The Use of Phentolamine Mesylate to Evaluate Mandibular Nerve Damage Following Implant Placement
Stuart J. Froum, DDS; Scott H. Froum, DDS; and Stanley F. Malamed, DDS;
Abstract: High success rates and long-term predictability of implant therapy have been well documented in the literature. However, complications in implant treatment can arise and include sensory disturbances, especially in the posterior mandible in areas close to the inferior alveolar nerve. Treatment efficacy of sensory disturbances caused by implant placement in this area relies on the expeditious diagnosis of an induced paresthesia. Recently, phentolamine mesylate has been introduced as a reversal agent of local anesthesia with the ability to decrease the requisite time for a patient to return to normal sensation. This article introduces a method for faster detection of a potential paresthesia induced by implant placement in the posterior mandible.
Implant restorations have shown high success rates for replacement of single and multiple missing teeth in the posterior mandible.1-4 However, implant complications specific to this area may occur, distressing both patient and clinician. One of the complications of major concern is transient or permanent paresthesia due to implant-related damage to the inferior alveolar nerve (IAN).5-7 Sensory disturbances may include paresthesia or dysesthesia of the lip, tongue, and/or gingiva.
When these problems are diagnosed, it is incumbent on the clinician to refer the patient or to attempt to treat the condition as soon as possible. However, available treatment modalities appear to have low success rates.8
Treatment procedures may include reversing the implant several millimeters to relieve pressure on the nerve caused by bone compression on the canal or removal of the implant if it has impinged on the nerve.9 Following nerve damage, researchers have proposed that early implant removal may lead to better healing with the return of sensation.10 However, in many cases, by the time the damage is diagnosed, days may have elapsed, which results in a delay of implant removal. Because the patient often leaves the surgeon’s office with pupal and soft-tissue numbness, an immediate diagnosis of impaired sensory function cannot be made. A determination that there is a problem may also be delayed because some clinicians use long-acting local anesthetics (0.5% bupivacaine HCl with epinephrine 1:200,000) to avoid pain onset as long as possible. Both of these scenarios may affect the diagnosis and treatment. In addition to signs of sensory disturbance, the decision to reverse torque or remove the implant is made in conjunction with the appropriate radiographs demonstrating the implant’s proximity to the inferior alveolar nerve (IAN) or mental foramen. Often, a computed axial tomography (CT) or cone-beam computed tomography (CBCT) scan is required to definitively determine whether the implant has violated the IAN or canal.
Recently, phentolamine mesylate (PM) 0.4 mg/1.7 mL (OraVerse®, Novalar Pharmaceuticals, https://www.novalar.com) an a-adrenergic antagonist (eg, vasodilator) has been introduced, which has been shown to “reverse” the effect of soft-tissue local anesthesia in a shorter period.11,12 This may enable the clinician to make an early determination as to the presence of nerve damage and help decide if implant removal is necessary. This technology also may aid in the decision to order a CT or CBCT scan soon after sensory problems have been diagnosed, which may be at a much earlier period than if the local anesthetic had been allowed to run its normal course.
Purpose
The present clinical investigation was designed to evaluate the time necessary for return to normal function, return to normal lip and tongue sensation, and the clinical experience in patients who received block and infiltration local anesthesia. The patients were completely numb for implant placement and received PM injections in the same areas immediately following implant placement.
Materials and Methods
The investigation enrolled 10 patients (six males, four females) ranging in age from 35 to 80 years who were scheduled to receive one or more implants in the mandibular posterior quadrant. These patients had been informed of alternate treatments, as well as the risks, benefits, and potential complications of implant placement. Each patient signed an informed consent form. The verbal and written consent included a statement regarding acknowledgement of the risk of nerve damage and any transient or permanent paresthesia that may result from implant surgery.
Patients were also informed that, following surgery, the time that they would have postoperative numbness could be decreased by an injection of PM, which might “reverse” these symptoms.
Participants were told that this drug could facilitate early detection of nerve damage. Furthermore, they were told that removal of the implant within 8 to 12 hours following placement might help restore sensation and allow the nerve to heal.
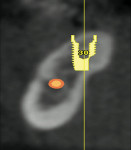
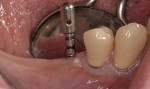
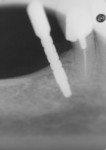
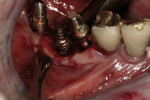
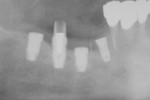
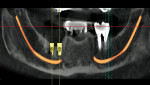
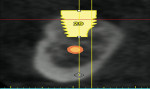
Ten patients who agreed to the injections were enrolled consecutively. Four patients received IAN blocks using one cartridge of 4% articaine (40 mg/mL) with 1:200,000 epinephrine (Septocaine with epinephrine 1:200,000, Septodont, https://www.septodontusa.com) and a buccal injection with one cartridge of the same anesthetic for buccal infiltration of the area. Five patients were injected with two cartridges of 2% lidocaine with 1:100,000 epinephrine (Novocol Pharmaceutical, https://www.novocol.com)—one as a block and one as an infiltration. One patient received an IAN block with one cartridge of 2% lidocaine 1:100,000 and a buccal infiltration with a second cartridge of 4% articaine 1:200,000. Implants were placed using preoperative CT or CBCT scans with an implant simulation program and surgical guide. Guide pins were used during surgery to ascertain the correct path of insertion. However, implants in many cases were in close proximity to the IAN, as seen on final peri-implant radiographs (Figures 1, 2, 3, 4, 5, 6, 7, 8, 9, 10).
Following completion of implant placement and suturing, each patient received two injections (one cartridge as an IAN block and one by infiltration) of PM 0.4 mg/1.7 mL in the same areas that were previously anesthetized prior to surgery. The time of injections was recorded. Patients were instructed to record the time that lip and tongue numbness subsided and the time it took for return of normal oral function. They were told that “normal function” meant they had complete feeling of the lips, gums, and tongue; they could drink water with full control; and they had no spilling, drooling, or accidental biting of the lip, cheek, or tongue. All patients were given postoperative analgesics by mouth immediately following surgery while in the office. These included ibuprofen 800 mg, codeine with acetaminophen, or oxycodine with acetaminophen.
Telephone calls were made 2 to 3 hours after the patients left the office, and their responses were documented. At time of injection of both the local anesthetic and PM, conditions at the site of injection, including redness, edema, and hemorrhage, were recorded by the operator.
The occurrence of any untoward bleeding in the area following the PM injections was recorded while the patient was in the office and asked of the patient that evening on the phone and at the first postsurgical visit.
At the first postoperative visit 7 to 14 days following surgery, each patient was asked if numbness “reversal” improved their dental experience. They were also asked to describe their pain level after surgery when the numbness wore off as “mild,” “moderate,” or “severe” discomfort.
Results
All 10 patients reported “yes” to the following question: “Do you feel the reversal injection improved your dental experience and reduced the expected duration of numbness?” Nine of the 10 patients reported mild discomfort, and one reported moderate discomfort when the numbness completely abated. The patients said lip numbness returned to normal in 30 to 69 mins (average 50.6 mins), tongue numbness returned to normal in 25 to 81 mins (average 60.5 mins), and oral function returned in 25 to 81 mins (average 59.9 mins). In patients who received 2% lidocaine with epinephrine 1:100,000, lip and tongue sensation and oral function return averaged 41.8 mins, 51.8 mins, and 50.8 mins, respectively. This contrasts to the longer time required for patients who received 4% articane with epinephrine 1:200,000 as the local anesthesia—lip, tongue, and oral functional return of sensation averaged 66.75 mins, 71.50 mins, and 71.25 mins, respectively. The one patient who received one cartridge of each local anesthetic reported return of lip, tongue, and oral function in 30 mins, 60 mins, and 60 mins, respectively (Table 1). All patients experienced the complete return of sensation.
In no case was abnormal bleeding seen in or reported by the patients following the PM injection. In six of the 10 patients, redness and some slight swelling were present at the site of both the local anesthesia and PM injections. In the remaining four patients, no soft-tissue redness or swelling was seen following injection of either the local anesthesia or PM. Five of the 10 patients experienced this complete return before leaving the office. In these patients, this occurred from 25 to 60 mins after the PM injections.
Discussion
The prevalence of altered sensation associated with implant surgery has been documented to range from 0% to 9% in early longitudinal studies13,14 and to 35% to 40% in more recent studies.15 In two separate findings using retrospective questionnaires, one in Toronto, Canada and the other in Adelaide, Australia, the prevalence of altered sensation in patients receiving implants was 37% and 36%, respectively.16,17 In the former study, 37% of patients receiving mandibular implants reported altered sensation (24% transient and 13% persistent).16 In the latter Australian study, 36% of the patients reported sensory changes, 23% had transient changes, and 13% experienced persistent changes.17 With current technology, including the use of surgical guides and CT and CBCT scans, the prevalence may be lower. However, persistent numbness remains a major risk for implant placement in the posterior mandible.
Neurosensory alterations associated with implant placement have been classified into three categories according to the severity of nerve damage. These include 1) neuropraxia (mild injury in which feeling is reversed within 4 weeks postsurgery); 2) axonotmesis (nerve compression, structure remains intact, and signs of feeling return 5 to 11 weeks postsurgery and continue to improve in the next 10 months); and 3) neurotmesis (disruption of the nerve with poor prognosis for return of feeling).18
The first two levels of damage may be caused by bone compression onto the canal during osteotomy preparation or implant placement. Damage to the IAN can occur when the twist drill or implant encroaches, transects, or lacerates the nerve. In the case of sensory alteration caused by nerve compression, slight withdrawal of the implant several turns may alleviate the problem. However, if there is any question of instrument or implant violation of the canal or IAN, the implant should be removed as soon as possible.
This is advisable from both a clinical and medico-legal perspective. Radiographs taken immediately postimplant placement may not facilitate detection of nerve or canal problems unless the implant is clearly in or through the nerve. Clinicians rely on the return of lip, tongue, and gingival feeling, as reported by the patient, to diagnose neurosensory alteration. Frequently, clinicians will not know the extent of the disturbance for days or weeks following the surgery. Consequently, prolonged paresthesia, dysesthesia, or anesthesia may become permanent or require corrective microsurgery. For the latter, “early referral and intervention can be essential to the restoration of sensation after trigeminal nerve injury.”8
Mandibular block anesthesia causes tongue, as well as lip numbness. This is why both were included in the evaluation and considered by the patients when they reported “on complete return of sensation and function.” IAN damage caused by an implant would affect the lip and gingiva sensation. However, to be certain that lingual nerve numbness was not present and reported as paraesthesia, the patients were asked to report when “all numbness” was completely gone.
The use of PM has been shown in this case series to reverse the symptoms of local anesthesia within 25 to 81 mins of injection. The average time of recovery of lip sensation in the present investigation (50.6 mins) compares favorably to the results reported recently in a study of 484 adult and adolescent dental patients (age 12 through 92 years) in which the median recovery time of normal lip sensation in the mandible was 70 mins for PM patients. This was 55% faster than the control. The average time for recovery of normal oral function was 59.9 mins in the present research compared with a median time of 60 mins for patients who received PM injections in the larger reported clinical trial. In the latter study, this represented a 50% faster recovery than the control. 12 If the reversal injections are given prior to or at the time of completion of implant placement, the clinician can be made aware of any neurosensory disturbance at an earlier period than when the local anesthetic is allowed to run its natural course. Both lidocaine with epinephrine and articane with epinephrine provide a duration of pulpal anesthesia of about 60 mins. However, residual soft-tissue anesthesia may persist from 3 to about 5 hours with both drugs.19 When there is prolonged neurosensory disturbance following injection with PM, CBCT or CT scan radiographs are indicated as soon as possible to help the clinician decide whether to completely remove the implant. This procedure, when indicated and if performed in the appropriate period, will allow the best chance for sensory recovery. In cases of nerve compression without nerve disruption, the “backing out” should result in a shorter and more complete recovery.
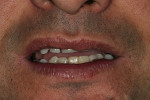
In cases of nerve damage, removal of the causative agent (when the implant is at fault) may allow a clear healing environment with the possibility of nerve regeneration.8 For all patients in the present investigation at whatever the level of recovery, it can be demonstrated medico-legally that the proper sequence of treatment was initiated as early as possible. Often, when clinicians are concerned about the proximity of the implant placement and the IAN position, they may hesitate to use inferior alveolar block anesthesia for fear of decreasing their ability to determine if the implant is too close to the nerve. They may also hesitate to use an IAN block for fear of masking nerve damage. This may compromise the attainment of deep anesthesia during the procedure and cause discomfort for the patient. The use of PM with its ability to reverse the block anesthesia allows the clinician to use this form of injection with a faster recovery of sensation. If there is any sensory dysfunction, this, too, can be deter mined early and treatment rendered, thus reducing the risk of permanent damage (Figure 11 and Figure 12).
The present investigation did not involve controls. This would have required a control group with sham injections of saline or water instead of PM. Because the goal of the present investigation was to determine the efficacy of PM in helping to detect early neurosensory dysfunction to initiate early treatment (while avoiding prolonged nerve damage), it was decided not to use a placebo. The placebo may have placed the patient at risk of permanent paresthesia, which could not be justified in a private clinical practice. Moreover, well-documented studies have been published showing the length of local soft-tissue anesthesia when obtained with lidocaine and articaine and evaluating the effect of PM for reversal of local anesthesia. These studies may be used for comparison when evaluating the results of the current inves tigation. Larger studies, using Internal Review Board protocols, are underway that will more accurately assess time and patient experience differences.
Conclusion
Use of PM has been shown to be effective in reversing the neurosensory effects of local anesthesia in a shorter period (25 to 81 mins) when compared with other research, which demonstrates a 3- to 5-hour range for the expected duration of soft-tissue anesthesia following the same two local anesthetics used in the current study.19-21 This helps the clinician to assess any IAN damage and consider measures to treat the problem at the earliest postsurgical time.
References
1. van Steenberghe D. A retrospective multicenter evaluation of the survival rate of osseointegrated fixtures supporting fixed partial prosthesis in the treatment of partial edentulism. J Pros thet Dent. 1989;61(2):217-222.
2. Jemt T, Lekholm U, Adell R. Osseointegrated implants in the treatment of partially edentulous patients: a preliminary study on 876 consecutively placed implants. Int J Oral Maxillofac Implants. 1989;4(3):211-217.
3. Jemt T, Lekholm U. Oral implant treatment in posterior partially edentulous jaws: a 5-year follow-up report. Int J Oral Maxillofac Implants 1993;8(6):635-640.
4. Buser D, Mericske-Stern R, Bernard JP, et al. Long-term evaluation of non-submerged ITI implants. Part 1: 8-year life table analysis of a prospective multi-center study with 2359 implants. Clin Oral Implant Res. 1997;8(3):161-172.
5. Greenstein G, Tarnow D. The mental foramen and nerve: clinical and anatomical factors related to dental implant placement: a literature review. J Periodontol. 2006;77(12):1933-1943.
6. Dao TT, Mellor A. Sensory disturbances associated with implant surgery. Int J Prosthodont. 1998;11(5):462-469.
7. Wismeijer D, van Waas MA, Vermeeren JI, et al. Patients’ perception of sensory disturbances of the mental nerve before and after implant surgery: a prospective study of 110 patients. Br J Oral Maxillofac Surg. 1997;35(4):254-259.
8. Hegedus F, Diecidue RJ. Trigeminal nerve injuries after mandibular implant placement–practical knowledge for clinicians. Int J Oral Maxillofac Implants. 2006;21(1):111-116.
9. Greenstein G, Cavallaro J, Romanos G, et al. Clinical recommendations for avoiding and managing surgical complications associated with implant dentistry: a review. J Periodontol. 2008;79(8):1317-1329.
10. Day RH. Diagnosis and treatment of trigeminal nerve injuries. J Calif Dent Assoc. 1994;22(6):48-54.
11. Tavares M, Goodson JM, Studen-Pavlovich D, et al; Soft Tissue Anesthesia Reversal Group. Reversal of soft tissue local anesthesia with phentolamine mesylate in pediatric patients. J Am Dent Assoc. 2008;139(8):1095-1104.
12. Hersh EV, Moore PA, Papas AS, et al. Reversal of soft-tissue local anesthetia with phentolamine mesylate in adolescent and adults. J Am Dent Assoc. 2008;139(8):1080-1093.
13. Adell R, Lekholm U, Rockler B, et al. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg. 1981;10(6):387-416.
14. Zarb GA, Schmitt A. The longitudinal clinical effectiveness of osseointegrated dental implants: the Toronto study. Part III: problems and complications encountered. J Prosthet Dent. 1990;64(2):185-194.
15. Kiyak HA, Beach BH, Worthington P, et al. The psychological impact of osseointegrated dental implants. Int J Oral Maxillofac Implants. 1990;5(1):61-69.
16. Ellies LG. Altered sensation following mandibular implant surgery: a retrospective study. J Prosthet Dent. 1992;68(4):664-671.
17. Ellies LG, Hawker PB. The prevalence of altered sensation associated with implant surgery. Int J Oral Maxillofac Implants.1993;8(6):674-679.
18. Jalbout Z, Tabourian G. Glossary of implant dentistry. Upper Montclair, NJ: International Congress of Oral Implantologists; 2004:16.
19. Malamed SF. Local anesthetics: dentistry’s most important drugs, clinical update 2006. J Calif Dent Assoc. 2006;34(12):971-976.
20. Malamed SF, Gagnon S, Leblanc D. Efficacy of articaine: a new amide local anesthetic. J Am Dent Assoc. 2000;131(5)635-642.
21. Laviola M, McGavin SK, Freer GA, et al. Randomized study of phentolamine mesylate for reversal of local anesthesia. J Dent Res. 2008;87(7):635-639.