Computer-Generated Implant Planning and Surgery: Case Selection
Gary Orentlicher, DMD; Douglas Goldsmith, DDS; Andrew Horowitz, DMD, MD
Implant dentistry is in the midst of a revolutionary change. Medical-grade computed tomography (CT) scanners and in-office cone-beam scanners allow the surgeon to see a patient’s anatomy in three dimensions. Visualization of the available bone for implant placement, soft-tissue thicknesses, and location of the maxillary sinuses and pertinent vital structures, such as the mandibular canal, are possible.1-3 The images can be imported into proprietary software programs (eg, Simplant® [Materialise Dental NV, Leuven, Belgium], Procera® [Nobel Biocare, Göteborg, Sweden], EasyGuide™ [Keystone Dental, Burlington, MA], Facilitate™ [Astra Tech, Mölndal, Sweden]) that allow the surgeon to “virtually” place implants. The type and size of the planned implant; its position within the bone; its relationship to the planned restoration and adjacent teeth and/or implants; and its proximity to the mandibular canal, incisive canal, and maxillary sinuses can be determined before surgery.1-5 Computer-generated drilling guides then can be fabricated from the virtual treatment plan. These surgical guides enable the clinician to surgically place the planned implants, in the patient’s mouth, in the same positions as planned on the virtual treatment plan. This process permits more accurate and predictable implant placement6-10 and reduces patient morbidity.11-14
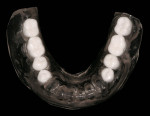
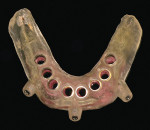
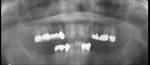
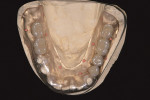
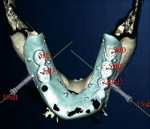
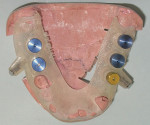
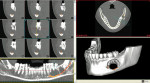
All of the current systems have similar protocols. First, the surgeon creates a prosthesis for the patient to wear during the CT scan. This is a partial or full denture (termed the scan prosthesis) that duplicates the planned final restorations (Figure 1a; Figure 1b and Figure 1c). The CT scan then is taken with the patient wearing the prosthesis. Next, the images are imported into one of the software programs (eg, Simplant, Procera, EasyGuide), which is used to virtually place implants into their ideal positions related to the planned restoration and underlying bone (Figure 2a; Figure 2b and Figure 2c). Then, the surgeon electronically sends the virtual treatment plan to the software company, which fabricates a surgical guide (Figure 3a ; Figure 3b and Figure 3c). With this guide, the surgeon uses implant-specific drilling instrumentation to place the implants precisely according to his or her virtual treatment plan.
Virtual treatment planning and computer-generated drilling guides benefit the patient by enabling flapless surgery, reduced surgical time, less discomfort and swelling, and a quicker return to life and work.6,11-19 These technologies help the surgeon by reducing chair time, lessening the patient’s stress at the time of surgery, facilitating an accurate means of placing dental implants, and reducing the risk for surgical complications.7-10,20 The potential for mistakes is greatly alleviated.
Computerized implantology can be used for any implant case. The increased time for treatment planning and the increased expense may outweigh its benefits in certain cases. In the authors’ practice, these techniques are the appropriate means of treatment for many cases, including those with:
- questionable bone volume,
- problems related to proximity of adjacent teeth,
- proximity to vital anatomic structures,
- three or more adjacent implants,
- placement of a multiple-unit or full-arch immediate restoration,
- significant alteration of the soft tissue or bony anatomy by prior surgery or trauma, and
- medical comorbidities.
This article will not only illustrate cases in which these techniques benefit the surgeon and patient, but it will also help dentists determine those cases in which these techniques are the appropriate means of treatment.
Planning for three or more adjacent implants and fully edentulous arches
When placing a dental implant, the surgeon commonly uses a surgical guide that is fabricated on articulated patient models. These types of guides can be simple (eg, vacuforms with the buccal or palatal/lingual facings of the planned restorations) or more complex (eg, 2-mm drill holes or metal tubes in the middle of hard acrylic appliances that reproduce a planned restoration). The one commonality of these appliances is their lack of correlation between the planned restoration and underlying bony anatomy. Only with virtual implant surgical guides can this anatomic relationship be established predictably and considered prior to surgery.
A surgical guide for implant placement is fabricated based on the patient’s anatomy and local references, such as the numbers and locations of teeth in the arch to be treated or in the opposing arch. As the length of the edentulous area increases, the surgeon has fewer anatomic references to accurately predict implant placement. When the case is fully edentulous, all local references, other than the soft-tissue ridge, are absent. Additionally, as the bone and soft tissue are lost from periodontal disease, bony atrophy, sinus pneumatization, and long-term denture wear, it becomes more difficult to predictably use a traditional surgical guide.
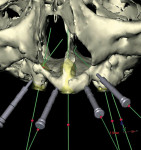
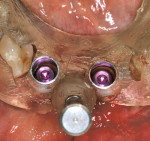
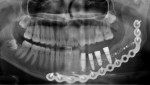
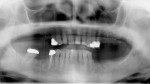
Because of these issues, virtual implant evaluation and treatment planning and implant placement using computer-generated guides is an ideal technology for most cases in which three or more adjacent implants are planned (Figure 4a; Figure 4b and Figure 4c). The spacing and angulation relationships of multiple planned implants, as well as implant to bone relationships, all can be established before surgery. Implants can be placed flaplessly and immediately loaded6,11-19 (Figure 5a; Figure 5b and Figure 5c).
Nerve proximity close to the planned implant position
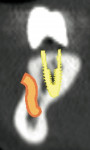
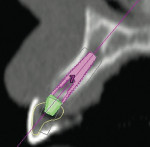
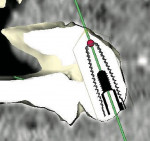
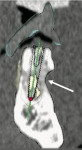
Panoramic and periapical radiographs are 2-dimensional representations of a patient’s 3-dimensional (3D) anatomy. Differences in x-ray machines and radiographic techniques commonly lead to distorted images, such as elongation and shortening of anatomic structures. Accurate evaluation and measurement of the relationship between the position of the mental nerve, the inferior alveolar nerve, or the nasopalatine/incisive nerve and the location of the planned implant can only truly be determined and planned by a 3D evaluation of the patient’s anatomy as it relates to the planned restoration3 (Figure 6a; Figure 6b ). These anatomic relationships are best visualized, evaluated, and measured by using CT images. Placement of implants when there is a question of nerve or sinus proximity to the patient’s available bone is most accurately performed using virtual implant treatment planning and implant placement with computer-generated surgical guides. With these techniques, the potential for patient morbidity because of possible implant placement in close proximity to vital structures is minimized (Figure 7; Figure 8 and Figure 9).
Questionable bone volume
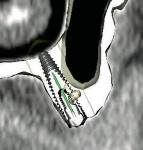
Frequently, dental implant surgeons confront surgical dilemmas that require the placement of dental implants in only one possible location and/or at only one possible implant depth. At times, surgeons are required to place dental implants into tight spaces with minimal bony leeway—mesial-distally, buccal-lingually, or both (Figure 10a; Figure 10b; Figure 10c; Figure 10d; Figure 11a; Figure 11b; Figure 11c; Figure 11d; Figure 11e; Figure 11f; Figure 11g; Figure 11h and Figure 11i). Proximity of adjacent tooth roots can require the surgeon to “thread the needle” with the implant placement. This is often a problem with congenitally missing teeth. Clinical situations of limited bone volume often leave the clinician with scenarios in which the patient’s anatomy dictates the location of implant placement, sometimes leaving only one available location.
3-D planning software allows for segmentation, enabling the virtual removal of surrounding bone tissues, leaving only the crowns and roots of the teeth visible. This visualization of the roots of the adjacent teeth enables the precise positioning of an implant in limited mesial-distal spaces. Selection of the best implant for that specific clinical application then can be determined. Some software allows for the selection and determination of virtual stock and custom abutments, as well. Implant placement then can be performed predictably and accurately according to the patient’s anatomy. Because of their precision and accuracy, 3D computer-generated technologies are ideally suited for these difficult cases.21
Flapless Surgery and Multiple-Unit Immediate Placement/Immediate-Load Cases
Today’s implant technologies and surface characteristics have dramatically shortened the time required from implant placement to implant loading. Immediate placement and immediate loading of implants is now common in many dental surgical offices. In some cases, teeth can be extracted, an implant immediately placed, and a temporary crown placed at the time of implant insertion. Concepts of cross-arch stabilization of implants and loading of multiple implant cases have changed the way many dentists approach treatment plans. These new technologies are available to place single, multiple, or a full arch of implants flaplessly, without making an incision or elevating a tissue flap. Patients experience less surgical trauma, pain, and swelling. Recovery time is reduced, and the ability to return to normal life is expedited.6,11-19
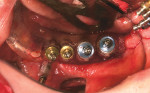
The utilization of virtual implant treatment planning allows the dentist to evaluate the patient’s bony anatomy as it relates to the planned restoration and then plan the ideal placement and position of a dental implant in that clinical situation. Computer-generated stereolithographic surgical guides are then fabricated from these virtual treatment plans. A dental laboratory then uses the surgical guide, along with mounted patient models, to fabricate temporary, and in some cases, final restorations, before implant placement surgery. At the time of surgery, the surgeon can use this guide to place the planned implants flaplessly, removing only a core of tissue in the planned implant sites. Abutments are then immediately placed on the implants, and temporary, or in some cases final, restorations are inserted (Figure 12a; Figure 12b; Figure 12c; Figure 12d; Figure 12e; Figure 12f; Figure 12g; Figure 12h; Figure 12i; Figure 12j; Figure 12k and Figure 12l).
Depending on the clinical circumstances and the dentist’s experience and comfort level, the technologies can be used to place implants as a two-stage, a single-stage with healing abutments, or an immediate-placement/immediate-load case. Implants can be positioned accurately with a tissue incision or flaplessly.7-10,20
Complex problems with a significant alteration of the bony anatomy
Implant dentistry is shifting from the days of “placing the implants where the bone is.” Current technologies allow the surgeon to do various soft-tissue and bone procedures to prepare the planned implant site. Sinus lift grafts, block bone grafts, ridge splitting, and alveolar distraction procedures, as well as large and small soft-tissue and connective-tissue grafts, are a few of the procedures that are performed routinely to prepare the recipient jaw site for implant placement. Previous surgical procedures, including the placement of many different types of dental implants (eg, blade and subperiosteal), can leave patients with challenging reconstructive bony defects (Figure 11a; Figure 11b; Figure 11c; Figure 11d; Figure 11e; Figure 11f; Figure 11g; Figure 11h; Figure 11i). Patients may have lost bone, teeth, and soft tissue from traumatic injuries (Figure 14a; Figure 14b; Figure 14c; Figure 14d; Figure 14e; Figure 14f and Figure 14g). Bony defects of various sizes can occur because of benign or malignant pathology of the jaws. Reconstructive surgical procedures to treat benign or malignant diseases can leave areas of abnormal bony anatomy and scarred soft tissue (Figure 15a; Figure 15b; Figure 15c; Figure 15d; Figure 15e; Figure 15f; Figure 15g; Figure 15h and Figure 15i).
After healing, graft maturation, and settling of graft materials, resultant bone and soft-tissue volumes can be unpredictable. Lateral block onlay grafts can resorb a portion of their bone volume before implant placement21-23 (Figure 13a and Figure 13b). In sinus-lift grafting, CT technology permits the surgeon to predict the volume of graft material necessary to augment the area to a desired height of bone.24 In alveolar distraction procedures, the shape and width of the new distracted bone can vary. Evaluating these areas with conventional panoramic or periapical radiographs is inadequate; a 3D analysis can facilitate a better assessment.
These are all examples of ideal situations in which computer-guided implant surgery should be used to evaluate and plan the placement of dental implants. These technologies allow visualization and evaluation of a patient’s distorted anatomy without making an incision or removing any soft tissue or bone.25 The surgeon can then accurately and predictably place the dental implants with clear knowledge of the patient’s underlying distorted bony anatomy as it relates to the planned dental restoration. Additionally, dental implants can be placed flaplessly, without making an incision and removing the periosteal vascularization from grafted areas.
Patients with medical comorbidities
Patients with head and neck cancers are often treated with pre- and postoperative radiation therapy, which potentially may alter healing capacity. Research and treatment protocols call for patients who received radiation therapy to undergo pre- and postoperative courses of hyperbaric oxygen therapy (HBO) to increase the vascularity of the bone prior to implant placement.26-29 To limit the likelihood of such patients developing osteoradionecrosis of the jaws, placing implants with minimal flap elevation and soft- and hard-tissue trauma is indicated.26,30 Bleeding, swelling, and alteration of bone and soft-tissue vascularization are minimized by using these technologies31 (Figure 16a; Figure 16b; Figure 16c; Figure 16d; Figure 16e; Figure 16f and Figure 16g).
Some patients may have associated medical problems, such as blood dyscrasias, anticoagulation problems, or significant cardiovascular disease. These issues may necessitate specific medication protocols that cannot be adjusted prior to surgery. Limiting surgical trauma to the soft and hard tissues, thus minimizing bleeding, is indicated in the management of these patients.
3D implant evaluation and planning with CT-guided implant placement allows the surgeon to place implants flaplessly, with precision.6,11-19 Implants can be placed accurately while minimizing soft- and hard-tissue trauma as much as possible.7-10 These technologies are ideally indicated for use in patients with these challenging medical management problems.31
Patients with emotional and physical problems
Dental patients come to offices with different historical experiences, most good, but sometimes bad. Their stress, anxiety, and phobias can prevent them from undergoing procedures that require long periods in a dental chair. Orthopedic and spinal problems may limit how long a patient can sit. Additionally, wheelchair-bound patients pose another set of logistical problems.
Patients such as these can require extensive planning and preparation prior to surgery, which must be performed quickly and efficiently, without compromising quality. With the use of 3D computer-generated implant planning, most anatomic issues that may be encountered during surgery are visualized before the patient is in the dental chair. By using surgical guides for implant placement, implants can be placed quickly and predictably, thus minimizing the patient’s stress, pain, and time in the dental chair. 3-D virtual technologies are well suited for use in patients with these unusual management issues.6-20
Conclusion
3D virtual implant planning and placement using surgical guides allows the surgeon to plan and place dental implants according to a restoratively driven treatment plan. Patients’ underlying bony anatomy and vital structures, as related to the planned dental restorations, are determined before surgery, thus decreasing potential patient morbidity. Surgical guides allow the placement of implants, with precision and accuracy, according to a predetermined plan. If desired, implants can be placed flaplessly, with predictability. The use of these technologies in planning and placing dental implants greatly benefits the patient and clinician. These technologies are ideal for use in the following cases: the placement of three or more adjacent implants; fully edentulous arches; when implants are planned in close proximity to nerves, teeth, and other vital anatomic structures; immediate-placement and immediate-load cases; complex cases with questionable bone volume and unusual bony contours and anatomy; difficult medical comorbidities; and patients with physical and emotional issues.
Disclosure
Dr. Orentlicher has received honorariums from Nobel Biocare and Materialise Dental. Dr. Goldsmith is a consultant for Keystone Dental and Materialise Dental and has received honorariums from Nobel Biocare and Materialise Dental.
References
1. Ramez J, Donazzan M, Chanavaz M, et al. The contribution of scanner imagery in implant surgery and sinus overflow using frontal oblique orthogonal reconstruction. Rev Stomatol Chir Maxillofac. 1992;93(3):212-214.
2. Pattijn V, Van Cleynenbreugel T, vander Sloten J, et al. Structural and radiological parameters for the nondestructive characterization of trabecular bone. Ann Biomed Eng. 2001;29(12): 1064-1073.
3. Sonick M, Abrahams J, Faiella R. A comparison of the accuracy of periapical, panoramic, and computerized tomographic radiographs in locating the mandibular canal. Int J Oral Maxillofacial Impl. 1994;9(4);455-460.
4. Todd AD, Gher ME, Quintero G, et al. Interpretation of linear and computed tomograms in the assessment of implant recipient sites. J Periodontol. 1993;64(12):1243-1249.
5. Gehr ME, Richardson AC. The accuracy of dental radiographic techniques used for evaluation of implant fixture placement. Int J Periodontics Restorative Dent. 1995;15(3):268-283.
6. van Steenberghe D, Glauser R, Blombäck U, et al. A computed tomographic scan-derived customized surgical template and fixed prosthesis for flapless surgery and immediate loading of implants in fully edentulous maxillae: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1): S111-S120.
7. Tardieu P, Vrielinck L. Implantologie assistèe par ordinateur: le propramme SimPlant/SurgiCase™ et le SAFE System™ mis en charge immediate d’un bridge mandibulaire avec des impalt transmuqueux. Implant. 2003;9:15-28.
8. Rosenfeld AL, Mandelaris GA, Tardieu PB. Prosthetically directed implant placement using computer software to ensure precise placement and predictable prosthetic outcomes. Part 3: ste-reolithographic drilling guides that do not require bone exposure and the immediate delivery of teeth. Int J Perio Rest Dent. 2006;26(5):493-499.
9. Vrielinck L, Politis C, Schepers S, et al. Image-based planning and clinical validation of zygoma and pterygoid implant placement in patients with severe bone atrophy using customized drill guides. Preliminary results from a prospective clinical follow-up study. Int J Oral Maxillofac Surg. 2003;32(1):7-14.
10. Sarment DP, Sukovic P, Clinthorne N. Accuracy of implant placement with a stereolithiographic surgical guide. Int J Oral Maxillofac Implants. 2003;18:571-577.
11. Hahn, J. Single-stage, immediate loading, and flapless surgery. J Oral Implantol. 2000;26(3): 193-198.
12. Campelo LD, Camara JR. Flapless implant surgery: a 10-year clinical retrospective analysis. J Oral Maxillofac Implants. 2002;17(2):271-276.
13. Becker W, Goldstein M, Becker BE, et al. Minimally invasive flapless implant surgery: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(suppl 1):S21-S27.
14. Becker W, Wikesjö UM, Sennerby L, et al. Histologic evaluation of implants following flapless and flapped surgery: a study in canines. J Periodontol. 2006;77(10):1717-1722
15. Tardieu PB, Vrielinck L, Escolano E. Computer-assisted implant placement. A case report: treatment of the mandible. Int J Oral Maxillofac Implants. 2003;18(4):599-604.
16. van Steenberghe D, Naert I, Andersson M, et al. A custom template and definite prosthesis allowing immediate implant loading in the maxilla: a clinical report. Int J Oral Maxillofac Implants. 2002;17(5):663-670.
17. van Steenberghe D, Ericsson I, Van Cleynenbreugel J, et al. High precision planning for oral implants based on 3D CT scanning. A new surgical technique for immediate and delayed loading. Appl Osseo Int Res. 2004;4;27-30.
18. Wendelhag I, van Steenberghe D, Blombäck U, et al. Immediate function in edentulous maxillae with flapless surgery including a 3-D CT-scan based treatment planning procedure. [Abstract. Poster 144] Clin Oral Implants Res. 2004.
19. Sanna AM, Molly L, van Steenberghe D. Immediately loaded CAD-CAM manufactured fixed complete dentures using flapless implant placement procedures: a cohort study of consecutive patients. J Prosthet Dent. 2007;97(6):331-339.
20. Orentlicher GP, Goldsmith D, Horowitz A. “Virtual” implant treatment planning—the future of implant dentistry? Inside Dentistry. 2008;4(1):44-47.
21. Johansson B, Grepe A, Wannfors K, et al. A clinical study of changes in the volume of bone grafts in the atrophic maxilla. Dentomaxillofac Radiol. 2001;30(3):157-161.
22. Verhoeven JW, Ruijter J, Cune MS, et al. Onlay grafts in combination with endosseous implants in severe mandibular atrophy: one year results of a prospective, quantitative radiological study. Clin Oral Implants Res. 2000;11(6):583-594.
23. Smolka W, Eggensperger N, Carollo V, et al. Changes in the volume and density of calvarial split bone grafts after alveolar ridge augmentation. Clin Oral Implants Res. 2006;17(2):149-155.
24. Krennmair G, Krainhöfner M, Maier H, et al. Computerized tomography-assisted calculation of sinus augmentation volume. Int J Oral Maxillofac Implants. 2005;21(6):907-913.
25. Orentlicher GP, Goldsmith D, Horowitz A. Thinking out of the box—the use of “virtual” implant treatment planning and surgery in challenging cases. Inside Dentistry. 2008;4(6): 58-64.
26. Marx RE. Osteoradionecrosis: a new concept of its pathophysiology. J Oral Maxillofac Surg. 1983;41(5):283.
27. Marx RE, Ames JR. The use of hyperbaric oxygen therapy in bony reconstruction of the irradiated and tissue-deficient patient. J Oral Maxillofac Surg. 1982;40(7):412-420.
28. Granström, G. Placement of dental implants in irradiated bone: the case for using hyperbaric oxygen. J Oral Maxillofac Surg. 2006;64(5):812-818.
29. Granström G. Radiotherapy, osseointegration, and hyperbaric oxygen therapy. Periodontol. 2000;33: 145-162.
30. Koga DH, Salvajoli JV, Alves FA. Dental extractions and radiotherapy in head and neck oncology: review of the literature. Oral Dis. 2008;14(1): 40-44.
31. Horowitz A, Orentlicher GP, Goldsmith DH. Case report: CT guided implant surgery in the head and neck cancer patient. J Oral Maxillofac Surg. 2009;67(3):619-623.
About the Authors
Gary Orentlicher, DMD
Private Practice
Scarsdale, New York
Douglas Goldsmith, DDS
Private Practice
Scarsdale, New York
Andrew Horowitz, DMD, MD
Private Practice
Scarsdale, New York