Prebrushing with a Potassium Nitrate Dentifrice to Reduce Tooth Sensitivity During Bleaching Evaluated in a Practice-Based Setting
William D. Browning, DDS, MS; Van B. Haywood, DMD; Nathan Hughes, BSc; and Rafael Cordero, DDS
Abstract
This study compared subjective occurrence of tooth sensitivity associated with tooth bleaching in patients who brushed with potassium nitrate toothpaste prior to and during a home tooth bleaching procedure to those who brushed with standard fluoride toothpaste. Methodology: This was a randomized, field-based, multicenter, two-arm, parallel-group, open-label study comparing the occurrence of tooth sensitivity (a visual analogue scale and questionnaire) associated with tooth bleaching. Also, patient satisfaction, shade change (whitening), and safety were compared. Results: During the first 7 days of bleaching, participants in the Sensodyne® Fresh Mint toothpaste group averaged less sensitivity than those in the Crest® Cavity Protection toothpaste group. However, the observed difference was not statistically significant (P = .2883). Similar results were observed for the various periods analyzed: days 1 to 3 (P = .348), days 1 to 14 (P = .405), days 4 to 7 (P = .309), and days 8 to 14 (P = .681). Significant differences did emerge, however, in patient perceptions of the treatment: Significantly more people were satisfied (P = .0125) with the whitening treatment in terms of sensitivity experienced. Significantly fewer people reported sensitivity (P = .0192) while undergoing bleaching. There were no statistically significant differences in VITA® Classical shade guide change improvement between the two groups (P > .05). There were no reported treatment-related adverse effects. Conclusion: The results indicate that the use of potassium nitrate toothpaste before and during bleaching treatment may reduce tooth sensitivity. Brushing with potassium nitrate toothpaste did not adversely affect bleaching efficacy.
Transient tooth and gingival sensitivity occur commonly with tooth bleaching.1,2 Recently, the addition of desensitizing agents have been shown to provide some reduction of both gingival and tooth sensitivity.3-5 However, many tooth-bleaching agents do not contain desensitizing materials. Potassium nitrate-containing toothpastes have been used in conjunction with tooth bleaching under these circumstances and found to be successful at reducing sensitivity levels.6,7
This study compared tooth sensitivity in two groups of participants undergoing the same bleaching regimen but using different commercially available dentifrices before and after bleaching. Both groups used the same tooth whitening agent, which was a commercially available 14% hydrogen peroxide-containing whitening strip. One group used a fluoride and potassium nitrate-containing dentifrice, and the other used a standard fluoride, nonsensitivity dentifrice. Tooth sensitivity was measured with a visual analogue scale (VAS). The hypothesis was that average VAS scores for the first 7 days of bleaching would be lower in the potassium nitrate group. This was tested against the null hypothesis that there would be no difference between groups at the 5% significance level. A secondary objective was to compare patient satisfaction in the two groups, using a posttreatment questionnaire. The hypothesis that satisfaction scores would be higher in the potassium nitrate group was tested against the null hypothesis that there would be no difference. As these were secondary objectives, each test was performed at a 5% significance level and reported as observations.
Methods and Materials
This was a randomized, controlled, multisite, parallel group study involving 14 sites. The study design and the informed consent process were approved by an independent review board (IRB) prior to the start of the study. A total of 14 centers were included, with a goal of enrolling between 12 and 20 people each.
At a screening appointment, participants were informed about the study and asked for written informed consent. Inclusion criterion was an age older than 21 years and a desire for teeth whitening. The procedure had no fee, and those deemed eligible were randomly assigned to receive either the potassium nitrate dentifrice (Sensodyne® Fresh Mint toothpaste GlaxoSmithKline, https://www.sensodyne.com) or the standard fluoride, nonsensitivity family dentifrice (Crest® Cavity Protection toothpaste, Procter & Gamble, https://www.crest.com). An evaluation of the oral soft tissue (OST) was completed. Participants were asked to rate their level of existing tooth sensitivity on a 100-mm line. This VAS had two descriptors. The left edge of the line was labeled “no sensitivity” while the right was labeled “extreme sensitivity.” Finally, they were provided with their assigned toothpaste and instructions, which were to apply a 1-inch strip of toothpaste to cover the head of the toothbrush and brush for 1 min twice daily: once in the morning and again in the evening.
After 14 days of the brushing protocol, they returned for the bleaching baseline visit in which the OST evaluation was completed and participants again assessed their present level of tooth sensitivity. Baseline tooth color was established using the VITA Classical shade guide (Vident™, https://www.vident.com). Only the maxillary right central incisor was matched to the shade guide. Tooth color was tracked only to assess any possible effect of the potassium nitrate dentifrice on the bleaching process. Participants were provided with a home bleaching pack and instructions. Crest Whitestrips® Supreme (Procter & Gamble), a strip-delivery whitener containing 14% hydrogen peroxide, was assigned to both groups. Wear was for 30 mins twice daily with at least 3 hours between applications. Participants were also provided with a diary card to record bleaching sensitivity and other product attributes and asked to complete it each day during active bleaching.
Active tooth bleaching occurred during the next 14 days. Participants used their assigned toothpaste during active bleaching. Immediately after this period, participants returned to their respective clinics for the final evaluation appointment. At this posttreatment appointment, the OST examination and the Vita shade color were measured. Bleaching diaries were collected with all used and unused whitening products. Within 7 days of completing the final visit, patients completed a patient satisfaction survey (PSS) via the telephone.
The PSS included two questions that were relevant to the level of sensitivity experienced by participants. For one question, the response consisted of a five-point ordinal scale, ie, extremely satisfied, very satisfied, somewhat satisfied, not very satisfied, and not at all satisfied; while the other called for a binomial response, ie, yes or no. With five categories in the ordinal scale question, it was considered appropriate to treat the response as a continual measure and to compare the mean score between the treatment groups, using an analysis of variance (ANOVA). The binomial response was analyzed using a logistic regression procedure.
Average VAS scores for the first 7 days of active bleaching were used to calculate the mean difference between the two treatment groups. An analysis of covariance (ANCOVA) with treatment and center as effects was used to compare the groups. In this analysis, the prebleaching VAS score was also included as a covariate. Mean VAS scores for the standard family dentifrice group were compared with the potassium nitrate dentifrice group with a positive mean difference indicating a higher mean VAS score for the potassium nitrate dentifrice group.
Results
Each of the 14 study sites enrolled 11 to 17 participants, totaling 184. Overall, males comprised 41% (75) of the study participants and females 59% (109). The average age of all participants was 30 years with the potassium nitrate dentifrice group averaging 30 years and the standard fluoride containing family dentifrice group 31 years. Respectively, the youngest and oldest participants were 22 and 37 years old.
In all, 180 participants (93 potassium nitrate dentifrice, 87 standard family dentifrice) were included in the intent-to-treat analysis. Four failed to attend any appointments beyond the baseline. One was from the potassium nitrate group and three from the standard family group.
On day 7, the mean treatment difference was -2.3 mm with a 95% confidence interval of -6.5 mm to -1.9 mm indicating that the potassium nitrate group had lower VAS scores on average than the standard family dentifrice group. This difference, however, was not statistically significant (ANCOVA; P = .29). Mean treatment differences for this primary outcome and all other intervals during the active bleaching phase are listed in Table 1. Table 2 lists the mean VAS scores for each group at every evaluation period. While both tables depict a clear data trend that indicates the potassium nitrate group experienced lower VAS scores, as was the case with the primary comparison of interest, none of the periods compared were statistically significant.
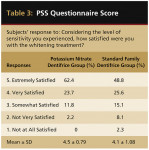
The PSS asked participants if they had experienced tooth sensitivity. In the potassium nitrate and standard family groups, 49.5% and 66.3% responded positively, respectively. A logistic regression procedure was performed, using treatment group and centers as effects. The difference between groups was significant (logistic regression; P = .019). The second question asked about patient satisfaction, considering the level of sensitivity that was experienced. This question used an ordinal scale with five choices, with 1 representing “not at all satisfied” and 5 representing “extremely satisfied” (Table 3). There was a statistically significant difference between groups with participants in the potassium nitrate group expressing more satisfaction (ANOVA; P = .013). The mean treatment difference was 0.36 with a 95% confidence interval of 0.08 to 0.64.
The color difference from baseline to the end of the study was 3.0 and 3.1 tabs for the potassium nitrate and standard family groups, respectively. This represents positive tooth whitening for both groups, with no significant difference between the two groups (ANCOVA; P = .653).
Discussion
Wide variation was observed between participants regarding the level of sensitivity as recorded using the VAS. This is similar to the findings in two reviews of bleaching studies.8,9 The differences between the two treatments were consistently small.
The amount of sensitivity recorded in this study was extremely small, and therefore, the ability of the study to differentiate between groups was limited. The VAS data did not show significant differences in tooth sensitivity levels among the two groups at any time. However, a statistically significant difference between the participants’ report of sensitivity on the PSS was noted. These data are different from the VAS data in important ways. The VAS data are based on daily assessment of tooth sensitivity whereas the PSS asks the participant to make a global assessment of his or her overall experience following the end of the study. The VAS was presented as a 100-mm line with potential responses from 0 to 100. Accordingly, some participants may have inferred that they were being asked to make a very fine determination. They may have reported sensitivity levels that they personally considered inconsequential. The responses available to the PSS question about sensitivity were broader and thus likely to allow participants latitude to decide importance regarding what to report. This is especially true of the PSS question regarding satisfaction level after considering the sensitivity experienced. It should also be noted that both these questions were asked during the phone call that occurred 7 days following the end of treatment. As a result, the answers may have been affected by the participants’ ability for accurate recall.
The present study was conducted within the community (practice-based) as opposed to an academic research center. Academic studies typically involve a single or small number of investigators. Using a smaller number of investigators makes it easier to gain agreement on study procedures, calibrate examiners, etc. In short, it is easier to develop consistency among the evaluators and control all aspects of the study more closely. These steps help ensure that the outcomes observed are truly as a result of the research intervention(s) and not differences between evaluators or other factors unrelated to the study procedures. All of this creates a more scientifically sound study. In contrast to these positive aspects, there are concerns. A study that involves just a few operators may be greatly affected by the skills and knowledge of the practitioners involved. A single operator with superior or inferior skills may lead to a study result unrelated to what “most” practitioners would achieve. Similarly, a researcher who has ample time to practice a procedure preceding the research and to judiciously follow a complicated set of steps during a clinical trial may be able to achieve a result that is impractical for practitioners. These issues lower the relevance of such tightly controlled studies to a clinical practice setting.
By contrast, a study that involved a large number of investigators at multiple, unrelated sites (practice or otherwise) makes it difficult to obtain rigorous control from a scientific standpoint but would align with the current concept of evidence-based research for general practices. There would be limited opportunity to calibrate study personnel in terms of procedural technique, precise measurements, etc. A study involving a large number of practitioners is likely to be representative of most practitioners. Similarly, procedures would be completed under conditions relevant to dental practice. All study outcomes were self-assessments made by the participants, so potential differences between dentists regarding evaluations were minimized and controlled. The dentists in this study had a limited role. They performed oral soft-tissue examinations aimed at ensuring the safety of the treatments. Accordingly, the study reported herein combined some of the best elements of both the study designs discussed above.
The present results are very similar to a previous study7 with the same trend noted at each of the three evaluation periods examined. In addition, patients using the potassium nitrate toothpaste had fewer days of sensitivity and the onset of sensitivity was delayed relative to that in the control group. As with the present study, after considering the level of sensitivity experienced, participants in the potassium nitrate group were significantly more satisfied with their treatment.
The bleaching material was self applied as a 14% hydrogen peroxide on a strip, also different from the previous study, which used a 9% hydrogen peroxide product containing potassium nitrate and delivered in a custom-made tray. Because both the material in this study and the previous study are hydrogen peroxide products, they are designed for day wear. The delivery methods were thus very different between the studies (tray vs strip). Contact time and the absolute amount of hydrogen peroxide available significantly differed, which may result in disparities in efficacy and sensitivity. Using a single bleaching product in this study allowed the variable of the dentifrice to be evaluated but did not easily allow comparisons with other studies on color changes.
Measurement of color change during bleaching with the Vita Classical shade guide would be one issue in which the decision to conduct this study within the community presents a potential problem. The number of practitioners involved and the fact that the practitioners operated independently might lead a critical reader to question the reliability of this measure. It is important to recall that the focus of this measure is on the relative performance of the two groups. The purpose was to determine if the potassium nitrate had any deleterious effect on the effectiveness of the whitening agent. As such, there is no intent to compare the absolute size of the color change to any previous studies. Each evaluator made color measurements of participants in both groups, and any measurement errors were likely to occur in both groups equally. Given the fact that the color change for the two groups was significant and almost identical despite one group using the potassium nitrate dentifrice. There is little evidence to support the conclusion that the efficacy of tooth bleaching was affected.
Conclusion
Within the limitations of the study, use of a toothpaste with potassium nitrate before and during tooth bleaching has the potential to reduce sensitivity without affecting the effectiveness of the whitening product.
Acknowledgments
The authors would like to thank Kimberley Clark, Farzana Sufi, John Durocher, and Joanne Liebman for their contribution to this study.
Disclosure
This study was sponsored by GlaxoSmithKline Consumer Healthcare and was performed by a contract research organization in a practice-based setting in Florida with Dr. Cordero (Principal Investigator).
References
1. Haywood VB. Dentine hypersensitivity: bleaching and restorative considerations for successful management. Int Dent J. 2000;52:376-384.
2. Haywood VB. Frequently asked questions about bleaching. Compend Cont Edu Dent. 2003;24(4A):324-338.
3. Pohjola RM, Browning WD, Hackman SH, et al. Sensitivity and tooth whitening agents. J Esthet Restor Dent. 2002;14(2):85-91.
4. Callan RS, Browning WD, Downey MC, et al. Comparison of two low sensitivity whiteners. Am J Dent. 2008;21(1):17-20.
5. Browning WD, Chan DC, Myers ML, et al. Comparison of traditional and low sensitivity whiteners. Oper Dent. 2008;33(4):379-385.
6. Haywood VB, Caughman WF, Frazier KB, et al. Tray delivery of potassium nitrate-fluoride to reduce bleaching sensitivity. Quintessence Int. 2001;32(2):105-109.
7. Haywood VB, Cordero R, Wright K, et al. Brushing with a potassium nitrate dentifrice to reduce bleaching sensitivity. J Clin Dent. 2005;16(1):17-22.
8. Browning WD, Blalock JS, Frazier KB, et al. Duration and timing of sensitivity related to bleaching. J Esthet Restor Dent. 2007;19(5):256-264.
9. Browning WD. Critical appraisal. Comparison of the effectiveness and safety of carbamide peroxide whitening agents at different concentrations. J Esthet Restor Dent. 2007;19(5):290-297.
About the Authors
William D. Browning, DDS, MS
Professor and IDA Endowed Chair in Restorative Dentistry
Indiana University, School of Dentistry
Indianapolis, Indiana
Van B. Haywood, DMD
Professor, Department of Oral Rehabilitation
Medical College of Georgia, School of Dentistry
Augusta, Georgia
Nathan Hughes, BSc
Clinical Research Scientist
GlaxoSmithKline Consumer Healthcare
Weybridge, Surrey, United Kingdom
Rafael Cordero, DDS
Principal Coordinating Investigator
Applied Clinical Research, LLC
West Palm Beach, Florida