Plaque Formation and Marginal Gingivitis Associated with Restorative Materials
Abstract
The presence of restorative materials on tooth surfaces is perceived to be a contributing factor to periodontal disease. This observation is a result of the increased accumulation of plaque on restorations adjacent to the gingiva, which may lead to gingivitis. Plaque is believed to adhere better to restorations than to enamel. This may be due to the surface characteristics of restorative materials such as surface roughness and surface-free energy inherent in the materials. This article reviews the experimental studies of plaque formation on different restorative materials. In addition, clinical studies analyzing and comparing restorative materials and the consequent formation of gingivitis are reviewed. While in vitro and in vivo studies show variations in plaque formation among restorative materials and enamel, clinical studies demonstrate that the progression of gingivitis can be prevented if patients maintain adequate oral hygiene and home care. Therefore, instructing the patient to maintain proper oral hygiene and home care is more important than the choice of restorative material.
Plaque-induced gingivitis is gingival inflammation caused by bacterial plaque at the gingival margin. Prevalent in all ages of the dentate population, it is the most common form of periodontal disease.1 Plaque is a biofilm composed of various microbial species in a matrix of microbial byproducts and host-derived factors and attaches to the tooth.2 Local factors such as crowded teeth, tooth anatomy, and restorative margin discrepancies may alter the formation of bacterial plaque.3 Throughout the years, studies have considered the effects of the integrity of the restorative surface, type of restorative materials, and location of the restorative margin placement on the periodontal tissues as factors that exacerbate gingival diseases.4 One concern of clinicians is the placement of restorative margins and its impact on the violation of the biologic width. This may lead to gingival inflammation, clinical attachment loss, and bone loss due to the inflammatory response to the microbial plaque situated in deeply placed restorative margins. It appears that a minimum distance of 3 mm between alveolar bone and restorative margins is a prudent dimension in restorative treatment planning.5 A review6 of several clinical studies has shown varying degrees of microbial colonization when comparing restorative materials or when comparing them to natural teeth.
The topic of how different restorative materials affect plaque formation is important. The clinician may choose a specific restorative material over another based on its known attraction to plaque and its potential to initiate gingival disease.
Formation of Plaque
Within seconds of polishing the teeth, a thin layer called the pellicle covers the tooth. This layer is composed of salivary components such as glycoproteins, phosphoproteins, enzymes, and other bacterial byproducts that coat the tooth surface. Then, the pellicle attracts bacteria through physiochemical interactions (such as van der Waals forces, electrostatic repulsive forces, and covalent, ionic, or hydrogen bonding). Through coaggregration and coadhesion, the motile bacteria attract other bacteria onto the tooth surface. Colonization of more bacteria ensues and forms a complex plaque biofilm.2
Plaque forms on both natural tooth and artificial surfaces. Surface roughness7-10 and surface-free energy11-14 may influence adhesion. Microscopy observations have shown that early microbial colonization of enamel surfaces tends to start in surface irregularities such as cracks, grooves, and abrasion defects. Other data have indicated roughening of intraoral surfaces spurs growth rate and maturation of supragingival plaque.12 Conversely, physiochemical interactions between bacteria and the surface are affected by their charges and forces (van der Waals force and electrostatic repulsion). This surface-free energy influences bacterial adhesion. High-energy surfaces collect more plaque, bind the plaque more strongly, and select specific bacteria.12 Although these factors interact, surface roughness is more important than surface-free energy.10,13 Alternatively, subgingival plaque formation is influenced by other conditions such as a favorable environment for bacterial growth, making surface characteristics (roughness and energy) less influential. Overall, smooth surfaces and low surface-free energy are better conditions for minimizing supragingival plaque formation.10,14 The complexity and pathogenicity of subgingival plaque formation in restorations placed subgingivally has been confirmed in a longitudinal study of 26 years, showing the placements of restorations below the gingiva are detrimental to gingival and periodontal health even in a population characterized as having good-to-moderate oral hygiene.15
Plaque Adhesion to Restorative Materials
Investigations comparing plaque adhesion to various restorative materials and to enamel have been performed using in vitro and in vivo models.9,10 One in vitro study16 concluded that bacterial adhesion to resin is associated with the characteristic extracellular ultrastructure of the streptococcal bacteria being studied. Hannig6 investigated the ultrastructural pattern of early plaque formation using transmission electron microscopy. This in vivo study compared amalgams, casting alloys, titanium, ceramics, glass polyalkenoate cements, composite resins, unfilled resins, and bovine enamel. The authors concluded early plaque formation behaves similarly in all materials, including enamel. Apparently, the pellicle layer is more important than the difference in material or enamel.6,17 The pellicle layer seems to mask any variance among materials with regard to surface properties and biocompatibility. While previous research has analyzed the qualitative difference among dental materials and enamel, an in vitro study18 examined the amount of bacteria adhering to different restorative materials using radioisotopes. This study compared amalgam, gold alloy, resin composite, and three ceramic materials. The investigation concluded that bacteria adhered most to amalgam, then to resin composite, then to casting gold restoration, and least to the ceramics. However, when amalgam and composite were polished, bacterial adhesion dramatically decreased in all materials, except porcelain. Regardless of the polishing method, no differences of bacterial adhesion were found in comparing the three ceramic materials.
Another in vitro study19 similarly showed that Streptococcus mutans adhered more to glass ionomer, amalgam, composite, and resin-modified glass ionomer compared to ceramics. Other investigations20,21 concluded amalgam and ceramics harbored less plaque compared to resins. A different in vitro study22 showed the effect of various degrees of surface roughness of porcelain on adhesion of bacteria. The researchers found the smoother surface had less plaque accumulation. Overall, smoother surfaces of restorative materials correlate with less plaque accumulation.19 Yet in another study comparing porcelain, acrylic resin, gold, and metal,23 the investigators found no difference in plaque retention between acrylic resin and porcelain. However, when contrasting porcelain with gold and metal, researchers found less plaque accumulation on porcelain than the two metals.
Other types of investigations looked at the viability of microbes on surfaces of the restorative materials. In vitro studies comparing plaque vitality to metals,24 glass ionomer, resin-modified glass ionomer, composites, and resins19,25-27 showed less plaque vitality in regard to the metals and fluoride-releasing restorative materials compared to resins and composites. Some investigators28,29 attribute this to the antimicrobial property of fluoride.
A clinical study30 investigated four finishing techniques on composite restorations. The composite restorations harbored plaque within 24 hours compared to 72 hours for the control teeth (enamel, polished metallic, or glazed porcelain). The authors attributed the statistically significant difference to the composite materials not being as smooth as the other restorative surfaces. A clinical experiment20 found two types of ceramic restorations harbored less plaque than enamel or composite and surmised that polishing of the composites did not achieve the smoothness of the ceramics' finish. A clinical study31 compared the amount of S mutans in plaque from supragingival margins of 2-year-old amalgam, composite, and glass-ionomer restorations in children. The investigators found more plaque mass on composites compared to amalgams and observed the least amount of plaque on glass-ionomer restorations. The authors surmised the reason for less plaque on glass ionomer is due to this material's fluoride release. Plaque acidogenicity is reduced by fluoride, thereby hindering the growth of S mutans. However, one study32 that compared 1-year-old subgingival composite and glass-ionomer restorations in an unspecified population age group found no difference in the presence of S mutans. All patients had good oral hygiene. The differences between the previous two studies might be the location of plaque samples and patient age.
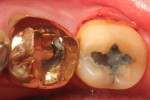
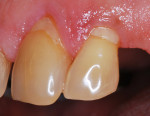
In summary, in vitro and in vivo experiments show that the pattern of early plaque formation is similar for different types of restorative materials compared to enamel due to the presence of the pellicle. As plaque matures, the amount of bacterial plaque adhesion depends on the smoothness of the restoration as seen by differences of plaque adhesion to different restorative materials and as observed in clinical investigations. Figure 1 shows plaque accumulation on two different restorative surfaces. The types of restorative material, such as glass ionomer, may also alter the formation of plaque; however, this needs further conclusive evidence.
Consequence: Gingivitis
The formation and maturation of plaque on the surface of enamel or restorative materials will affect the adjacent soft tissue and elicit an inflammatory host response. This occurs if plaque is not removed. Experiments in which patients are purposely told not to perform oral hygiene for days to weeks so that plaque and gingivitis develop are experimental gingivitis models.33 Using this model, one clinical study34 found no difference in plaque accumulation, gingival condition, and inflammatory response (as measured by the presence of interleukin-1 cytokine levels) when comparing ceramic or composite restorations and enamel. Van Dijken et al35 tested two types of composite materials (conventional and hybrid) and compared them to enamel using the model. Although the surface roughness of the composites varied, no significant difference in development of plaque or gingivitis between the two composites and the enamel surfaces resulted.
The previous group36 studied three types of subgingival composite restorations (conventional, hybrid, and microfilled) that were 1 year to 2 years old or 3 years to 4 years old. They investigated the plaque retention and gingival conditions surrounding the different types of composites and ages and found no significant difference. The investigators noted that all the participants had good oral hygiene as shown by low plaque scores, thus no detrimental responses were seen. In clinical studies,31,37 the authors compared the responses of the marginal gingiva to 1-year-old subgingivally located glass-ionomer and composite-resin restorations and enamel. They found no significant difference in the amount of plaque and degree of gingivitis. Participants also had good oral hygiene. In a clinical study34 in which patients had adequate oral hygiene, no difference in plaque accumulation, gingival condition, and inflammatory response was seen when comparing ceramic or composite restorations and enamel.
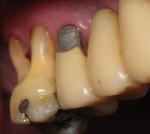
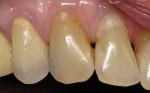
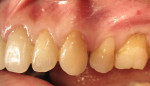
A 1-year study38 looked at the effects of subgingival Class V restorations (amalgam, glass ionomer, and composite) on the gingiva. These restorations did not significantly affect plaque scores, probing depth, and bleeding on probing when compared to sound control teeth. Another study39 compared a novel Class V ceramic filling material to resin composite and found no difference in plaque formation and gingival inflammation. In both studies, the authors noted that patients had good oral hygiene. A clinical investigation40 comparing interleukin-1 levels in gingival crevicular fluid adjacent to Class V restorations of calcium aluminate cement or resin composite determined no difference in a well-maintained patient population. One recent clinical study41 compared the effects of subgingival glass-ionomer and microfilled composites on the periodontal tissues and formation of subgingival biofilm for a 6-month period. Although the glass-ionomer restoration had more positive clinical and theoretical effects (good marginal adaptation, reduced surface roughness, aluminum and fluoride release that may interfere with bacterial adhesion and inhibit bacterial metabolism) on the subgingival biofilm composition than the microfilled composite, both well-finished restorations did not negatively affect periodontal health. Like previous studies, the participants had good oral hygiene compliance. Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6 show clinical situations of restorations and the presence or absence of gingivitis.
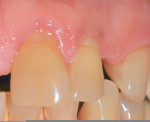
In summary, clinical studies investigating the effect of different restorative materials on the gingiva do not show a detrimental consequence as long as patients demonstrate adequate-to-good oral hygiene. In certain cases, patients with cervical lesions that harbor plaque may benefit from having a restoration placed because these areas become more cleansable. Figure 7, for example, shows plaque accumulation on teeth but not on the composite restoration.
Conclusion
It has long been postulated that rough surfaces and high-energy surfaces tend to accumulate more plaque. This has been shown in mathematical models and in vitro and in vivo studies in which surfaces of different materials are demonstrated to be rougher than others. Most notably, porcelain surfaces are smoother than composite surfaces; therefore, the former will accumulate less plaque. Although this has been shown in a number of in vitro and in vivo studies, it is more important to analyze investigations that include the effects of plaque formation on the gingiva. Clinical studies have shown that people with good or adequate oral hygiene will not develop a markedly diseased gingiva despite the surface roughness or composition of the restorative material and consequent plaque formation. Therefore, the choice of a material is not as important as providing oral hygiene instructions to the patient. If a patient presents with less-than-adequate oral hygiene, then periodontal problems may be expected regardless of restorative material.
Acknowledgment
The authors would like to thank Drs. Jackson Linger, Shilpa Kolhatkar, and Syed Khalid for their contributions in furnishing clinical photographs.
References
1. Mariotti A. Dental plaque-induced gingival diseases. Ann Periodontol. 1999;4(1):7-19.
2. Quirynen M, Teughels W, Kinder Haake S, et al. Microbiology of periodontal diseases. In: Newman MG, Takei HH, Carranza FA, Klokkevold PR, eds. Carranza's Clinical Periodontology. 10th ed. Philadelphia, PA: Saunders Elsevier; 2006:138-143.
3. Blieden TM. Tooth-related issues. Ann Periodontol. 1999;4(1):91-97.
4. Broadbent JM, Williams KB, Thomson WM, Williams SM. Dental restorations: a risk factor for periodontal attachment loss? J Clin Periodontol. 2006;33(11):803-810.
5. Padbury A Jr, Eber R, Wang HL. Interactions between the gingiva and the margin of restorations. J Clin Periodontol. 2003;30(5):379-385.
6. Hannig M. Transmission electron microscopy of early plaque formation on dental materials in vivo. Eur J Oral Sci. 1999;107(1):55-64.
7. Beyth N, Bahir R, Matalon S, et al. StreptococcuS mutans biofilm changes surface-topography of resin composites. Dent Mater. 2008;24(6):732-736.
8. Carlén A, Nikdel K, Wennerberg A, et al. Surface characteristics and in vitro biofilm formation on glass ionomer and composite resin. Biomaterials. 2001;22(5):481-487.
9. Nassar U, Meyer AE, Ogle RE, Baier RE. The effect of restorative and prosthetic materials on dental plaque. Periodontol 2000. 1995;8(1):114-124.
10. Teughels W, Van Assche N, Sliepen I, Quirynen M. Effect of material characteristics and/or surface topography on biofilm development. Clin Oral Implants Res. 2006;17(suppl 2):68-81.
11. Knorr SD, Combe EC, Wolff LF, Hodges JS. The surface free energy of dental gold-based materials. Dent Mater. 2005;21(3):272-277.
12. Quirynen M. The clinical meaning of the surface roughness and the surface free energy of intra-oral hard substrata on the microbiology of the supra- and subgingival plaque: results of in vitro and in vivo experiments. J Dent. 1994;22(suppl 1):S13-S16.
13. Quirynen M, Marechal M, Busscher HJ, et al. The influence of surface free energy and surface roughness on early plaque formation. An in vivo study in man. J Clin Periodontol. 1990;17(3):138-144.
14. Quirynen M, Bollen CM. The influence of surface roughness and surface-free energy on supra- and subgingival plaque formation in man. A review of the literature. J Clin Periodontol. 1995;22(1):1-14.
15. Schätzle M, Land NP, Anerud A, et al. The influence of margins of restorations of the periodontal tissues over 26 years. J Clin Periodontol. 2001;28(1):57-64.
16. Yamamoto K, Noda H, Kimura K. Adherence of oral streptococci to composite resin restorative materials. J Dent. 1989;17(5):225-229.
17. Meier R, Hauser-Gerspach I, Lüthy H, Meyer J. Adhesion of oral streptococci to all-ceramics dental restorative materials in vitro. J Mater Sci Mater Med. 2008;19(10):3249-3253.
18. Kawai K, Urano M. Adherence of plaque components to different restorative materials. Oper Dent. 2001;26(4):396-400.
19. Eick S, Glockmann E, Brandl B, Pfister W. Adherence of Streptococcu S mutans to various restorative materials in a continuous flow system. J Oral Rehabil. 2004;31(3):278-285.
20. Hahn R, Weiger R, Netuschil L, Brüch M. Microbial accumulation and vitality on different restorative materials. Dent Mater. 1993;9(5):312-316.
21. Dummer PM, Harrison KA. In vitro plaque formation on commonly used dental materials. J Oral Rehabil. 1982;9(5):413-417.
22. Kawai K, Urano M, Ebisu S. Effect of surface roughness of porcelain on adhesion of bacteria and their synthesizing glucans. J Prosthet Dent. 2000;83(6):664-667.
23. Wise MD, Dykema RW. The plaque-retaining capacity of four dental materials. J Prosthet Dent. 1975;33(2):178-190.
24. Auschill TM, Arweiler NB, Brecx M, et al. The effect of dental restorative materials on dental biofilm. Eur J Oral Sci. 2002;110(1):48-53.
25. Scherer W, Lippman N, Kaim J. Antimicrobial properties of glass-ionomer cements and other restorative materials. Oper Dent. 1989;14(2):77-81.
26. Kawai K, Takaoka T. Inhibition of bacterial and glucan adherence to various light-cured fluoride-releasing restorative materials. J Dent. 2001;29(2):119-122.
27. Brambilla E, Cagetti MG, Gagliani M, et al. Influence of different adhesive restorative materials on mutans streptococci colonization. Am J Dent. 2005;18(3):173-176.
28. Palenik CJ, Behnen MJ, Setcos JC, Miller CH. Inhibition of microbial adherence and growth by various glass ionomers in vitro. Dent Mater. 1992;8(1):16-20.
29. van Dijken JW, Kalfas S, Litra V, Oliveby A. Fluoride and mutans streptococci levels in plaque on aged restorations of resin-modified glass ionomer cement, compomer and resin composite. Caries Res. 1997;31(5):379-383.
30. Weitman RT, Eames WB. Plaque accumulation on composite surfaces after various finishing procedures. J Am Dent Assoc. 1975;91(1):101-106.
31. Svanberg M, Mjör IA, Orstavik D. Mutans streptococci in plaque from margins of amalgam, composite, and glass-ionomer restorations. J Dent Res. 1990;69(3):861-864.
32. van Dijken JW, Sjöström S. The effect of glass ionomer cement and composite resin fillings on marginal gingiva. J Clin Periodontol. 1991;18(3):200-203.
33. Löe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;36(3):177-187.
34. Konradsson K, Claesson R, van Dijken JW. Dental biofilm, gingivitis and interleukin-1 adjacent to approximal sites of a bonded ceramic. J Clin Periodontol. 2007;34(12):1062-1067.
35. van Dijken JW, Sjöström S, Wing K. Development of gingivitis around different types of composite resin. J Clin Periodontol. 1987;14(5):257-260.
36. van Dijken JW, Sjöström S, Wing K. The effect of different types of composite resin fillings on marginal gingiva. J Clin Periodontol. 1987;14(4):185-189.
37. van Dijken JW, Sjöström S. Development of gingivitis around aged restorations of resin-modified glass ionomer cement, polyacid-modified resin composite (compomer) and resin composite. Clin Oral Investig. 1998;2(4):180-183.
38. Paolantonio M, D'ercole S, Perinetti G, et al. Clinical and microbiological effects of different restorative materials on the periodontal tissues adjacent to subgingival class V restorations. J Clin Periodontol. 2004;31(3):200-207.
39. Konradsson K, van Dijken JW. Effect of a novel ceramic filling material on plaque formation and marginal gingiva. Acta Odontol Scand. 2002;60(6):370-374.
40. Konradsson K, van Dijken JW. Interleukin-1 levels in gingival crevicular fluid adjacent to restorations of calcium aluminate cement and resin composite. J Clin Periodontol. 2005;32(5)462-466.
41. Santos VR, Lucchesi JA, Cortelli SC, et al. Effects of glass ionomer and microfilled composite subgingival restorations on periodontal tissue and subgingival biofilm: a 6-month evaluation. J Periodontol. 2007;78(8):1522-1528.
About the Author
Luis A. Litonjua, DMD, DDS, MS
Clinical Assistant Professor
Department of Periodontology and Dental Hygiene and Department of Diagnostic Sciences
University of Detroit Mercy School of Dentistry
Detroit, Michigan
Leyvee L. Cabanilla, DDS, MSD
Associate Professor
Department of Periodontology and Dental Hygiene
University of Detroit Mercy School of Dentistry
Detroit, Michigan
Lawrence J. Abbott, DDS, MBA
Associate Professor
Department of Restorative Dentistry
University of Detroit Mercy School of Dentistry
Detroit, Michigan