-pigment–producing bacteria were at one time collectively referred to as BPBs (black- pigment–producing Bacteriodes). Due to advances in DNA and polymerase chain reaction (PCR) technology, many species of oral bacteria that formally belonged to the genus Bacteriodes have been reassigned to the genera of Porphyromonas, Prevotella, and Tannerella, and, of course, Bacteriodes still exists. Colonies of Porphyromonas, Prevotella, and Bacteriodes have been shown to produce pigment when grown on blood agar plates.52
If the possibility of selective eradication of bacteria by diode lasers is considered, those making such claims must also consider that periodontitis is a mixed infection involving at least 30 or more microbial species.52-55 Indeed, it may be that Porphyromonas gingivalis need not be present in large numbers to dictate disease initiation or progression. Recent reports have identified Porphyromonas gingivalis as a “keystone” member of the subgingival microbial biofilm. The “keystone” designation indicates that while this microbe resides in low numbers within the periodontal pocket it still has the ability to dictate host immune cell response.56 Further, it must be considered that, collectively, microbes of the genus Porphyromonas, Prevotella, and Bacteriodes constitute less than 20% of the total subgingival microbial flora.57 The remaining 80% are very capable of initiating a host response leading to periodontal tissue degradation. As so aptly stated by Mombelli et al,58 “Given the large diversity of the microbiota associated with all forms of periodontitis, …the concept of specifically identifying and eradicating particular pathogens may be illusionary.” One might also add, with regards to subgingival laser therapy, that relatively low numbers of highly pathogenic microbes, eg, Porphyromonas gingivalis, make it difficult to totally eradicate such microbes.
Case Report
A 73-year-old female was referred to one of the authors, a periodontist, with a chief complaint of excessive tooth mobility involving the mandibular left second bicuspid (tooth No. 20). The tooth had been mobile for approximately 3 months. The patient was classified as ASA Class II due to the following: baseline blood pressure of 147/85; pulse: 53/minute; allergy to sulfa; and medications that included atorvastatin calcium for high cholesterol and atenolol and ramipril for hypertension.
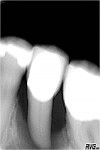
A comprehensive periodontal examination revealed that all probing depths were 2 mm to 4 mm, except tooth No. 20, which exhibited facial measurements of 9 mm, 12 mm, and 9 mm and lingual of 7 mm, 4 mm, and 7 mm. Further, this tooth exhibited BOP and purulent exudate and Class III mobility. It was noted that the gingival biotype was thin and that all mandibular cuspid and premolar teeth exhibited pronounced facial root prominence. A vitality test was not performed as the radiographic examination showed extensive bone loss (Figure 1). Given the clinical presentation and extent of bone loss, tooth extraction was recommended. Restoration of the extraction site required an extensive osseous graft procedure due to the loss of buccal cortical bone.
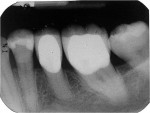
Following extraction, three small dark root surface cavitations and one large area of roughly 2-mm diameter were noted (Figure 2). The referring dentist was contacted regarding previous treatment of tooth No. 20, and it was revealed that his dental hygienist had used a gallium-aluminum-arsenide (GaAlAs) diode laser to perform subgingival curettage and to reduce subgingival bacteria. A copy of the pretreatment radiograph was requested; it showed a normal periodontium with no evidence of bone loss (Figure 3). Tooth No. 20 had a 1:1 crown-to-root ratio and was restored with a full-coverage porcelain-fused-to-metal (PFM) restoration. The referring dentist also noted that the patient was seen on April 19, 2009, at which time the tooth presented a 4-mm to 5-mm facial probing depth. SRP was performed, followed by subgingival treatment with the diode laser. The remainder of the dentition was treated with the diode where required, as the majority of probing depths were within normal range. The patient was seen 3 months later, July 28, 2009. The facial pocket was still in the 4-mm to 5-mm range with BOP and again treated by SRP and diode laser therapy. Subsequent to the July appointment the patient complained of acute tooth pain when chewing. At this point, she was referred to a local periodontist for evaluation and/or treatment.
The extracted tooth was submitted for examination by scanning electron microscopy (SEM) to determine the extent and nature of the cavitational defect (Figure 4). The SEM evaluation revealed a severe, heat-related, irregular-shaped, slightly oval cavitation of the root surface measuring 1.7 mm x 2 mm, which exhibited melting and resolidification of root surface mineral. Given the presence of heat-induced damage and charring, it is hypothesized that the laser burned the root which, in turn, may have resulted in an irreversible pulpal necrosis leading to severe loss of adjacent bone. It is interesting to note that a thin gingival biotype associated with facial root prominence, as evidently was the case with this patient, is often associated with an underlying bony dehiscence. Such a combination of clinical anatomy could explain why the diode laser had easy access to the root surface.
Assuming appropriate laser parameter settings and avoidance of root surface contact, damage from a diode laser is unlikely. In the case presented, the root damage may have resulted from an improper choice of laser parameters or possibly an extreme duration of exposure. However, the authors decided to test another hypothesis using the manufacturer’s recommended settings. It is well known that dark-colored stains or calculus deposits will activate a diode laser. Thus, with each successive pass of the laser, the dark-stained area absorbs increasing amounts of heat which, in turn, leads to surface charring, a darker surface coloration, and yet more heat absorption.
To test the hypothesis, the roots of two freshly extracted teeth—removed because of advanced periodontitis—were marked with a graphite pencil. Four parallel lines were placed, each separated from the adjacent line by 2 mm to 3 mm of root surface (Figure 5). An 810-nm diode laser GaAlAs fitted with a 400-nm optical delivery fiber was used to deliver either 0.5 W, 1 W, 1.5 W, or 2 W of power via a continuous waveform. The fiber was placed parallel to the root surface and moved along the pencil line for one pass using only one of the power settings for each line. The fiber was moved at a rate of roughly 3 mm/sec. Each tooth received the same sequence of laser exposures, after which both specimens were submitted for SEM evaluation.
SEM examination revealed obvious parallel-elongated areas of damage on the root surface that featured heat-induced surface fracturing and crazing (Figure 6). Such areas of damage were present regardless of the laser power setting, ie, 0.5 W through 2 W. The fractured areas of cementum appeared slightly raised from the undamaged adjacent surface. Backscatter SEM imaging revealed extended areas of heat damage featuring well-defined borders. Also noted was a slight gradient in severity of damage that correlated to increased power settings (Figure 7). Another interesting feature was the sharp delineation of an interface between the melted root surface, which was devoid of microbial deposits, and the immediately adjacent areas of undisturbed biofilm (Figure 8 and Figure 9).
Discussion
Testing of the hypothesis in this case might be considered a limited proof-of-principle study, the results of which offer another possible reason for root-surface damage following improper diode laser application within periodontal pockets. The patient’s tooth exhibited not only one larger area of charring and cavitation, but three smaller areas (Figure 2) whose diameters nearly matched that of the laser delivery optical fiber (400 nm) were also noted. If the hypothesis is accepted, it might be argued that the smaller areas also represented activation of the laser due to passage over localized areas of brown/black-stained residual calculus.
It has been known for several years that heat-induced tooth root charring results in formation of surface ammonia,59 cyanate, and cyanamide,60 all of which are incompatible with epithelial and/or connective tissue reattachment to the affected surface.61,62 To gain cellular reattachment requires that the root surface be cleaned of the char layer. In vivo evidence of root surface charring subsequent to a flapless crown-lengthening procedure with an Er:YAG laser has been reported.63 The authors stated that, “Despite great care to avoid laser contact with root surfaces, and despite the end-cutting-only nature of the Er:YAG laser, root-surface damage could not be predictably avoided.”63
The abrupt interface between lased biofilm/calculus and immediately adjacent undisturbed biofilms noted in the SEM examination has been reported previously.64,65 The clinical implication of this sharply marked interface is simply that, in this case, the diode laser energy beam must make direct contact with the biofilm in order to “kill” the microbes. SEM evidence indicates there is no adjacent zone of collateral destruction. This observation probably explains why reduction in subgingival microbial loads following adjunctive use of the diode laser with SRP is no better, on average, than SRP alone (Table 2).
Conclusion
“Lasers are not a magic wand” but do provide advantages depending on the application and goals of treatment.66 When making treatment decisions, clinicians must understand the limitations of nonsurgical laser periodontal therapy (ie, curettage), the limitations of laser application to reduce subgingival bacterial loads, and the potential for adverse clinical outcomes. Further, one must critically evaluate the laser research literature, particularly human clinical trials, and understand the strength of evidence or lack thereof.
References
1. He S, Wang Y, Li X, Hu D. Effectiveness of laser therapy and topical desensitizing agents in treating dentine hypersensitivity: a systematic review. J Oral Rehabil. 2011;38(5):348-358.
2. Yilmaz HG, Kurtulmus-Yilmaz S, Cengiz E, et al. Clinical evaluation of Er,Cr:YSGG and GaAlAs laser therapy for treating dentine hypersensitivity: a randomized controlled clinical trial. J Dent. 2011;39(3):249-254.
3. Pesevska S, Nakova M, Ivanovski K, et al. Dentinal hypersensitivity following scaling and root planing: comparison of low-level laser and topical fluoride treatment. Lasers Med Sci. 2010;25(5):647-650.
4. Sekhejane PR, Houreld NN, Abrahamse H. Irradiation at 636 nm positively affects diabetic wounded and hypoxic cells in vitro. Photomed Laser Surg. 2011;29(8):521-530.
5. Goncalves RV, Novaes RD, Matta SL, et al. Comparative study of the effects of gallium-aluminum-arsenide laser photobiomodulation and healing oil on skin wounds in wistar rats: a histomorphometric study. Photomed Laser Surg. 2010;28(5):597-602.
6. Ezzati A, Bayat M, Khoshvaghti A. Low-level laser therapy with a pulsed infrared laser accelerates second-degree burn healing in rat: a clinical and microbiologic study. Photomed Laser Surg. 2010;28(5):603-611.
7. Bayat M, Azari A, Golmohammadi MG. Effects of 780-nm low-level laser therapy with a pulsed gallium aluminum arsenide laser on the healing of a surgically induced open skin wound in the rat. Photomed Laser Surg. 2010;28(4):465-470.
8. Yilmaz S, Kuru B, Kuru L, et al. Effect of gallium arsenide diode laser on human periodontal disease: a microbiological and clinical study. Lasers Surg Med. 2002;30(1):60-66.
9. Andersen R, Loebel N, Hammond D, Wilson M. Treatment of periodontal disease by photodisinfection compared to scaling and root planing. J Clin Dent. 2007;18(2):34-38.
10. de Oliveira RR, Schwartz-Filho HO, Novaes AB Jr, Taba M Jr. Antimicrobial photodynamic therapy in the non-surgical treatment of aggressive periodontitis: a preliminary randomized controlled clinical study. J Periodontol. 2007;78(6):965-973.
11. Braun A, Dehn C, Krause F, Jepsen S. Short-term clinical effects of adjunctive antimicrobial photodynamic therapy in periodontal treatment: a randomized clinical trial. J Clin Periodontol. 2008;35(10):877-884.
12. Christodoulides N, Nikolidakis D, Chondros P, et al. Photodynamic therapy as an adjunct to non-surgical periodontal treatment: a randomized, controlled clinical trial. J Periodontol. 2008;79(9):1638-1644.
13. Polansky R, Haas M, Heschl A, Wimmer G. Clinical effectiveness of photodynamic therapy in the treatment of periodontitis. J Clin Periodontol. 2009;36(7):575-580.
14. Al-Zahrani MS, Barnshmous SO, Alhassani AA, Al-Sherbini MM. Short-term effects of photodynamic therapy on periodontal status and glycemic control of patients with diabetes. J Periodontol. 2009;80(10):1568-1573.
15. Lulic M, Leiggener GI, Salvi GE, et al. One-year outcomes of repeated adjunctive photodynamic therapy during periodontal maintenance: a proof of principle randomized controlled clinical trial. J Clin Periodontol. 2009;36(8):661-666.
16. Chondros P, Nikolidakis D, Christodoulides N, et al. Photodynamic therapy as adjunct to non-surgical periodontal treatment in patients on periodontal maintenance: a randomized controlled clinical trial. Lasers Med Sci. 2009;24(5):681-688.
17. Rühling A, Fanghanel J, Houshmand M, et al. Photodynamic therapy of persistent pockets in maintenance patients-a clinical study. Clin Oral Invest. 2010;14(6):637-644.
18. Pinheiro SL, Donegá JM, Seabra LM, et al. Capacity of photodynamic therapy for microbial reduction in periodontal pockets. Lasers Med Sci. 2010;25(1):87-91.
19. Sigusch BW, Engelbrecht M, Völpel A, et al. Full-mouth antimicrobial photodynamic therapy in Fusobacterium nucleatum-infected periodontitis patients. J Periodontol. 2010;81(7):975-981.
20. Lui J, Corbet EF, Jin L. Combined photodynamic and low-level laser therapies as an adjunct to nonsurgical treatment of chronic periodontitis. J Periodont Res. 2011;46(1):89-96.
21. Ge L, Shu R, Li Y, et al. Adjunctive effect of photodynamic therapy to scaling and root planing in the treatment of chronic periodontitis. Photomed Laser Surg. 2011;29(1):33-37.
22. Cappuyns I, Cionca N, Wick P, et al. Treatment of residual pockets with photodynamic therapy, diode laser, or deep scaling. A randomized split-mouth controlled clinical study. Lasers Med Sci. 2011;November 22:DOI 10.1007/s10103-011-1027-6.
23. Cobb CM. Lasers in periodontics: a review of the literature. J Periodontol. 2006;77(4):545-564.
24. Karlsson MR, Diogo Löfgren CI, Jansson HM. The effect of laser therapy as an adjunct to non-surgical periodontal treatment in subjects with chronic periodontitis: a systematic review. J Periodontol. 2008;79(11):2021-2028.
25. Schwarz F, Aoki A, Becker J, Sculean A. Laser application in non-surgical periodontal therapy: a systematic review. J Clin Periodontol. 2008;35(suppl 8):29-44.
26. Slot DE, Kranendonk AA, Paraskevas S, van der Weijden F. The effect of a pulsed Nd:YAG laser in non-surgical periodontal therapy. J Periodontol. 2009;80(7):1041-1056.
27. Cobb CM, Low SB, Coluzzi DJ. Lasers in the treatment of chronic periodontitis. Dent Clin North Am. 2010;54(1):35-53.
28. Matthews DC. Seeing the light—the truth about soft tissue lasers and nonsurgical periodontal therapy. J Can Dent Assoc. 2010;76(2):a30-a35.
29. Christopher H, Blackburn PR. It’s not rocket science! (but it is laser physics). RDH. 2011;31(7):66-72.
30. Ainslie PT, Caffessee RG. A biometric evaluation of gingival curettage (I). Quintessence Int Dent Dig. 1981;12(5):519-527.
31. Ainslie PT, Caffessee RG. A biometric evaluation of gingival curettage (II). Quintessence Int Dent Dig. 1981;12(6):609-614.
32. Smith BA, Echeverri M, Caffesse RG. Mucoperiosteal flaps with and without removal of the pocket epithelium. J Periodontol. 1987;58(2):78-85.
33. Echeverria JJ, Caffesse RG. Effects of gingival curettage when performed 1 month after root instrumentation. A biometric evaluation. J Clin Periodontol. 1983;10(3):277-286.
34. Lindhe J, Nyman S. Scaling and granulation tissue removal in periodontal therapy. J Clin Periodontol. 1985;12(5):374-388.
35. Ramfjord SP, Caffesse RG, Morrison EC, et al. 4 modalities of periodontal treatment compared over 5 years. J Clin Periodontol. 1987;14(8):445-452.
36. Kalkwarf KL. Tissue attachment. In: Proceedings of the World Workshop in Clinical Periodontics. Chicago, IL: American Academy of Periodontology; 1989:V1-V19.
37. Kieser JB. Nonsurgical periodontal therapy. In: Lang NP, Karring T, eds. Proceedings of the 1st European Workshop on Periodontology. Hanover Park, IL: Quintessence Publishing Co., Ltd.; 1993:131-158.
38. Forgas LB, Gound S. The effects of antiformin-citric acid chemical curettage on the microbial flora of the periodontal pocket. J Periodontol. 1987;58(3):153-158.
39. Kalkwarf KL, Tussing GJ, Davis MJ. Histologic evaluation of gingival curettage facilitated by sodium hypochlorite solution. J Periodontol. 1982;53(2):63-70.
40. Yukna RA, Lawrence JJ. Gingival surgery for soft tissue new attachment. Dent Clin North Am. 1980;24(4):705-718.
41. Litch JM, O’Leary TJ, Kafrawy AH. Pocket epithelium removal via crestal and subcrestal scalloped internal beveled incisions. J Periodontol. 1984;55(3):142-148.
42. ADA Council on Scientific Affairs. Statement on Lasers in Dentistry. https://www.ada.org/1860.aspx. Updated April 2009. Accessed December 26, 2011.
43. Moritz A, Schoop U, Goharkhay K, et al. Treatment of periodontal pockets with a diode laser. Lasers Surg Med. 1998;22(5):302-311.
44. Borrajo JL, Varela LG, Castro GL, et al. Diode laser (980 nm) as adjunct to scaling and root planing. Photomed Laser Surg. 2004;22(6):509-512.
45. Kreisler M, Al Haj H, d’Hoedt B. Clinical efficacy of semiconductor laser application as an adjunct to conventional scaling and root planing. Lasers Surg Med. 2005;37(5):350-355.
46. Qadri T, Miranda L, Tunér J, Gustafsson A. The short-term effects of low-level lasers as adjunct therapy in the treatment of periodontal inflammation. J Clin Periodontol. 2005;32(7):714-719.
47. Kamma JJ, Vasdekis VGS, Romanos GE. The short-term effect of diode laser (980 nm) treatment of aggressive periodontitis: evaluation of clinical and microbiological parameters. Photomed Laser Surg. 2009;27(1):11-19.
48. Caruso U, Nastri L, Piccolomini R, et al. Use of diode laser 980 nm as adjunctive therapy in the treatment of chronic periodontitis. A randomized controlled clinical trial. New Microbiol. 2008;31(4):513-518.
49. Kamma JJ, Vasdekis VG, Romanos GE. The effect of diode laser (980 nm) treatment on aggressive periodontitis: Evaluation of microbial and clinical parameters. Photomed Laser Surg. 2009;27(1):11-19.
50. Lin J, Bi L, Wang L, et al. Gingival curettage study comparing a laser treatment to hand instruments. Lasers Med Sci. 2011;26(1):7-11.
51. De Micheli G, de Andrade AK, Alves VT, et al. Efficacy of high intensity diode laser as an adjunct to non-surgical periodontal treatment: a randomized controlled trial. Lasers Med Sci. 2011;26(1):43-48.
52. Mayrand D, Holt SC. Biology of asaccharolytic black-pigmented Bacteroides species. Microbiol Rev. 1988;52(1):134-152.
53. Socransky SS, Haffajee AD, Cugini MA, et al. Microbial complexes in subgingival plaque. J Clin Periodontol. 1998;25(2):134-144.
54. Loomer PM, Armitage GC. Microbiology of periodontal diseases. In: Rose LF, Mealey BL, Genco RJ, Cohen DW, eds. Periodontics: Medicine, Surgery and Implants. St. Louis, MO: Mosby; 2004:75.
55. Paster BJ, Olsen I, Aas JA, Dewhirst FE. The breadth of bacterial diversity in the human periodontal pocket and other oral sites. Periodontol 2000. 2006;42:80-87.
56. NIH News. Mouse study explains bacterium’s unique role in periodontitis. National Institutes of Health. https://www.nih.gov/news/health/nov2011/nidcr-29.htm. Accessed February 24, 2012.
57. Socransky SS, Haffajee AD, Ximenez-Fyvie FA, et al. Ecological considerations in the treatment of Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis periodontal infections. Periodontol 2000. 1999;20:341-362.
58. Mombelli A, Cionca N, Almaghlouth A. Does adjunctive antimicrobial therapy reduce the perceived need for periodontal surgery? Periodontol 2000. 2011;55(1):205-216.
59. Spencer P, Trylovich D, Cobb CM. Chemical characterization of lased root surfaces using Fourier transform infrared photoacoustic spectroscopy. J Periodontol. 1992;63(7):633-636.
60. Spencer P, Cobb CM, McCollum MH, Wieliczka DM. The effects of CO2 laser and Nd:YAG with and without water/air surface cooling on tooth root structure: Correlation between FTIR spectroscopy and histology. J Periodontal Res. 1996;31(7):453-462.
61. Thomas D, Rapley J, Cobb C, et al. Effects of the Nd:YAG laser and combined treatments on in vitro fibroblast attachment to root surfaces. J Clin Periodontol. 1994;21(1):38-44.
62. Gopin BW, Cobb CM, Rapley JW, Killoy WJ. Histologic evaluation of soft tissue attachment to CO2 laser-treated root surfaces: an in vivo study. Int J Periodontics Restorative Dent. 1997;17(4):316-325.
63. McGuire MK, Scheyer ET. Laser-assisted flapless crown lengthening: a case series. Int J Periodontics Restorative Dent. 2011;31(4):357-364.
64. Tucker D, Cobb CM, Rapley JW, Killoy WJ. Morphologic changes following in vitro CO2 laser treatment of calculus-ladened root surfaces. Lasers Surg Med. 1996;18(2):150-156.
65. Sennhenn-Kirchner S, Schwarz P, Schliephake H, et al. Decontamination efficacy of erbium:yttrium-aluminium-garnet and diode laser light on oral Candida albicans isolates of a 5-day in vitro biofilm model. Lasers Med Sci. 2009;24(3):313-320.
66. Yukna RA. Lasers in periodontal therapy. Today’s FDA. 2011;23(3):40-41.
About the Authors
Charles M. Cobb, DDS, MS, PhD
Professor Emeritus
Department of Periodontics
School of Dentistry
University of Missouri-Kansas City
Kansas City, Missouri
Mark S. Blue, DDS
Private Practice of Periodontics
Durango, Colorado
Nabil E. Beaini, DDS
Private Practice of Periodontics
Plano, Texas
Michael R. Umaki, DDS
Private Practice of Periodontics
Honolulu, Hawaii
Keerthana M. Satheesh, DDS, MS
Assistant Professor
Department of Periodontics
School of Dentistry
University of Missouri-Kansas City
Kansas City, Missouri