Reactive Oxygen Species and Antioxidant Defense Mechanisms in the Oral Cavity: A Literature Review
Symone M. San Miguel, DMD, PhD;1 Lynne A. Opperman, PhD;2 Edward P. Allen, DDS, PhD;3 and Kathy K.H. Svoboda, PhD4
Abstract:
Through dental procedures and environment, periodontal tissues are exposed to many types of reactive oxygen species (ROS). Recently, various forms of antioxidants have been introduced as an approach to fight dental diseases and improve general gingival health. This article focuses on the classification of antioxidants and the link between oxidative stress and periodontal disease. The protective mechanisms of antioxidants and how routine dental procedures may increase ROS is discussed. The final section reviews the effect of tobacco products on gingival health and disease.
An antioxidant is any substance that significantly delays or prevents oxidation of an oxidizable substrate1 (Figure 1) . Antioxidants are classified by five methods: mode of function, location of action, solubility, structural dependence, and origin and source. Each method is summarized in Table 1.2
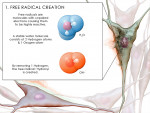
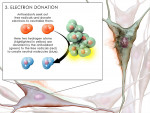
The physiologic and pathologic changes in the human body depend on free radical (FR) and reactive oxygen species (ROS) interactions (Figure 2 and Figure 3) to maintain normal cellular activities.3 In aerobic organisms, the imbalance between ROS generation and antioxidant levels leads to increased oxidative stress and cellular degeneration (Figure 3 ). Antioxidants can counter the formation of free radicals (Figure 4 and Figure 5) and prevent free radical damage by donating electrons.
The involvement of ROS and the antioxidant defense mechanisms in human saliva has been demonstrated in various processes of the oral cavity: healing periodontal disease, preventing oral carcinogenesis, reducing oral mucosa inflammatory reactions, and ameliorating metal-based restoration reactions.4
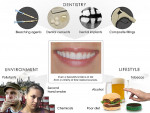
The salivary antioxidant system has an essential anticarcinogenic role in the oral cavity, aimed at fighting ROS and reactive nitrogen species (RNS) caused by smoking, alcoholic beverages, food, carbonated drinks, dental restorations and/or various other volatile sources freely entering the oral cavity through the body's largest open gate—the mouth (Figure 6).5 The salivary antioxidant system includes various molecules and enzymes. The most important are the uric acid molecule and the peroxidase enzyme; both are water-soluble. Uric acid contributes approximately 70% of the total salivary antioxidant capacity.6 An animal model showed the anticarcinogenic capability of saliva significantly inhibits the initiation and progression of oral cancer.7 In a study using the Ames test, saliva inhibited the mutagenicity of oral cancer inducers: cigarette smoke and 4-nitroquinoline 1-oxide (4NQ0).8 Saliva also plays an important role in preventing cigarette-induced deoxyribonucleic acid (DNA) damage.9 This antioxidant capacity of saliva protects against oral cancers.10 However, in cases of periodontal disease formation and progression when normal cellular mechanisms are hampered, the oxidation process occurs due to an increased ROS production induced by other etiologic factors of periodontitis, such as bacterial plaque formation./p>
Bacterial plaque is the most important substrate in periodontal disease development. A study of dental plaque bacteria has implicated its role in inducing "oxygen shock" to activate free radicals and the collagen-destroying enzymes. The process of collagen matrix degradation affects not only the amount of bone destruction and rate of inflammation but also the free radical damage, mechanical trauma, and tissue destruction.11 Antioxidants cause the degradation of mineral deposits, making them water-soluble. The calcified dental plaque is composed of calcium carbonate, which is converted to calcium bicarbonate, becoming water-soluble as mentioned above. Antioxidants can prevent the formation of dental stains that occur from oxidation of both organic and inorganic compounds.12
Thus, oxidative stress is caused by an imbalance between the production of ROS and the body's ability to produce sufficient antioxidants and repair the resulting damage (Figure 4 and Figure 5). The action and protective mechanism of a single antioxidant depends on the concentration, specific reactivity of the ROS, and condition of the antioxidant interaction (Figure 1).13
Link Between Oxidative Stress and Periodontal Disease
Periodontitis affects the teeth's supportive tissues (gingiva, cementum, periodontal ligament, and alveolar bone). Periodontitis is caused by disequilibrium between periodontal tissue destruction and repair involving the host response to bacterial challenge. Polymorphonuclear leukocytes (PMNs) are the primary host defense against periodontal pathogens. Individuals with periodontal disease have an imbalance between oxidants and antioxidants. Following bacterial antigen stimulation, ROS or free radical molecular species are generated by PMNs, as a result of the inflammatory tissue response mechanisms. These mechanisms have been implicated in ROS involvement in periodontal disease, including the possible interactions of PMNs with respect to the level of oxidation products and transition metal ions, neutrophil dysfunction, and antioxidant levels.14
The possible association between low antioxidant levels and periodontal infection has been the subject of several studies. A previous study indicated 8-hydroxydeoxyguanosine (8-OHdG) levels (a marker for oxidative damage in chronic inflammatory diseases) correlate with periodontal health.15 Chemically, 8-OHdG is generated from guanosine by the action of reagents that generate oxygen radicals.15 Individuals with periodontal disease have a significant increase in the activities of enzymatic and nonenzymatic antioxidants in gingival tissue compared to healthy control patients. These results show an important correlation between oxidative stress biomarkers and periodontal disease.16 Another important biomarker for oxidative damage is protein carbonylation. This process of incorporating the carbonyl groups into proteins by various oxidative pathways has been shown in some studies17—an ROS reacts with proteins to generate products with a reactive carbonyl species, leading to the formation of protein carbonyl derivatives (aldehydes and ketones).16 The elevated protein carbonylation in serum and increased levels of gingival crevicular fluid (GCF) indicated the amount of both systemic and local/periodontal protein carbonylation correlate with periodontal status.18
In the oral microflora, the increased level of systemic oxidative stress will lead to a generalized decrease of serum immunoglobulin G (IgG) antibody.19 Other minor modifiers, such as the level or condition of the oral microbial environment, status of periodontal disease, and smoking, can also directly influence the serum IgG levels.19 To identify microbial species associated with destructive periodontal disease progression, one study used serum IgG antibody against suspected periodontal pathogens in order to detect disease-susceptible or disease-resistant individuals.20 The formation of IgG subclasses in humans is necessary to react with different types of antigens, and its expression may help define the diseased condition.19,21 The isotype-specific and IgG subclass-specific antibody synthesis can influence the stress and mediators of oxidative stress. These mechanisms were shown in a study in which increased plasma IgG2 levels was not observed in smokers. The oxidative stress generated by smoking decreased the reaction of IgG2 antibody to oral microbes.22
One of the key ROS sources in periodontal tissues is the phagocyte-generated nicotinamide adenine dinucleotide phosphate oxidase (NOX2). In cases of aggressive periodontitis, this increased ROS is attributed to the patient's genetics and the action of oral pathogens to increase the NOX2 function.23 A positive association was found between plasma reactive oxygen metabolites (ROM)—a measure of blood ROS—and clinical gingival attachment levels in patients who are in the maintenance phase of periodontal therapy.24 Although it remains unclear whether the increased plasma ROM level is a cause or a result of periodontal disease, some research has shown periodontal therapy is effective in reducing plasma ROM level.25 A study reported, "...plasma glutathione peroxidase levels in gingival crevicular fluid of patients with gingivitis or periodontitis increased proportionality with the severity of disease."26 The relationships between the levels of GCF, interleukin1ß (IL-1ß), and thiobarbituric acid is an important experimental parameter for treatment and monitoring the patterns of periodontal disease progression.27 Although most university-based clinical studies have shown promising results, further research is needed to try some assays that may help define specific markers in periodontal disease.28
A particularly important aspect of oxidative stress is the systemic oxidative stress that is involved in many diseases, such as diabetes mellitus, heart failure, atherosclerosis, myocardial infarction, and respiratory disease. An association has been observed that people who have periodontal disease often show increased glucose levels. Patients with well-controlled diabetes (measured by blood glycated hemoglobin levels) have less severe periodontal disease than those who have poorly controlled diabetes.29 In cardiovascular disease (CVD), the same bacteria that causes periodontal infection can enter the bloodstream and contribute to the chemistry that forms arterial plaques and clogs arteries.30-31 Consequently, these inflammatory products may cause atherosclerosis, which reduces blood supply to the tissues and organs. People who have periodontal disease are at high risk for coronary heart disease and carotid intima-medial thickening.32 The moderate elevation of C-reactive protein (CRP) is a predictor of increased risk for CVD.33 Periodontal infections also can lead to a higher risk for respiratory disease. People who normally smoke and those with poor immune response are more susceptible to respiratory diseases, including pneumonia, bronchitis, emphysema, and chronic obstructive pulmonary disease.34
Sources of Free Radical Insults in Dental Therapy
In dentistry, many commonly used dental materials may form free radicals (Figure 6). Although there are no major health concerns about the components of dental materials used in clinical procedures, studies have evaluated the following:
Bleaching agents: During tooth whitening procedures, 3% to 35% hydrogen peroxide (H2O2) can leak onto the oral mucosa membrane and irritate soft tissues, causing pain. H2O2 has oxidative ability, and the hydroxyl radical (-OH) that is generated exert biologic reactions, such as inflammation, carcinogenesis, aging, and mutation.35
Composite fillings: Composite resin components may be cytotoxic-damaging or destroying cells. Primary human periodontal ligament and pulp fibroblasts were more responsive than 3-day transfer, inoculum 3 x 105 cells (3T3) and gingival fibroblasts with respect to the tested materials, including urethane dimethacrylate, Bowen monomer, isopropyliden-bis (2-hydroxy-3-[4-phenoxy]-propylmethacrylate, dimethoxybenzoin, and N,N-dimethyltetradecylamine or moderate (2-hydroxyethyl methacrylate), benzyl methacrylate, camphorquinone, dimethyl-p-toluidine, and 2-(4-dimethyl-aminophenyl) ethanol.36 In addition, chemically cured composite resins contain the initiator benzoyl peroxide,37 which is carcinogenic38 and can impair the cellular pro- and antioxidant redox balance.39-40
Dental cements: Eugenol is widely used as a component of zinc oxide eugenol cement in dentistry and is used in the oral environment.41 Eugenol produced radicals in alkaline solutions, with an optimum pH of 9.5. The cytotoxicity of eugenol-related compounds was significantly associated with the production of phenoxyl radicals.42
Ceramic restorations: These have radioactive fluorescing agents that may lead to silica granuloma.43 Silica granuloma is one type of chronic inflammatory reaction that has been shown to generate free radicals in vivo where elevated levels of inducible nitric oxide synthase (iNOS) are in the tumor tissue. Nitric oxide (NO) thus facilitates vascular permeability, which accelerates nutritional supply to the tumor tissue, and hence sustains the rapid tumor group.44
Metals in restorations: A current issue in dental practice is the use of metals in fillings, braces, bridges and other prosthodontic restorations. Toxicity studies on commonly used metal salts found in fixed prosthodontic restorations, showed that Zn and Cu released from gold alloys, and Ni released from nickel-chromium alloys, have a highly significant cytotoxic activity on fibroblast cell cultures.45-46
Dental implants: Current dental implants are made of titanium, which is efficacious for oral rehabilitation procedures. The presence of nanoscale titanium dioxide (TiO2) at implant surfaces47 provides a wide-band gap semiconductor48 that produces radicals O2-, H2O2, or OH-, when illuminated with ultraviolet light.49 These free radicals are used to destroy bacteria,50 and the nanostructured TiO2 on most dental implant surfaces acts to absorb the incident radiation and become activated by sterilizing the surface chemically.51 However, some in vitro studies on other cell types showed that titanium dioxide had an inductive effect on oxidative stress and apoptosis in PC12 cells.52 The testing of different particle sizes (nano to micro) of titanium dioxide demonstrated an increased production of IL-1ß as mediated by ROS and cathepsin B in macrophage-like human THP-1 cells, suggesting its involvement in inflammatory reactions.53
Nicotine-Induced Free Radical Formation and Periodontal Disease
Recent studies have shown tobacco use may be one of the most significant risk factors in the development and progression of periodontal disease because it promotes a high degree of ROS release, culminating in heightened oxidative damage to gingival tissue, periodontal ligament, and alveolar bone (Figure 6).54 Many studies have shown the effects of continued smoking on persistent gingival bleeding,55 vertical bone loss,56 and poor treatment outcomes.57 Similarly, in vitro studies have demonstrated nicotine inhibits the attachment and growth of gingival and periodontal ligament fibroblasts58 and decreases fibroblast migration through the Rac-GTP signaling pathway.59 Furthermore, chemicals in tobacco also can affect healing and cause poor treatment outcomes.57 Other tobacco products can harm periodontal health. Smokeless tobacco can cause gingival recession and worsen periodontal disease. Both cigar and cigarette smokers have the same degree of tooth and bone loss; however, pipe and cigarette smokers had a similar rate of tooth loss. All tobacco products cause a higher oral cancer risk, halitosis, stained teeth, bone loss, loss of taste, less successful periodontal treatment, less success with dental implants, gingival recession, mouth sores, and facial wrinkling.57,60
Several studies have evaluated the cellular mechanisms induced by tobacco smoking that negatively influence oral health. In addition to stimulating production of ROS, smoking may reduce antioxidant levels. A dose-related reduction of salivary and gingival crevicular fluid superoxide dismutase levels was found in both light and heavy smokers compared to nonsmokers.61 Smokers also had significantly lower serum levels of vitamin C than other levels of nonenzymatic antioxidants, such as vitamins A and E and coenzyme Q10. Nicotine is a chemical substance that can affect everyone by either direct contact or secondhand inhalation.62 Song et al63 employed a urine cotinine test for the determination of smoking status. Cotinine is a neuroactive metabolite of nicotine, which is useful in monitoring patients with cognitive and behavioral disorders.64 High levels of urine cotinine have been found in nonsmokers, illustrating the significance of passive smoking exposure.63 At the cellular level, protein content was significantly decreased and cell membranes were damaged in the presence of nicotine. Nicotine concentrations higher than 3.9 mm revealed changes in the morphology of the cytoskeletal elements, microtubules, and vimentin. Moreover, cells exposed to nicotine have increased vacuoles and decreased fibroblast growth at concentrations higher than 7.8 mM.65 Nicotine doses between 10.5 mM and 15.5 mM cause irreversible toxicity compared with lower concentrations.65 In addition, nicotine decreased cell-signaling molecules and altered the response to transforming growth factor-ß1 (TGF-ß1) by decreasing the morphologic change from fibroblast to myofibroblast cells,66 supporting the theory that nicotine interferes with wound closure by changing the gingival fibroblast ability to contract wounds.
Human periodontal ligament (PDL) cells respond to nicotine and tobacco extracts nearly the same way as gingival fibroblasts by changing morphology and structure, with decreased growth and attachment through cytoskeletal disruption.67 PDL cells were flattened in the control groups but rounded in the smoking groups, indicating a change in cytoskeletal structure. The investigators suggested cigarette smoking compromises PDL cell adhesion to root surfaces, which might affect periodontal regeneration following therapy.68 Recent studies have also shown that nicotine decreases PDL and gingival fibroblast migration but treatment with antioxidants reverse the cellular behavior and increase migration rates.69
Conclusion
ROS compromises the body's ability to fight infection and repair tissue in periodontal disease, as well as in many systemic conditions. Based on a new classification system of periodontal disease,70 further studies can help establish the link between periodontal disease and other elevated markers of inflammation in systemic diseases, such as diabetes, heart failure, atherosclerosis, myocardial infarction, and respiratory disease.71-76
Dental products as well as dietary and environmental compounds are among the many sources of ROS. In particular, tobacco smoking promotes ROS release, resulting in heightened oxidative damage to periodontal tissues. Tobacco plays a significant role in many diseases, because it affects almost every organ and reduces general and oral health of both smokers and nonsmokers.
Antioxidant treatment can block the production of ROS or obstruct its effects and may be therapeutically valuable in reducing the risk for many dental maladies. Recent abstracts presented at national meetings have focused on the application of bioactive antioxidant compounds that can counteract effects of nicotine on gingival and periodontal ligament cells. Some of the most promising technologies use an engineered mammalian phloretin-adjustable control element to program expression of difficult-to-produce protein therapeutics during standard bioreactor operation in subcutaneous implants.71 This research could provide alternative modes of drug delivery for antioxidant compounds for future clinical trials in the treatment of chronic periodontitis.
About the Authors
Symone M. San Miguel, DMD, PhD1
Postdoctoral Fellow, Biomedical Sciences,
Baylor College of Dentistry,
Texas A&M Health Science Center,
Baylor College of Dentistry,
Dallas, Texas
Lynne A. Opperman, PhD2
Professor of Biomedical Sciences and Director of Technology Development,
Texas A&M Health Science Center,
Baylor College of Dentistry,
Dallas, Texas
Edward P. Allen, DDS, PhD3
Adjunct Professor, Department of Periodontics,
Texas A&M Health Science Center,
Baylor College of Dentistry,
Dallas, Texas
Kathy K.H. Svoboda, PhD4
Professor of Biomedical Sciences and Graduate Program Director,
Texas A&M Health Science Center,
Baylor College of Dentistry,
Dallas, Texas
References
1. Halliwell B. Antioxidants: the basics—what they are and how to evaluate them. Adv Pharmacol. 1997;38:3-20.
2. Chapple IL, Matthews JB. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontol 2000. 2007;43:160-232.
3. Chapple IL, Brock GR, Milward MR, et al. Compromised GCF total antioxidant capacity in periodontitis: cause or effect? J Clin Periodontol. 2007;34(2):103-110.
4. Sakagami H, Oi T, Satoh K. Prevention of oral diseases by polyphenols (review). In Vivo. 1999;13(2):155-171.
5. Hershkovich O, Shafat I, Nagler RM. Age-related changes in salivary antioxidant profile: possible implications for oral cancer. J Gerontol A Biol Sci Med Sci. 2007;62(4):361-366.
6. Moore S, Calder KA, Miller NJ, et al. Antioxidant activity of saliva and periodontal disease. Free Radic Res. 1994;21(6):417-425.
7. Nagler RM, Klein I, Zarzhevsky N, et al. Characterization of the differentiated antioxidant profile of human saliva. Free Radic Biol Med. 2002;32(3):268-277.
8. Dayan D, Hirshberg A, Kaplan I, et al. Experimental tongue cancer in desalivated rats. Oral Oncol. 1997;33(2):105-109.
9. Petti S, Scully C. Polyphenols, oral health and disease: A review. J Dent. 2009 Jun;37(6):413-23.
10. Wu HJ, Chi CW, Liu TY. Effects of pH on nicotine-induced DNA damage and oxidative stress. J Toxicol Environ Health A. 2005;68(17-18):1511-1523.
11. DuPont GA. Understanding dental plaque: biofilm dynamics. J Vet Dent. 1997;14(3):91-94.
12. Izumita D. New approach in dentistry. Clinical and Basic Medical Research on EM-X--A Collection of Research Papers, 2001;2:77-81.
13. Vertuani S, Angusti A, Manfredini S. The antioxidants and pro-antioxidants network: an overview. Curr Pharm Des. 2004;10(14):1677-1694.
14.Waddington RJ, Moseley R, Embery G. Reactive oxygen species: a potential role in the pathogenesis of periodontal diseases. Oral Dis. 2000;6(3):138-151.
15. Takane M, Sugano N, Iwasaki H, et al. New biomarker evidence of oxidative DNA damage in whole saliva from clinically healthy and periodontally diseased individuals. J Periodontol. 2002;73(5):551-554.
16. Borges I Jr, Moreira EA, Filho DW, et al. Proinflammatory and oxidative stress markers in patients with periodontal disease. Mediators Inflamm. 2007;2007:45794.
17. Dalle-Donne I, Aldini G, Carini M, et al. Protein carbonylation, cellular dysfunction, and disease progression. J Cell Mol Med. 2006;10(2):389-406.
18. Baltacioğlu E, Akalin FA, Alver A, et al. Protein carbonyl levels in serum and gingival crevicular fluid in patients with chronic periodontitis. Arch Oral Biol. 2008;53(8):716-722.
19. Singer RE, Moss K, Beck JD, et al. Association of systemic oxidative stress with suppressed serum IgG to commensal oral biofilm and modulation by periodontal infection. Antioxid Redox Signal 2009;11(12):2973-2983.
20. Craig RG, Boylan R, Yip J, et al. Serum IgG antibody response to periodontal pathogens in minority populations: relationship to periodontal disease status and progression. J Periodontal Res. 2002;37(2):132-146.
21. King PT, Hutchinson PE, Johnson PD, et al. Adaptive immunity to nontypeable Haemophilus influenzae. Am J Respir Crit Care Med. 200315;167(4):587-592.
22. Graswinckel JE, van der Velden U, van Winkelhoff AJ, et al. Plasma antibody levels in periodontitis patients and controls. J Clin Periodontol. 2004;31(7):562-568.
23. Giannopoulou C, Krause KH, Müller F. The NADPH oxidase NOX2 plays a role in periodontal pathologies. Semin Immunopathol. 2008;30(3):273-278.
24. Tamaki N, Tomofuji T, Maruyama T, et al. Relationship between periodontal condition and plasma reactive oxygen metabolites in patients in the maintenance phase of periodontal treatment. J Periodontol. 2008;79(11):2136-2142.
25.Tamaki N, Tomofuji T, Ekuni D, et al. Short-term effects of non-surgical periodontal treatment on plasma level of reactive oxygen metabolites in patients with chronic periodontitis. J Periodontol. 2009;80(6):901-906.
26. Patel SP, Pradeep AR, Chowdhry S. Crevicular fluid levels of plasma glutathione peroxidase (eGPx) in periodontal health and disease. Arch Oral Biol. 2009;54(6):543-548.
27. Tüter G, Kurtiş B, Serdar M. Interleukin-1beta and thiobarbituric acid reactive substance (TBARS) levels after phase I periodontal therapy in patients with chronic periodontitis. J Periodontol. 2001;72(7):883-888.
28. Ozmeric N. Advances in periodontal disease markers. Clin Chim Acta. 2004;343(1-2):1-16.
29. Soskolne WA. Epidemiological and clinical aspects of periodontal diseases in diabetics. Ann Periodontol. 1998;3(1):3-12.
30. Pucar A, Milasin J, Lekovic V, et al. Correlation between atherosclerosis and periodontal putative pathogenic bacterial infections in coronary and internal mammary arteries. J Periodontol. 2007;78(4):677-682.
31. Haraszthy VI, Zambon JJ, Trevisan M, et al. Identification of periodontal pathogens in atheromatous plaques. J Periodontol. 2000;71(10):1554-1560.
32. Mustapha IZ, Debrey S, Oladubu M, et al. Markers of systemic bacterial exposure in periodontal disease and cardiovascular disease risk: a systematic review and meta-analysis. J Periodontol. 2007;78(12):2289-2302.
33. Noack B, Genco RJ, Trevisan M, et al. Periodontal infections contribute to elevated systemic C-reactive protein level. J Periodontol. 2001;72(9):1221-1227.
34. Seymour GJ, Ford PJ, Cullinan MP, et al. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007;13(suppl 4):3-10.
35. Kashima-Tanaka M, Tsujimoto Y, Kawamoto K, et al. Generation of free radicals and/or active oxygen by light or laser irradiation of hydrogen peroxide or sodium hypochlorite. J Endod. 2003;29(2):141-143.
36. Kinoshita N, Yamamura T, Teranuma H, et al. Interaction between dental metals and antioxidants, assessed by cytotoxicity assay and ESR spectroscopy. Anticancer Res. 2002;22(6C):4017-4022.
37. Ruyter IE. Physical and chemical aspects related to substances released from polymer materials in an aqueous environment. Adv Dent Res. 1995;9:344-347.
38. Slaga TJ, Klein-Szanto AJ, Triplett LL, et al. Skin tumor-promoting activity of benzoyl peroxide, a widely used free radical-generating compound. Science. 1981;28;213 (4511):1023-5.
39 Di Pietro A, Visalli G, La Maestra S, et al. Biomonitoring of DNA damage in peripheral blood lymphocytes of subjects with dental restorative fillings. Mutat Res. 2008;29;650(2):115-122.
40. Schweikl H, Spagnuolo G, Schmalz G. Genetic and cellular toxicology of dental resin monomers. J Dent Res. 2006;85(10):870-877
41. Markowitz K, Moynihan M, Liu M, et al. Biologic properties of eugenol and zinc oxide-eugenol. A clinically oriented review. Oral Surg Oral Med Oral Pathol. 1992;73(6):729-737.
42. Fujisawa S, Atsumi T, Kadoma Y, et al. Antioxidant and prooxidant action of eugenol-related compounds and their cytotoxicity. Toxicology. 2002;177(1):39-54.
43. Mackert JR Jr. Side-effects of dental ceramics. Adv Dent Res. 1992;6:90-93.
44. Maeda H, Akaike T. Nitric oxide and oxygen radicals in infection, inflammation, and cancer. Biochemistry (Mosc). 1998;63(7):854-865.
45. Elshahawy WM, Watanabe I, Kramer P. In vitro cytotoxicity evaluation of elemental ions released from different prosthodontic materials. Dent Mater. 2009; 12:1551-1555.
46. Elshahawy W, Watanabe I, Koike M. Elemental ion release from four different fixed prosthodontic materials. Dent Mater. 2009;8:976-981.
47. Placko HE, Mishra S, Weimer JJ, et al. Surface characterization of titanium-based dental implant materials. Int J Oral Maxillofac Implants. 2000;15(3):355-363.
48. Ding RG, Lu GQ, Yan ZF, et al. Recent advances in the preparation and utilization of carbon nanotubes for hydrogen storage. J Nanosci Nanotechnol. 2001;1(1):7-29.
49. Wu TX, Liu GM, Zhao JC, et al. Evidence for H2O2 generation during the TiO2-assisted photodegradation of dyes in aqueous dispersions under visible light illumination. J Phys Chem B 1999;103:4862-4867.
50. Tatsuma T, Takeda S, Saitoh S, et al. Bactericidal effect of an energy storage TiO2-Wo3 photocatalyst in dark. Electrochem Commun. 2003;5:793-796.
51. Riley DJ, Bavastrello V, Covani U, et al. An in-vitro study of the sterilization of titanium dental implants using low intensity UV-radiation. Dent Mater. 2005;21(8):756-760.
52. Liu S, Xu L, Zhang T, et al. Oxidative stress and apoptosis induced by nanosized titanium dioxide in PC12 cells. Toxicology. 2010;267(1-3):172-177.
53. Gonzalez R, Arancibia R, Cáceres M, et al. Cigarette smoke condensate stimulates urokinase production through the generation of reactive oxygen species and activation of the mitogen activated protein kinase pathways in human gingival fibroblasts. J Periodontal Res. 2009;44(3):386-394.
54. Morishige T, Yoshioka Y, Tanabe A, et al. Titanium dioxide induces different levels of IL-1beta production dependent on its particle characteristics through caspase-1 activation mediated by reactive oxygen species and cathepsin B. Biochem Biophys Res Commun. 2010;5;392(2):160-165.
55. Dietrich T, Bernimoulin JP, Glynn RJ. The effect of cigarette smoking on gingival bleeding. J Periodontol. 2004;75(1):16-22.
56. Baljoon M. Tobacco smoking and vertical periodontal bone loss. Swed Dent J Suppl. 2005;(174):1-62.
57. Johnson GK, Hill M. Cigarette smoking and the periodontal patient. >J Periodontol. 2004;75(2):196-209.
58. James JA, Sayers NM, Drucker DB, et al. Effects of tobacco products on the attachment and growth of periodontal ligament fibroblasts. J Periodontol. 1999;70(5):518-525.
59. Fang Y, Svoboda KK. Nicotine inhibits human gingival fibroblast migration via modulation of Rac signalling pathways. J Clin Periodontol. 2005;32(12);1200-1207.
60. Albandar JM, Streckfus CF, Adesanya MR, et al. Cigar, pipe, and cigarette smoking as risk factors for periodontal disease and tooth loss. J Periodontol. 2000;71(12):1874-1881.
61. Agnihotri R, Pandurang P, Kamath SU, et al. Association of cigarette smoking with superoxide dismutase enzyme levels in subjects with chronic periodontitis. J Periodontol. 2009;80(4):657-662.
62. González YM, De Nardin A, Grossi SG, et al. Serum cotinine levels, smoking, and periodontal attachment loss. J Dent Res. 1996;75(2):796-802.
63. Song SM, Park YS, Lee A, et al. Concentrations of blood vitamin A, C, E, coenzyme Q10 and urine cotinine related to cigarette smoking exposure. Korean J Lab Med. 2009;29(1):10-16.
64. Terry AV Jr, Hernandez CM, Hohnadel EJ, et al. Cotinine, a neuroactive metabolite of nicotine: potential for treating disorders of impaired cognition. CNS Drug Rev. 2005;11(3):229-252.
65. Alpar B, Leyhausen G, Sapotnick A, et al. Nicotine-induced alterations in human primary periodontal ligament and gingiva fibroblast cultures. Clin Oral Investig. 1998;2(1):40-46.
66. Fang Y, Svoboda KK. Nicotine inhibits myofibroblast differentiation in human gingival fibroblasts. J Cell Biochem. 2005;95(6):1108-1119.
67. Xu Y, Jiang Y, Li S, et al. Effects of tobacco on proliferation and attachment of human periodontal ligament fibroblast. Zhonghua Kou Qiang Yi Xue Za Zhi. 2003;38(5):367-369.
68. Gamal AY, Bayomy MM. Effect of cigarette smoking on human PDL fibroblasts attachment to periodontally involved root surfaces in vitro. J Clin Periodontol. 2002;29(8):763-770.
69.San Miguel S M, Opperman LA, Allen EP, et al. Antioxidants counteract nicotine and promote migration via RacGTP in oral fibroblast cells. J Periodontol 2010:81:1675-1690.
70. Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol. 1999;4(1):1-6.
71. Gitzinger M, Kemmer C, El-Baba MD, et al. Controlling transgene expression in subcutaneous implants using a skin lotion containing the apple metabolite phloretin. Proc Natl Acad Sci U S A. 2009;106(26):10638-10643.
72. Svardal AM, Mansoor MA, Ueland PM. Determination of reduced, oxidized, and protein-bound glutathione in human plasma with precolumn derivatization with monobromobimane and liquid chromatography. Anal Biochem. 1990;184(2):338-346.
73. Avissar N, Whitin JC, Allen PZ, et al. Antihuman plasma glutathione peroxidase antibodies: immunologic investigations to determine plasma glutathione peroxidase protein and selenium content in plasma. Blood. 1989;73(1):318-323.
Dean RT, Fu S, Stocker R, et al. Biochemistry and pathology of radical-mediated protein oxidation. Biochem J. 1997;324(pt 1):1-18.
75. Marriott BM.Functional foods: an ecologic perspective,Am J Clin Nutr. 2000;71(suppl 6):1728S-1734S.
76. Whiteman M, Halliwell B. Prevention of peroxynitrite-dependent tyrosine nitration and inactivation of alpha1-antiproteinase by antibiotics. Free Radic Res. 1997;26(1):49-56.